Chlamydia LPS antibody | CF 6J12



Mouse anti Chlamydia LPS
- Product Type
- Monoclonal Antibody
- Clone
- CF 6J12
- Isotype
- IgG2a
- Specificity
- Chlamydia LPS
| Mouse anti Chlamydia LPS antibody, clone CF 6J12 recognizes a genus specific epitope within Chlamydia lipopolysaccharide (LPS). LPS is a common feature of the outer envelope of gram negative bacteria, which acts as a potent endotoxin, triggering an innate immune response. Whilst the LPS of Chlamydia trachomatis does evoke an immune response, it displays only weak endotoxic activity when compared to that of other bacteria such as Salmonella minnesota or Neisseria gonorrhoeae (Ingalls et al. 1995). |
- Target Species
- Bacterial
- Species Cross-Reactivity
-
Target Species Cross Reactivity Chlamydophila sp. - N.B. Antibody reactivity and working conditions may vary between species.
- Product Form
- Purified IgG - liquid
- Preparation
- Purified IgG prepared by affinity chromatography on Protein A from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline.
- Preservative Stabilisers
- <0.1% Sodium Azide (NaN3)
- Immunogen
- Elementary bodies from C. trachomatis strain SAF2.
- Approx. Protein Concentrations
- IgG concentration 1.0mg/ml
- Fusion Partners
- Spleen cells from immunised BALB/c mice were fused with cells of the NS0/U mouse myeloma cell line.
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| ELISA | |||
| Immunofluorescence | |||
| Western Blotting |
Source Reference
-
Thornley, M.J. et al. (1985) Properties of monoclonal antibodies to the genus-specific antigen of Chlamydia and their use for antigen detection by reverse passive haemagglutination.
J Gen Microbiol. 131 (1): 7-15.
References for Chlamydia LPS antibody
-
Campbell, S. et al. (1994) Lipopolysaccharide in cells infected by Chlamydia trachomatis.
Microbiology.140: 1995-2002. -
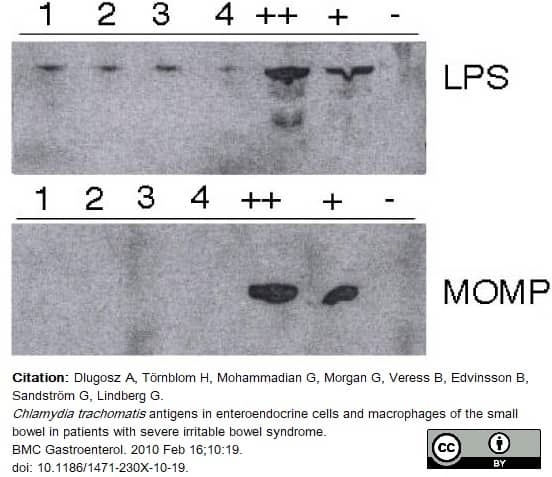
Dlugosz, A. et al. (2010) Chlamydia trachomatis. antigens in enteroendocrine cells and macrophages of the small bowel in patients with severe irritable bowel syndrome.
BMC Gastroenterol. 10: 19. -
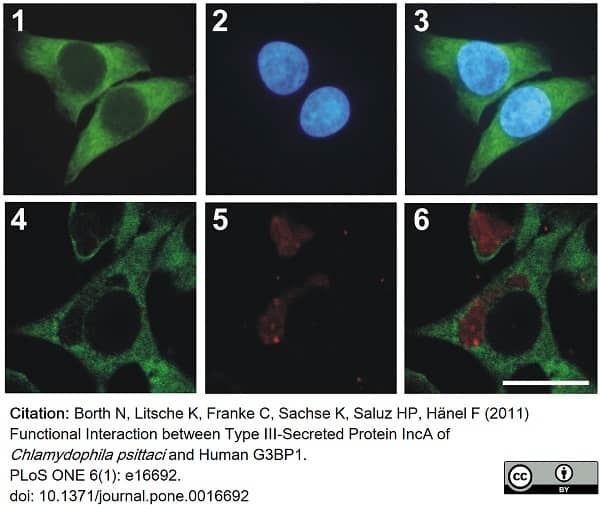
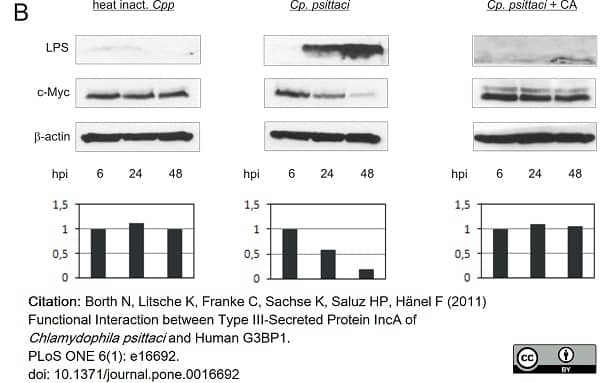
Borth, N. et al. (2011) Functional interaction between type III-secreted protein IncA of Chlamydophila psittaci. and human G3BP1.
PLoS One. 6 (1): e16692. -
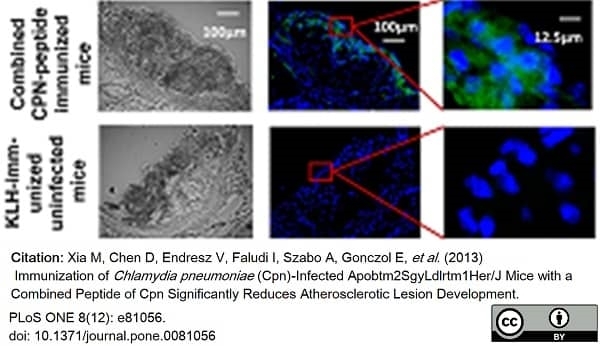
Xia, M. et al. (2013) Immunization of Chlamydia pneumoniae. (Cpn)-infected Apob(tm2Sgy)Ldlr(tm1Her)/J mice with a combined peptide of Cpn significantly reduces atherosclerotic lesion development.
PLoS One. 8 (12): e81056. -
Xia M et al. (2013) Immunization of Chlamydia pneumoniae (Cpn)-infected Apob(tm2Sgy)Ldlr(tm1Her)/J mice with a combined peptide of Cpn significantly reduces atherosclerotic lesion development.
PLoS One. 8 (12): e81056. -
Mosolygó, T. et al. (2019) Selenocompounds as Novel Antibacterial Agents and Bacterial Efflux Pump Inhibitors.
Molecules. 24(8):1487. -
Kókai, D. et al. (2021) Ambroxol Treatment Suppresses the Proliferation of Chlamydia pneumoniae in Murine Lungs.
Microorganisms. 9 (4): 880.
Further Reading
-
Ingalls, R.R. et al. (1995) The inflammatory cytokine response to Chlamydia trachomatis infection is endotoxin mediated.
Infect Immun. 63 (8): 3125-30.
- RRID
- AB_915244
MCA2718
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Bacterial ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up