Conversion of Fab to Full Immunoglobulin Format
-
Monoclonal Generation
-
Custom Recombinant Monoclonal Antibody Generation
-
HuCAL® Antibody Generation Process
- Antigens and antigen expression services
- Guided Selection Strategies
- Affinity maturation
- Selection of antibodies based on binding strength
- Extended QC services
- Fab antibody formats and epitope tags
- Conversion of Fab to full immunoglobulin format
- Fab Antibody Production
- Screening and Pair Selection using the Bio-Plex® System
- Flow cytometry antibody screening
-
HuCAL® Antibody Generation Process
-
Custom Recombinant Monoclonal Antibody Generation
s
Simplified Sourcing via Scientist.com and ScienceExchange

Access Bio-Rad custom antibody generation services through the Scientist.com and ScienceExchange marketplaces.
s
Custom Antibody Project Inquiry Form
A personal, no obligation quotation for a custom monoclonal antibody generation project
s
My Green Lab Certified!

Our custom antibody services based in Neuried (Munich), Germany, are My Green Lab certified!
s
Contact our Custom Antibody Specialists
Tel: +49 (0) 89 80 90 95 45
Fax: +49 (0) 89 80 90 95 50
Office: Bio-Rad AbD Serotec GmbH, Campus Neuried, Anna-Sigmund-Str. 5, 82061 Neuried, Germany
HuCAL® Fab antibodies can be converted into full length human or chimeric antibodies when an Fc region is required for your application. The variable heavy and light chain genes are cloned into vectors with the desired constant regions and co-transfected for expression in mammalian cells. Antibodies are purified and tested by quality control ELISA.
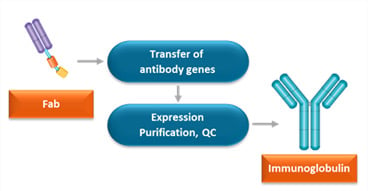
Fig. 1. Overview of the process for conversion of Fab to immunoglobulin.
Fully human HuCAL immunoglobulins (Igs) of various isotypes are expressed in a human cell line, with secure long term supply. This makes them ideal for bioanalytical assays supporting biotherapeutic development and as surrogates for human sera controls and calibrators in immunoassay applications.
Table 1. Choice of Fc region for full immunoglobulin format.
|
Human |
IgG1, IgG2, IgG3, IgG4 |
|
Other species |
On request e.g. mouse, rat, rabbit |
There are three options for obtaining your antibodies in Ig format:
1. Receive all Fab antibodies generated from your project
- Test all in your application
- Select your preferred candidates for conversion to Ig
- Order from Bio-Rad
- Available for all isotypes in table 1
- Up to 1 mg of each antibody supplied (IgM as supernatant, all other isotypes purified)
2. Receive all Fab antibodies generated from your project
- Test all in your application
- Select your preferred candidates for conversion to Ig
- Order the Fab antibody gene sequences for your selected antibodies from Bio-Rad
- Carry out the cloning, expression and purification in your own lab
3. Opt to have a number of Fab antibodies from your project converted directly, without testing the Fabs in your application
- Receive the full Ig antibodies for testing
- Available for human IgG1, IgG2 and IgG4
- Conversion of 4, 8, 10 or 12 antibodies in parallel
- Up to 0.2 mg of each antibody initially supplied for testing
Custom Services Available |
Catalog Number |
|---|---|
|
Conversion of one Fab antibody to full Ig format and test expression of up to 1 mg, all isotypes available |
CAB11571 |
|
Conversion of 4, 8, 10 or 12 Fab antibodies in parallel to full Ig format and microscale expression of up to 0.2 mg; IgG1, IgG2, IgG4 |
CAB11603 |
|
Production of Ig antibody, 250 µg |
CAB11558 |
|
Production of Ig antibody, per mg |
CAB11559 |
|
Antibody DNA and amino acid sequence, light chain, Fd chain and tags in electronic format |
CAB11582 |
Critical reagents for bioanalytical assays
With HuCAL technology it is possible to develop a single high-affinity antibody for use in Fab and/or Ig format for pharmacokinetic (PK) studies. In the fully human Ig format it can also be used as a calibrator for immunogenicity testing, or anti-drug antibody (ADA) assays. This eliminates the need for patient or animal sera and greatly reduces assay development time and effort.
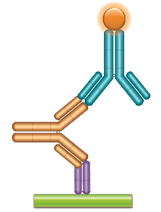
Schematic image of PK bridging ELISA. Anti-idiotypic capture antibody, Fab format (purple), monoclonal antibody drug (gold), anti-idiotypic detection antibody, Ig format (blue), labeled with HRP.
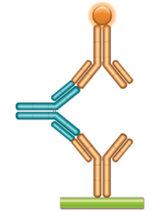
Schematic image of ADA bridging assay. Monoclonal antibody drug as capture antibody and detection antibody labeled with HRP (gold), fully human anti-idiotypic antibody, Ig format (blue).
Fig. 2. Examples of the use of HuCAL antibodies in full immunoglobulin format in PK and ADA assays.
Controls for autoimmune and infectious disease assays
HuCAL immunoglobulins have been successfully used to replace patient sera as a consistent, secure, and fully characterized source of control human antibodies for immunoassays.
Publication: Knappik A et al. (2009). Development of recombinant human IgA for anticardiolipin antibodies assay standardization. Ann N Y Acad Sci. 1173:190-8