Flow Cytometry: Proliferation and Cell Cycle
Author: Mike Blundell | Reviewer: Chloe Fenton
Cell proliferation can be measured by flow cytometry using several methods. For an overview of the techniques and reagents available from Bio-Rad visit our dedicated proliferation measurement by flow cytometry website. Whilst simple differences in the forward and side scatter will indicate if there are some changes in the cell cycle more accurate methods are available. One method is to stain with an antibody against a proliferation marker such as Ki67, MCM2 or PCNA. Alternatively you can incubate your cells with BrdU, which incorporates into DNA during S-phase of the cell cycle. Incorporated BrdU can be detected using fluorescently labeled anti BrdU antibodies. When combined with a DNA stain such as PI or DAPI, the relative proportion of cells in S-phase can be determined.
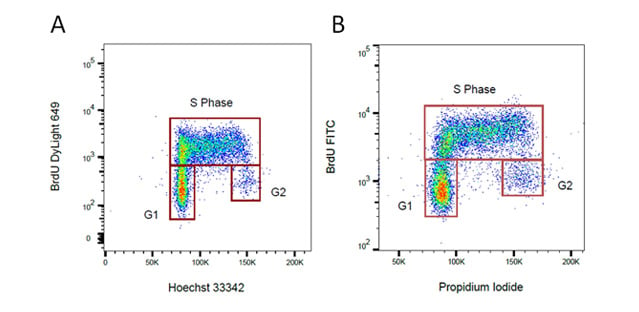
Fig. 34. BrdU staining for proliferation. Proliferating cells were stained for incorporated BrdU against total DNA content using A. Hoechst or B. Propidium Iodide. BrdU was detected by labeling a mouse primary anti-BrdU antibody (MCA2483) with the appropriate secondary antibody attached to a fluorophore.
In addition to using antibodies, cytoplasmic dyes, such as CFDA-SE, can be used to measure proliferation. Cells are incubated with the protein binding dyes and as the labeled cells divide, the concentration of the dye is halved and the proliferation measured based upon the reduced levels of fluorescence in subsequent generations. The advantage of these dyes is that they are non-toxic and available in a wide variety of colors, so can be combined with immunophenotyping and do not require the sample to be fixed.
Cell Cycle Determination
The proportion of cells within each stage of the cell cycle can be determined using DNA binding dyes such as PI, 7-AAD, Hoechst 33342 and DAPI that bind in a stoichiometric manner. This way cells in G2, which have twice as much DNA as cells in G1, will fluoresce twice as bright. To ensure good staining the cells should be fixed in cold 70% ethanol. However this can interfere with other staining protocols, see our protocols section for advice on the different methods. Cell cycle analysis is usually measured on a linear scale unlike most flow cytometry which uses a logarithmic scale as the differences in fluorescence are usually smaller. It is important to gate out any doublets from the data and the data can be improved by using a low flow rate on the cytometer. Many flow cytometry software programs now offer algorithms to accurately estimate the cell cycle phases.
Flow Cytometry Basics Guide Download
Get your own copy to peruse at your leisure
Download our updated Flow Cytometry Basics Guide to have practical advice, best practice examples, and a basic overview of all the important flow cytometry principles in one handy location.
Resources
Bio-Rad is committed to helping you succeed in Flow Cytometry by sharing knowledge and best practices from our experts. Below are some extremely useful resources to enhance your journey of discovery and support your success.
