Flow Cytometry Guide: Immunophenotyping
Author: Mike Blundell | Reviewer: Chloe Fenton
The most common use of flow cytometry is in the identification of markers on cells, particularly the immune system or immunophenotyping. It is the desire to detect an increased number of cell subsets of the immune system that has driven the increase in multicolor flow cytometry.
Immunophenotyping (Figure 32) can be simply identifying a cell by a single marker or more complex identification of cells, using homing profile, activation states and cytokine release all in one panel. As a consequence, experimental protocols are often a combination of surface and intracellular staining. In addition to basic research, immunophenotyping is now routinely used in clinical applications to diagnose disease or to monitor and evaluate residual disease.
A simple example of immunophenotyping using surface staining is shown below where a simple four color staining panel can help identify four major cell subsets in human peripheral blood using markers that require no compensation. Additional information in a more complex panel may include the subsets of T cells, e.g. T helper, T cytotoxic and T regulatory, using markers such as CD4, CD8, CD25, CD127 and FOXP3, whether they are naïve, memory or activated, using markers such as CD45RA, CD45RO, CD62L, CD69 and HLA-DR and their cytokine profile such as IFN-g, IL-2, IL-17, IL-9. It is easy to see how an 18 color panel can be quickly built. Refer to Chapter 6 for more information on building multicolor panels.
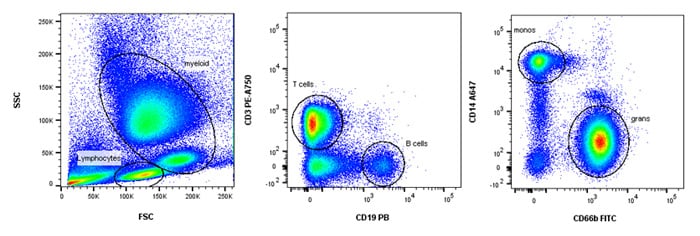
Fig. 32. Immunophenotyping of whole blood. Simple 4 color immunophenotyping panel using CD3, CD19, CD66b and CD14 to identify T, B, granulocyte and monocytes respectively.
Flow Cytometry Basics Guide Download
Get your own copy to peruse at your leisure
Download our updated Flow Cytometry Basics Guide to have practical advice, best practice examples, and a basic overview of all the important flow cytometry principles in one handy location.
Resources
Bio-Rad is committed to helping you succeed in Flow Cytometry by sharing knowledge and best practices from our experts. Below are some extremely useful resources to enhance your journey of discovery and support your success.
