Identification of the Immunological Correlates of Protection in Ruminants

- On This Page
- Overview
- Research objectives
Professor Gary Entrican, Principal Research Scientist at The Moredun Research Institute and Honorary Professor within the College of Medicine and Veterinary Medicine at University of Edinburgh, UK
Effective vaccines are the most efficient way of preventing the transmission and spread of infectious diseases of both humans and animals. Some vaccines have been so effective they have resulted in eradication of disease. Examples of these are smallpox in humans and rinderpest in cattle. However, there are still many major diseases for which no effective vaccines exist. This is either because prototype vaccines that have been developed do not protect, they present a hazard in themselves (benefit versus risk) or because they interfere with diagnostic testing, especially for infections that can persist and cause chronic disease. The latter element is particularly true for management of livestock diseases where development of effective vaccines and diagnostic tests go hand-in-hand to allow discrimination between infected and vaccinated animals (DIVA).
The trial-and-error approach to vaccine development that has been so successful in the past has now largely been exhausted. The ‘easy’ vaccines have been made. It is becoming increasingly clear that alternative approaches to vaccine design are required to control the more difficult diseases. A major part of this process is gaining an understanding of how the immune system is activated and regulated so that optimum protection is achieved (benefit) with minimum adverse effects (risk). Investigation of the immune system can reveal the correlates of protection that should be induced by an effective vaccine. Different infections (viruses, bacteria, parasites) are controlled by different arms of the immune response, hence the immunological correlates of protection need to be tailored accordingly to individual pathogens for maximal vaccine efficacy (Fig. 1).
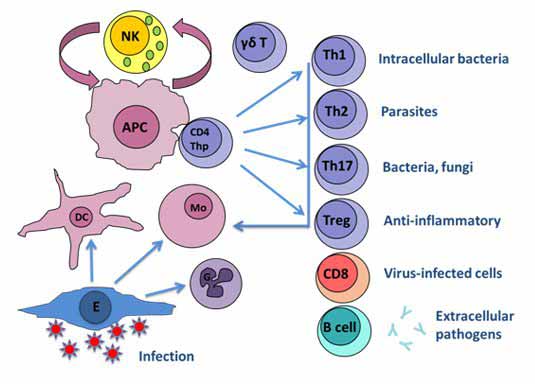
Fig. 1: Effective responses to infection are dependent on interactions between multiple cell types of the innate and adaptive immune systems, involving induction and effector phases. These different cell types can be characterized though a combination of cell-surface markers, intracellular transcription factors and the cytokines they produce. E: epithelial cell; G: granulocyte; Mo: monocyte; DC: dendritic cell; APC: antigen presenting cell; Thp: T helper precursor; NK: natural killer cell.
Infectious agents behave differently in different hosts, so a vaccine should ideally be designed for (and tested in) the appropriate host. In this regard, the challenge facing ruminant immunology is the relatively poor capability to identify immunological correlates of protection (compared to rodent models). This is due to the relative lack of reagents to fully characterize cattle and sheep immune responses. To help address this gap in capability, researchers at the Moredun Research Institute and The Roslin Institute (University of Edinburgh) are collaborating on a project to specifically develop and characterize immunological reagents for cattle and sheep.
This project is an Industrial Partnership Award (IPA) funded by the Biotechnology and Biological Sciences Research Council (BBSRC) in conjunction with Bio-Rad. The project is being led by Professor Gary Entrican (Moredun) and Professor Liz Glass (Roslin). Summaries of the project can be found in the Research in a Nutshell videos at University of Edinburgh:
The overarching aim is to characterize myeloid cell subsets and CD4+ve T helper cell subsets in cattle and sheep (Fig. 1) and investigate how they can be modulated to influence immune activation and immune function. There are five specific aims (listed below).
Research Objectives
- Clone and express bovine and ovine cell-surface molecules associated with myeloid subsets, the intracellular transcription factors and the cytokines associated with T cell subsets.
- Assess the species cross-reactivity of commercially-available antibodies on transfected Chinese Hamster Ovary cells developed within Objective 1.
- Produce and assess biologically-active recombinant cytokines for differentiation of myeloid cell and T cell subsets and characterize these subsets and their interactions.
- Produce and characterize monoclonal antibodies to molecules that define myeloid cell subsets and T cell subsets in cattle and sheep.
- Commercialize, promote and distribute reagents and knowledge through the Industrial Partnership with Bio-Rad.
This work will build on the expertise of the research teams at Moredun and Roslin and expand the portfolio of reagents they have already produced and commercialized. This includes dendritic cell growth kits for cattle and monoclonal antibodies to ruminant cytokines. The group works closely with overseas collaborators in the United States Veterinary Immunology Reagent Network (US VIRN) to share knowledge and ensure that there is no duplication of effort. It is important that the main gaps in capability are filled. To help prioritize the targets, the project team has an open call for information from other researchers on reagent function/gaps. Anyone who has information they wish to share can send an email to immunecorrelates@moredun.ac.uk.

The reagents developed in this project will facilitate the identification of immunological correlates of protection to many different diseases of cattle and sheep and will also inform on the best ways to deliver new vaccines to maximize their effects. Ultimately this will lead to more strategic approaches to the management and control of infectious diseases of farmed ruminants. The link with Bio-Rad will ensure that the techniques and reagents developed will not be limited to use within this project but will be made available to the research community on a commercial basis.
‘The route to identification of the immunological correlates of protection in ruminants’ is funded through the BBSRC Industrial Partnership Award scheme (grant numbers BB/I019863/1 & BB/I020519/1) in conjunction with Bio-Rad. The project is supported by the Scottish Government Rural and Environment Science and Analytical Services Division (RESAS) through special arrangement with BBSRC.


