CD31 antibody | ER-MP12





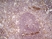

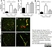

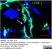


Rat anti Mouse CD31
- Product Type
- Monoclonal Antibody
- Clone
- ER-MP12
- Isotype
- IgG2a
- Specificity
- CD31
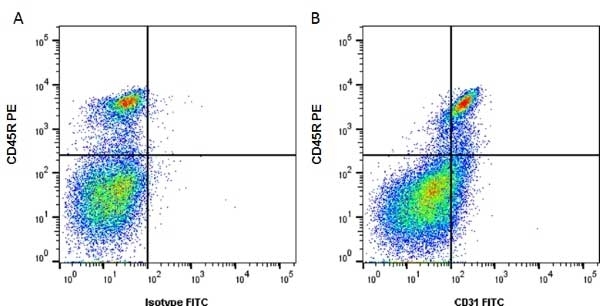
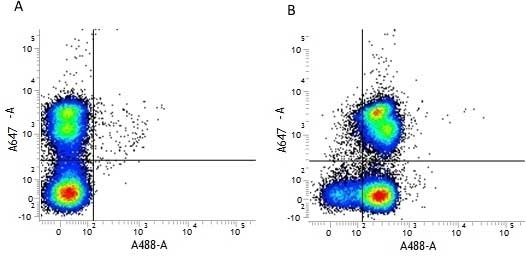
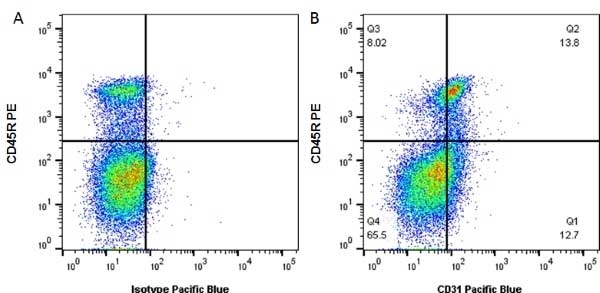
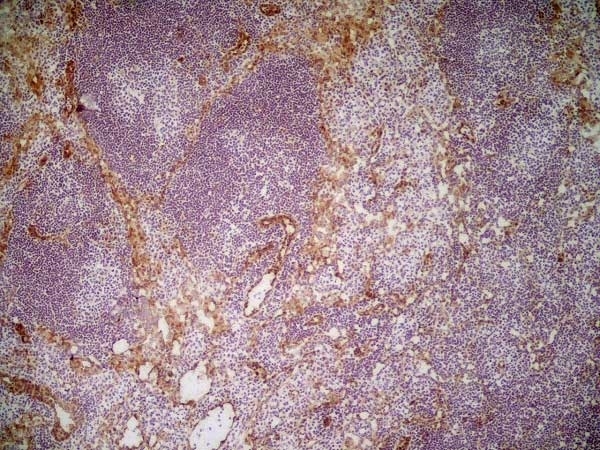
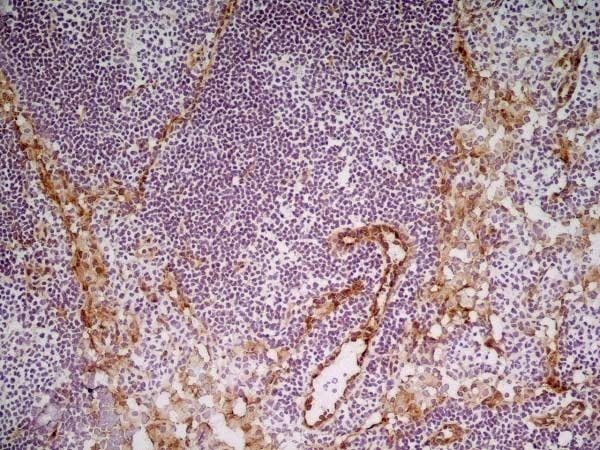
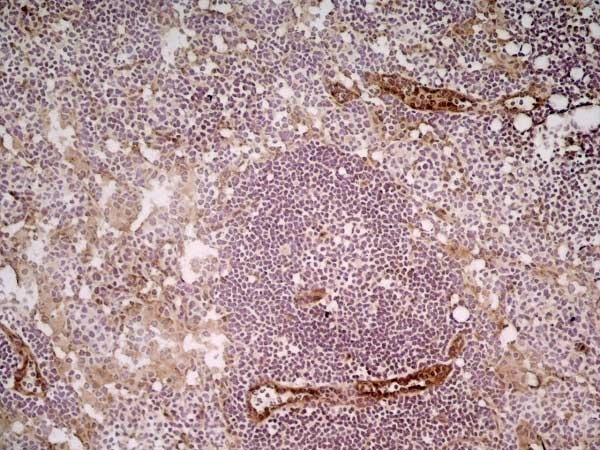
| Rat anti Mouse CD31 antibody, clone ER-MP12 recognizes mouse CD31, a 140 kDa cell surface glycoprotein expressed at high levels on endothelial cells, platelets and most leukocyte subpopulations. CD31 is also expressed on a major population of macrophage / dendritic cell precursors in the bone marrow. Rat anti Mouse CD31 antibody, clone ER-MP12 can be used in conjunction with clone ER-MP20 (MCA2389GA) in two colour flow cytometric analysis, to identify different stages of myeloid progenitor cells in mouse bone marrow (de Bruijn et al. 1998). |
- Target Species
- Mouse
- Product Form
- Purified IgG - liquid
- Preparation
- Purified IgG prepared by affinity chromatography on Protein G from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- 0.09% sodium azide (NaN3)
- Carrier Free
- Yes
- Immunogen
- BALB/c macrophage precursor cell hybrids
- Approx. Protein Concentrations
- IgG concentration 1.0 mg/ml
- Fusion Partners
- Cells from immunised rats were fused with the cells of the rat Y3-Ag1.2.3 myeloma cell line
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Flow Cytometry | 1/50 Pack Size: 0.25 mg, 25 µg 1/10 Pack Size: 0.1 mg |
1/200 Pack Size: 0.25 mg, 25 µg 1/100 Pack Size: 0.1 mg |
|
| Immunofluorescence | |||
| Immunohistology - Frozen 1 | 1/10 | 1/100 | |
| Immunohistology - Paraffin | |||
| Immunoprecipitation |
- 1The epitope recognised by this antibody is reported to be sensitive to formaldehyde fixation and tissue processing. Bio-Rad recommends the use of acetone fixation for frozen sections.
- Flow Cytometry
- Use10μl of the suggested working dilution to label 106 cells in 100μl
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Rat IgG2a Negative Control | MCA1212 | E F | 1 ml |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Rat IgG2a Negative Control | ||||||
References for CD31 antibody
-
Leenen, P.J. et al. (1990) Murine macrophage precursor characterization. II. Monoclonal antibodies against macrophage precursor antigens.
Eur J Immunol. 20 (1): 27-34. -
van der Loo, J. et al. (1995) Identification of hematopoietic stem cell subsets on the basis of their primitiveness using antibody ER-MP12.
Blood. 85:952-62. -
Ling, V. et al. (1997) Structural identification of the hematopoietic progenitor antigen ER-MP12 as the vascular endothelial adhesion molecule PECAM-1 (CD31).
Eur J Immunol. 27:509-14. -
de Bruijn, M.F. et al. (1998) Bone marrow cellular composition in Listeria monocytogenes infected mice detected using ER-MP12 and ER-MP20 antibodies: a flow cytometric alternative to differential counting.
J Immunol Methods. 217 (1-2): 27-39. -
Wynn, A.A. et al. (2001) Role of granulocyte/macrophage colony-stimulating factor in zymocel-induced hepatic granuloma formation.
Am J Pathol. 158 (1): 131-45. -
van Rijt, L. et al. (2002) Allergen-induced accumulation of airway dendritic cells is supported by an increase in CD31(hi)Ly-6C(neg) bone marrow precursors in a mouse model of asthma.
Blood. 100:3663-71. -
Tagoh, H. et al. (2002) Transcription factor complex formation and chromatin fine structure alterations at the murine c-fms (CSF-1 receptor) locus during maturation of myeloid precursor cells.
Genes Dev. 16:1721-37. -
Nikolic, T. et al. (2002) Developmental stages of myeloid dendritic cells in mouse bone marrow.
Int Immunol. 15:515-24.
View The Latest Product References
-
Baumeister, T. et al. (2003) Interleukin-3Ralpha+ myeloid dendritic cells and mast cells develop simultaneously from different bone marrow precursors in cultures with interleukin-3.
J Invest Dermatol. 121: 280-8. -
Geutskens, S.B. et al. (2005) Macrophages in the murine pancreas and their involvement in fetal endocrine development in vitro.
J Leukoc Biol. 78: 845-52. -
Revermann, M. et al. (2010) Soluble epoxide hydrolase deficiency attenuates neointima formation in the femoral cuff model of hyperlipidemic mice.
Arterioscler Thromb Vasc Biol. 30: 909-14. -
Sumagin, R. and Sarelius, I.H. (2010) Intercellular adhesion molecule-1 enrichment near tricellular endothelial junctions is preferentially associated with leukocyte transmigration and signals for reorganization of these junctions to accommodate leukocyte passage.
J Immunol. 184: 5242-52. -
Ross, E.A. et al. (2011) CD31 Is Required on CD4+ T Cells To Promote T Cell Survival during Salmonella Infection.
J Immunol. 187: 1553-65. -
Thum, T. et al. (2011) Impairment of endothelial progenitor cell function and vascularization capacity by aldosterone in mice and humans.
Eur Heart J. 32: 1275-86. -
Thorp, E. et al. (2011) A reporter for tracking the UPR in vivo reveals patterns of temporal and cellular stress during atherosclerotic progression.
J Lipid Res. 52 (5): 1033-8. -
Schledzewski, K. et al. (2011) Deficiency of liver sinusoidal scavenger receptors stabilin-1 and -2 in mice causes glomerulofibrotic nephropathy via impaired hepatic clearance of noxious blood factors.
J Clin Invest. 121: 703-14. -
Loureiro, J. et al. (2011) Blocking TGF-{beta}1 Protects the Peritoneal Membrane from Dialysate-Induced Damage.
J Am Soc Nephrol. 22: 1682-95. -
Matsakas, A. et al. (2012) Exercise training attenuates the hypermuscular phenotype and restores skeletal muscle function in the myostatin null mouse.
Exp Physiol. 97 (1): 125-40. -
Trottier MD et al. (2012) Enhancement of hematopoiesis and lymphopoiesis in diet-induced obese mice.
Proc Natl Acad Sci U S A. 109 (20): 7622-9. -
Moen, I. et al. (2012) Gene expression in tumor cells and stroma in dsRed 4T1 tumors in eGFP-expressing mice with and without enhanced oxygenation.
BMC Cancer. 12: 21. -
Trottier, M.D. et al. (2012) Enhanced production of early lineages of monocytic and granulocytic cells in mice with colitis
Proc Natl Acad Sci U S A.109: 16594-9. -
Kroon, P. et al. (2013) JAK-STAT blockade inhibits tumor initiation and clonogenic recovery of prostate cancer stem-like cells.
Cancer Res. 73 (16): 5288-98. -
Ono, N. et al. (2014) A subset of chondrogenic cells provides early mesenchymal progenitors in growing bones.
Nat Cell Biol. 16 (12): 1157-67. -
Eskilsson, A. et al. (2014) Distribution of microsomal prostaglandin E synthase-1 in the mouse brain.
J Comp Neurol. 522 (14): 3229-44. -
Nakamura, Y. et al. (2015) Mesenchymal-stem-cell-derived exosomes accelerate skeletal muscle regeneration.
FEBS Lett. 589 (11): 1257-65. -
Fraccarollo, D. et al. (2015) Efficacy of mineralocorticoid receptor antagonism in the acute myocardial infarction phase: eplerenone versus spironolactone.
ESC Heart Fail. 2 (3): 150-8. -
Stein-Merlob, A.F. et al. (2015) Blood Accessibility to Fibrin in Venous Thrombosis is Thrombus Age-Dependent and Predicts Fibrinolytic Efficacy: An In Vivo Fibrin Molecular Imaging Study.
Theranostics. 5 (12): 1317-27. -
Cao Y et al. (2016) IL-1β differently stimulates proliferation and multinucleation of distinct mouse bone marrow osteoclast precursor subsets.
J Leukoc Biol. 100 (3): 513-23. -
Ryan, T.E. et al. (2016) Mitochondrial therapy improves limb perfusion and myopathy following hindlimb ischemia.
J Mol Cell Cardiol. 97: 191-6. -
Shi, H. et al. (2016) Hiding inside? Intracellular expression of non-glycosylated c-kit protein in cardiac progenitor cells.
Stem Cell Res. 16 (3): 795-806. -
Chowdhury, B. et al. (2016) Hyaluronidase 2 (HYAL2) is expressed in endothelial cells, as well as some specialized epithelial cells, and is required for normal hyaluronan catabolism.
Histochem Cell Biol. 145 (1): 53-66. -
Yip, H.K. et al. (2016) Tissue plasminogen activator deficiency preserves neurological function and protects against murine acute ischemic stroke.
Int J Cardiol. 205: 133-41. -
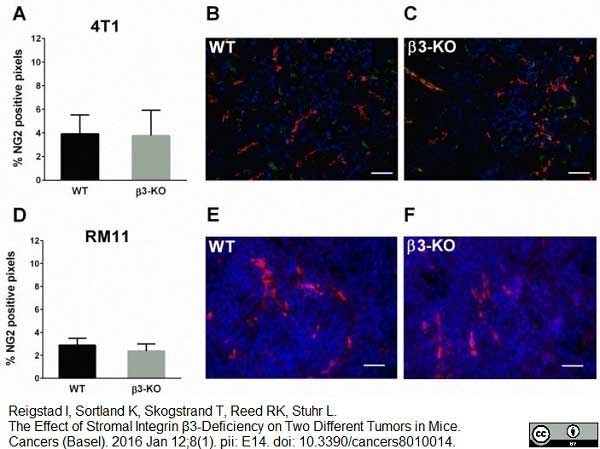
Reigstad, I. et al. (2016) The Effect of Stromal Integrin β3-Deficiency on Two Different Tumors in Mice.
Cancers (Basel). 8 (1): pii: E14. -
Bongiorno, E.K. et al. (2017) Type 1 Immune Mechanisms Driven by the Response to Infection with Attenuated Rabies Virus Result in Changes in the Immune Bias of the Tumor Microenvironment and Necrosis of Mouse GL261 Brain Tumors.
J Immunol. 198 (11): 4513-4523. -
Cao, Y. et al. (2017) TNF-α has both stimulatory and inhibitory effects on mouse monocyte-derived osteoclastogenesis.
J Cell Physiol. 232 (12): 3273-85. -
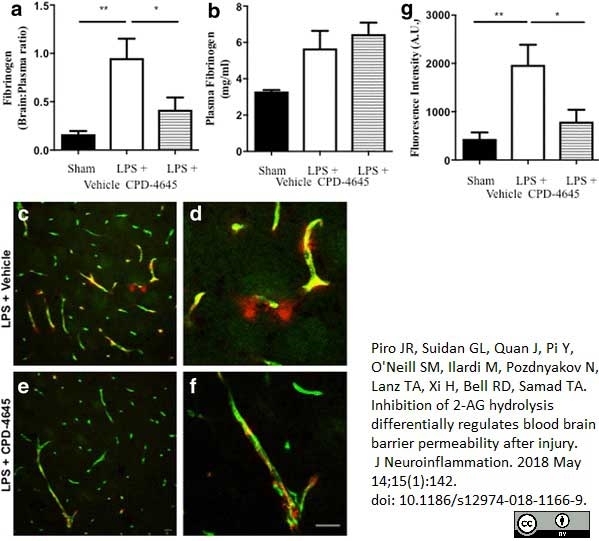
Piro, J.R. et al. (2018) Inhibition of 2-AG hydrolysis differentially regulates blood brain barrier permeability after injury.
J Neuroinflammation. 15 (1): 142. -
Oikawa, S. et al. (2018) Role of endothelial microRNA-23 clusters in angiogenesis in vivo.
Am J Physiol Heart Circ Physiol. 315 (4): H838-H846. -
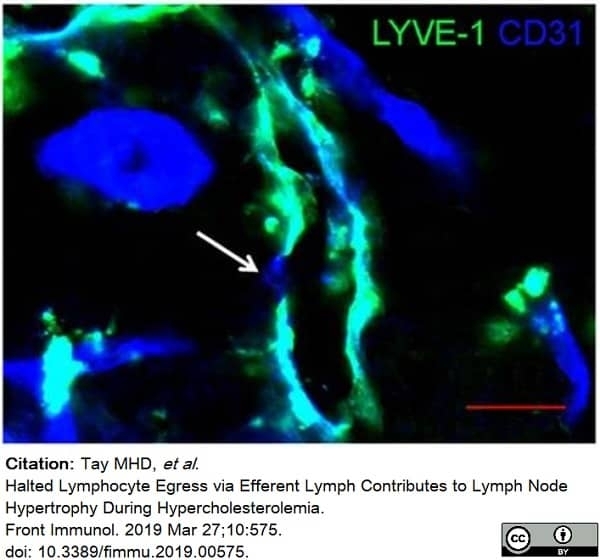
Tay, M.H.D. et al. (2019) Halted Lymphocyte Egress via Efferent Lymph Contributes to Lymph Node Hypertrophy During Hypercholesterolemia.
Front Immunol. 10: 575. -
Iring, A. et al. (2019) Shear stress-induced endothelial adrenomedullin signaling regulates vascular tone and blood pressure.
J Clin Invest. 129 (7): 2775-91. -
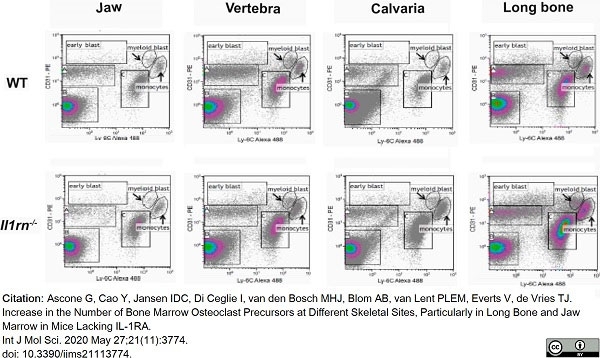
Ascone, G. et al. (2020) Increase in the Number of Bone Marrow Osteoclast Precursors at Different Skeletal Sites, Particularly in Long Bone and Jaw Marrow in Mice Lacking IL-1RA.
Int J Mol Sci. 21(11):3774. -
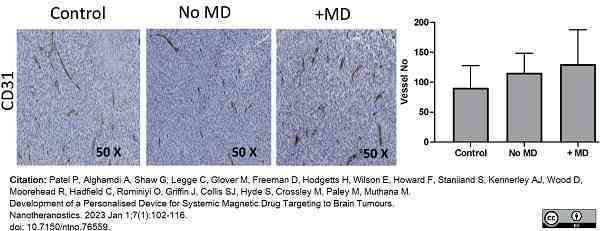
Patel, P. et al. (2023) Development of a Personalised Device for Systemic Magnetic Drug Targeting to Brain Tumours.
Nanotheranostics. 7 (1): 102-16. -
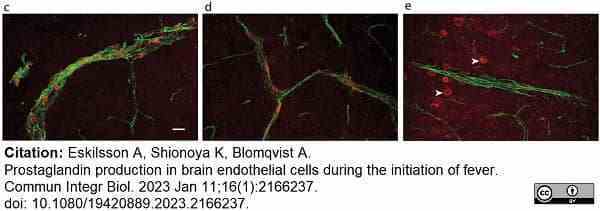
Eskilsson, A. et al. (2023) Prostaglandin production in brain endothelial cells during the initiation of fever.
Commun Integr Biol. 16 (1): 2166237. -
Yamashita, J. et al. (2021) Effect of bisphosphonates on healing of tooth extraction wounds in infectious osteomyelitis of the jaw.
Bone. 143: 115611. -
Molás, R.B. et al. (2020) The involvement of annexin A1 in human placental response to maternal Zika virus infection.
Antiviral Res. 179: 104809.
- Synonyms
- PECAM-1
- RRID
- AB_2161026
- UniProt
- Q08481
- Entrez Gene
- Pecam1
- GO Terms
- GO:0007155 cell adhesion
- GO:0016021 integral to membrane
- GO:0005625 soluble fraction
- GO:0007266 Rho protein signal transduction
- GO:0030054 cell junction
- GO:0030334 regulation of cell migration
- GO:0030485 smooth muscle contractile fiber
- GO:0042060 wound healing
- GO:0045121 membrane raft
- View More GO Terms
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Mouse ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up