CD169 antibody | 3D6.112


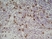



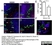
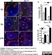
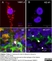
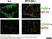
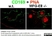
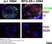
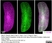
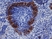
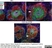
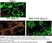
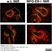








Rat anti Mouse CD169
- Product Type
- Monoclonal Antibody
- Clone
- 3D6.112
- Isotype
- IgG2a
- Specificity
- CD169
| Rat anti Mouse CD169 antibody, clone 3D6.112 recognizes mouse CD169 also known as sialoadhesin, Sheep erythrocyte receptor or Siglec-1. CD169 is a 1695 amino acid, ~180 kDa single pass, type 1 transmembrane glycoprotein containing a single Ig-like V-type domain and sixteen Ig-like C2-type domains. CD169 is a macrophage restricted receptor, preferentially binding to alpha 2,3 linked sialic acid residues (Crocker et al. 1991) and is expressed on stromal macrophages in many tissues, particularly in lymph nodes, bone marrow and on marginal metallophilic macrophages in the spleen (Morris et al. 1991). CD169 has been implicated in a number of roles including cell-cell interactions with lymphocytes (van den Berg et al. 1992) and granulocytes (Crocker et al. 1995). CD169 expressing macrophages have also been suggested to play a role in host resistance to lymphoma metastasis (Umansky et al. 1996). In pigs CD169 has also been identified as a macrophage restricted receptor for porcine reproductive and respiratory syndrome virus (Delputte et al. 2007). CD169 expressing macrophages have also been implicated in the regulation of autoimmune disease progression through their interaction with regulatory T cells via CD169 (Wu et al. 2009). CD169 has also been shown to play a critical role in the recognition and elimination of invasive sialylated microorganisms including Campylobacter jejuni (Klass et al. 2012) and group B Streptococcus (Chang et al. 2014). The functional activity of rat anti mouse CD169 antibody, clone 3D6.112, its ability to inhibit binding of red blood cells to CD169 can be considerably enhanced by derivitization of the antibody with polyethylene glycol (Ducreux et al. 2008). |
- Target Species
- Mouse
- Product Form
- Purified IgG - liquid
- Preparation
- Purified IgG prepared by affinity chromatography on Protein G from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
0.09% Sodium Azide - Carrier Free
- Yes
- Immunogen
- Purified murine sialoadhesin.
- Approx. Protein Concentrations
- MCA884GA: IgG concentration 1 mg/ml
- MCA884: IgG concentration 1.0 mg/ml
- Fusion Partners
- Spleen cells from an immunized AO rat were fused with the cells of the Y3 rat myeloma cell line.
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
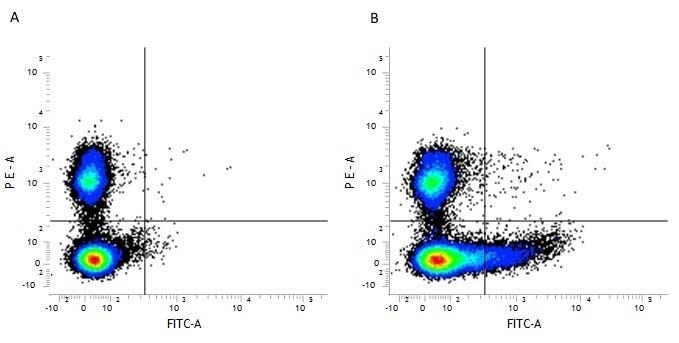
| Flow Cytometry | 1/100 | 1/1000 | |
| Immunofluorescence | |||
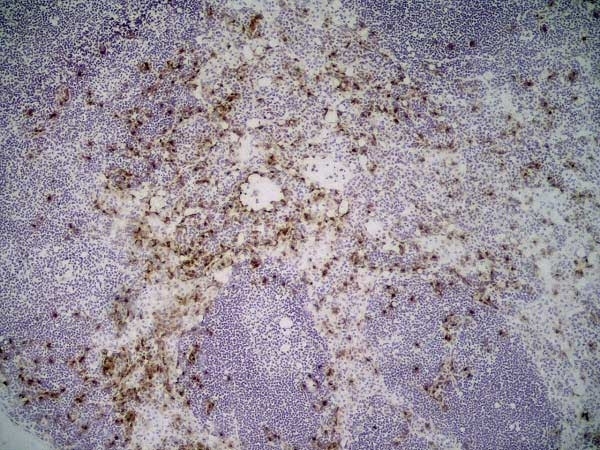
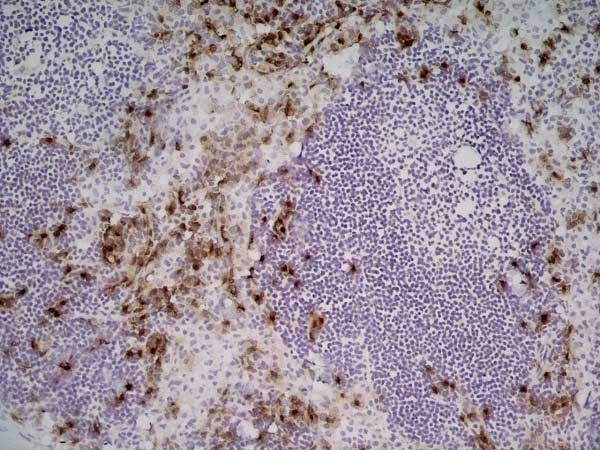
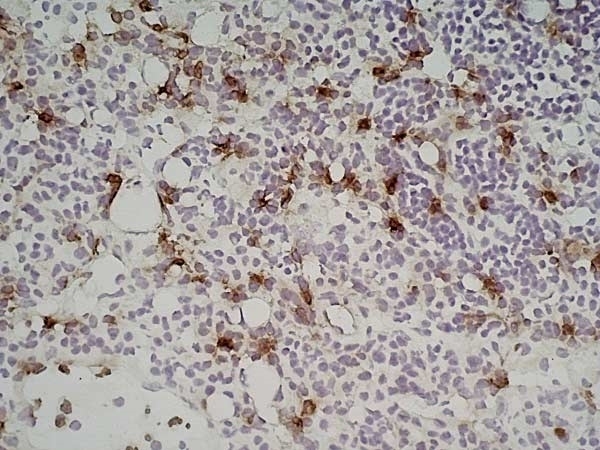
| Immunohistology - Frozen 1 | 1/50 | 1/100 |
- 1Bio-Rad recommend using fixation with either 2% paraformaldehyde or ethanol for optimal results.
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Rat IgG2a Negative Control | MCA1212 | E F | 1 ml |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Rat IgG2a Negative Control | ||||||
References for CD169 antibody
-
Crocker, P.R. et al. (1991) Purification and properties of sialoadhesin, a sialic acid-binding receptor of murine tissue macrophages.
EMBO J. 10 (7): 1661-9. -
Barnes, Y.C. et al. (1999) Sialylation of the sialic acid binding lectin sialoadhesin regulates its ability to mediate cell adhesion.
Blood. 93: 1245-52. -
Kaisho, T. et al. (2001) IkappaB kinase alpha is essential for mature B cell development and function.
J Exp Med. 193: 417-26. -
Hughes, E.H. et al. (2003) Generation of Activated Sialoadhesin-Positive Microglia during Retinal Degeneration.
Invest Ophthalmol Vis Sci. 44: 2229-34. -
Reid, D.M. et al. (2004) Expression of the beta-glucan receptor, Dectin-1, on murine leukocytes in situ correlates with its function in pathogen recognition and reveals potential roles in leukocyte interactions.
J Leukoc Biol. 76: 86-94. -
Lin, H.H. et al. (2005) The macrophage F4/80 receptor is required for the induction of antigen-specific efferent regulatory T cells in peripheral tolerance.
J Exp Med. 201: 1615-25. -
Krücken, J. et al. (2005) Massive destruction of malaria-parasitized red blood cells despite spleen closure.
Infect Immun. 73: 6390-8. -
Vora, K.A. et al. (2005) Sphingosine 1-phosphate receptor agonist FTY720-phosphate causes marginal zone B cell displacement.
J Leukoc Biol. 78: 471-80.
View The Latest Product References
-
Cunningham, C. et al. (2005) Comparison of inflammatory and acute-phase responses in the brain and peripheral organs of the ME7 model of prion disease.
J Virol. 79: 5174-84. -
Miyake, Y. et al. (2007) Protective role of macrophages in noninflammatory lung injury caused by selective ablation of alveolar epithelial type II Cells.
J Immunol. 178: 5001-9. -
Sancho-Pelluz, J. et al. (2008) Sialoadhesin expression in intact degenerating retinas and following transplantation.
Invest Ophthalmol Vis Sci. 49: 5602-10. -
Chtanova, T. et al. (2008) Dynamics of neutrophil migration in lymph nodes during infection.
Immunity. 29: 487-96. -
Taylor, P.R. et al. (2008) Development of a specific system for targeting protein to metallophilic macrophages.
Proc Natl Acad Sci U S A. 101: 1963-8. -
Anthony, R.M. et al. (2008) Identification of a receptor required for the anti-inflammatory activity of IVIG.
Proc Natl Acad Sci U S A. 105: 19571-8. -
Peng, Y. et al. (2009) Ly6C(low) monocytes differentiate into dendritic cells and cross-tolerize T cells through PDL-1.
J Immunol. 182: 2777-85. -
Toda, M. et al. (2009) Ligation of tumour-produced mucins to CD22 dramatically impairs splenic marginal zone B-cells.
Biochem J. 417: 673-83. -
Qiu, C.H. et al. (2009) Novel subset of CD8{alpha}+ dendritic cells localized in the marginal zone is responsible for tolerance to cell-associated antigens.
J Immunol. 182: 4127-36. -
Malkiel, S. et al. (2009) The loss and gain of marginal zone and peritoneal B cells is different in response to relapsing fever and Lyme disease Borrelia.
J Immunol. 182: 498-506. -
Hemmi, H. et al. (2009) A new triggering receptor expressed on myeloid cells (Trem) family member, Trem-like 4, binds to dead cells and is a DNAX activation protein 12-linked marker for subsets of mouse macrophages and dendritic cells.
J Immunol. 182: 1278-86. -
Hsu, K.M. et al. (2009) Murine cytomegalovirus displays selective infection of cells within hours after systemic administration.
J Gen Virol. 90:33-43. -
Idoyaga, J. et al. (2009) Antibody to Langerin/CD207 localizes large numbers of CD8alpha+ dendritic cells to the marginal zone of mouse spleen.
Proc Natl Acad Sci U S A. 106: 1524-9. -
Huang, Q.Q. et al. (2010) FLIP: a novel regulator of macrophage differentiation and granulocyte homeostasis.
Blood. 116: 4968-77. -
Barral, P. et al. (2010) CD169(+) macrophages present lipid antigens to mediate early activation of iNKT cells in lymph nodes.
Nat Immunol. 11: 303-12. -
Albacker, L.A. et al. (2010) TIM-4, a receptor for phosphatidylserine, controls adaptive immunity by regulating the removal of antigen-specific T cells.
J Immunol. 185: 6839-49. -
Iannacone M (2010) Subcapsular sinus macrophages prevent CNS invasion on peripheral infection with a neurotropic virus.
Nature. 465: 1079-83. -
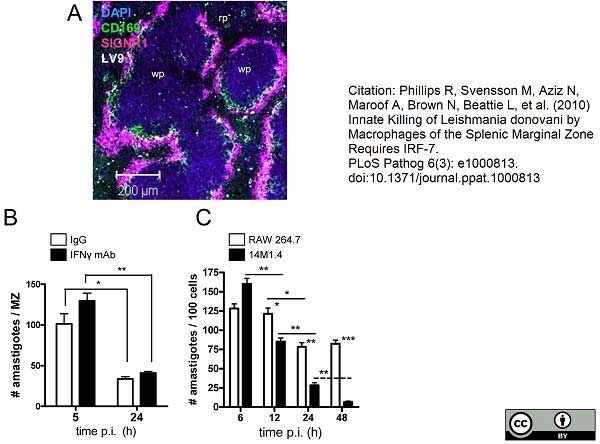
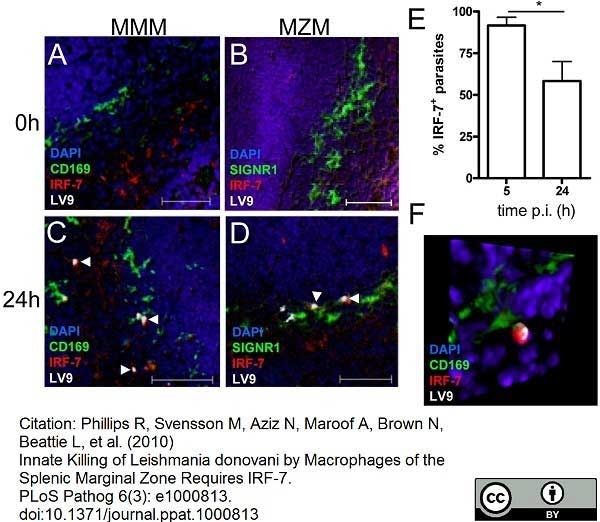
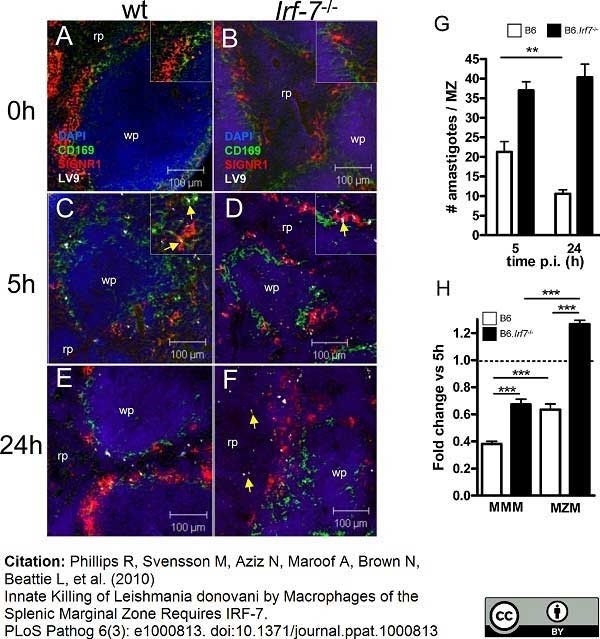
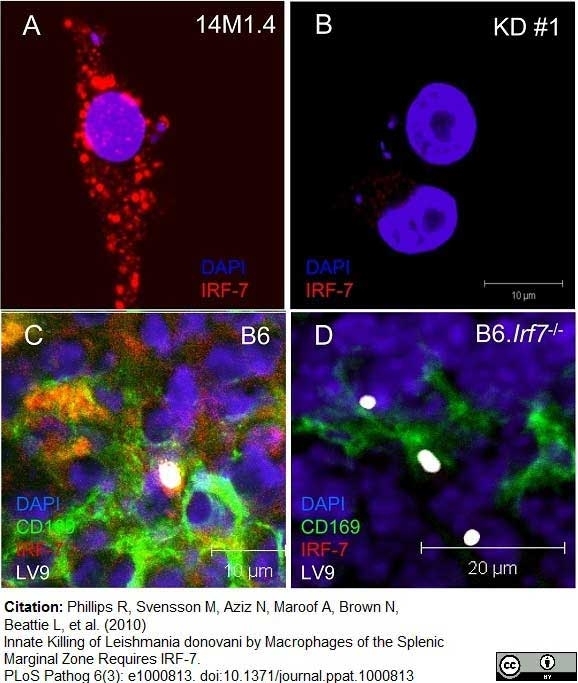
Phillips, R. et al. (2010) Innate killing of Leishmania donovani by macrophages of the splenic marginal zone requires IRF-7.
PLoS Pathog. 6(3):e1000813. -
Hashimoto, D. et al. (2011) Pretransplant CSF-1 therapy expands recipient macrophages and ameliorates GVHD after allogeneic hematopoietic cell transplantation.
J Exp Med. 208: 1069-82. -
Notley, C.A. et al. (2011) Natural IgM is required for suppression of inflammatory arthritis by apoptotic cells.
J Immunol. 186: 4967-72. -
Lu, M. and Munford, R.S. (2011) The transport and inactivation kinetics of bacterial lipopolysaccharide influence its immunological potencyin vivo.
J Immunol. 187: 3314-20. -
Chow, A. et al. (2011) Bone marrow CD169+ macrophages promote the retention of hematopoietic stem and progenitor cells in the mesenchymal stem cell niche.
J Exp Med. 208: 261-71. -
Mansour, A. et al. (2012) Osteoclasts promote the formation of hematopoietic stem cell niches in the bone marrow.
J Exp Med. 209: 537-49. -
Vagaja, N.N. et al. (2012) Changes in murine hyalocytes are valuable early indicators of ocular disease.
Invest Ophthalmol Vis Sci. 53: 1445-51. -
Hemmi, H. et al. (2012) Treml4, an Ig superfamily member, mediates presentation of several antigens to T cells in vivo, including protective immunity to HER2 protein.
J Immunol. 188: 1147-55. -
Chen, W.C. et al. (2012) Antigen delivery to macrophages using liposomal nanoparticles targeting sialoadhesin/CD169.
PLoS One. 7e39039. -
Chang, Y.C. et al. (2014) Role of macrophage sialoadhesin in host defense against the sialylated pathogen group B Streptococcus.
J Mol Med (Berl). 92(9):951-9. -
McCabe, A. et al. (2015) Macrophage-Lineage Cells Negatively Regulate the Hematopoietic Stem Cell Pool in Response to Interferon Gamma at Steady State and During Infection.
Stem Cells. 33 (7): 2294-305. -
Cantisani, R. et al. (2015) Vaccine adjuvant MF59 promotes retention of unprocessed antigen in lymph node macrophage compartments and follicular dendritic cells.
J Immunol. 194 (4): 1717-25. -
Gao, L. et al. (2015) Infiltration of circulating myeloid cells through CD95L contributes to neurodegeneration in mice.
J Exp Med. 212 (4): 469-80. -
Park, M.H. et al. (2015) Neuropeptide Y regulates the hematopoietic stem cell microenvironment and prevents nerve injury in the bone marrow.
EMBO J. 34 (12): 1648-60. -
Farrell, H.E. et al. (2015) Lymph Node Macrophages Restrict Murine Cytomegalovirus Dissemination.
J Virol. 89 (14): 7147-58. -
Asai, H. et al. (2015) Depletion of microglia and inhibition of exosome synthesis halt tau propagation.
Nat Neurosci. 18 (11): 1584-93. -
Xu, H.C. et al. (2015) Deficiency of the B cell-activating factor receptor results in limited CD169+ macrophage function during viral infection.
J Virol. 89 (9): 4748-59. -
Prokopec, K.E. et al. (2016) Marginal Zone Macrophages Regulate Antigen Transport by B Cells to the Follicle in the Spleen via CD21.
J Immunol. 197 (6): 2063-8. -
Li, Z. et al. (2016) The Macrophage-depleting Agent Clodronate Promotes Durable Hematopoietic Chimerism and Donor-specific Skin Allograft Tolerance in Mice.
Sci Rep. 6: 22143. -
Farrell, H.E. et al. (2016) Type 1 Interferons and NK Cells Limit Murine Cytomegalovirus Escape from the Lymph Node Subcapsular Sinus.
PLoS Pathog. 12 (12): e1006069. -
Perrotta, M. et al. (2018) Deoxycorticosterone acetate-salt hypertension activates placental growth factor in the spleen to couple sympathetic drive and immune system activation.
Cardiovasc Res. 114 (3): 456-67. -
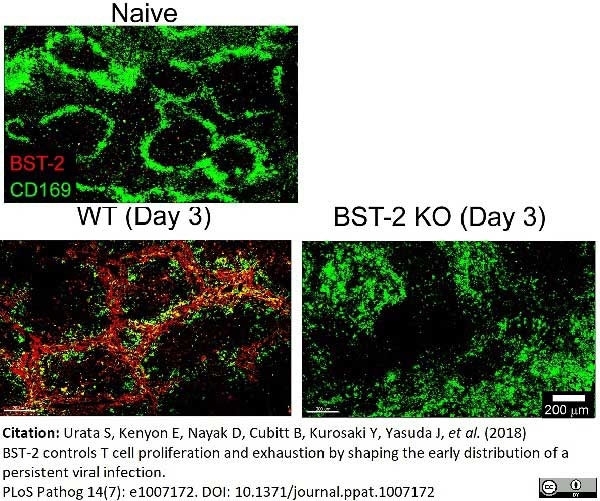
Urata, S. et al. (2018) BST-2 controls T cell proliferation and exhaustion by shaping the early distribution of a persistent viral infection.
PLoS Pathog. 14 (7): e1007172. -
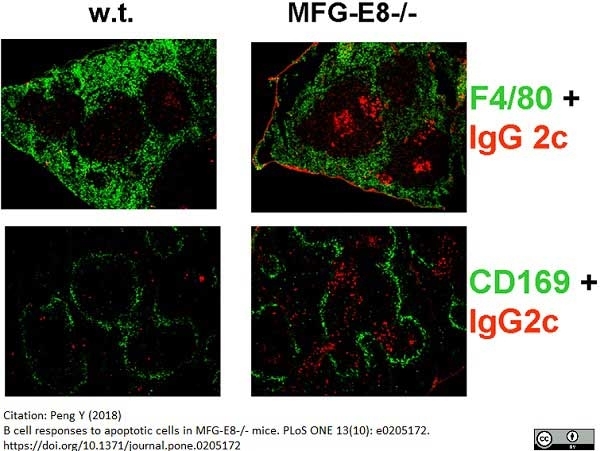
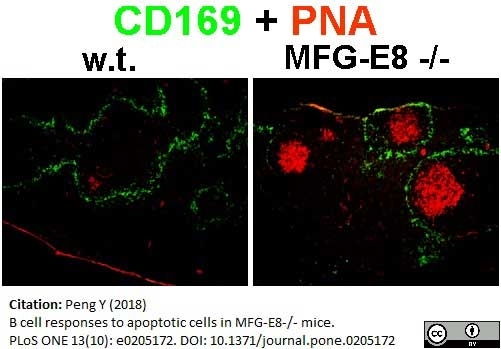
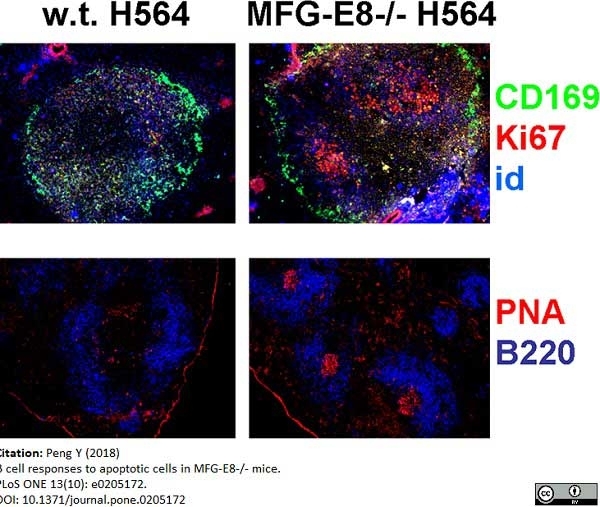
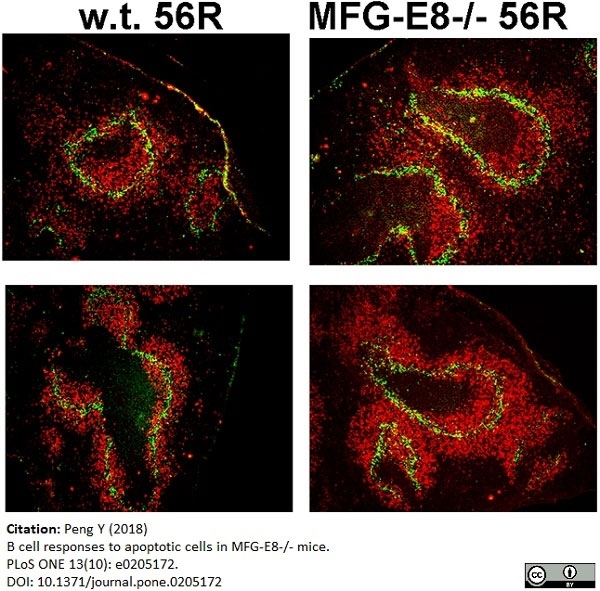
Peng, Y. (2018) B cell responses to apoptotic cells in MFG-E8-/- mice.
PLoS One. 13 (10): e0205172. -
Groh, J. et al. (2018) Teriflunomide attenuates neuroinflammation-related neural damage in mice carrying human PLP1 mutations.
J Neuroinflammation. 15 (1): 194. -
Tay, M.H.D. et al. (2019) Halted Lymphocyte Egress via Efferent Lymph Contributes to Lymph Node Hypertrophy During Hypercholesterolemia.
Front Immunol. 10: 575. -
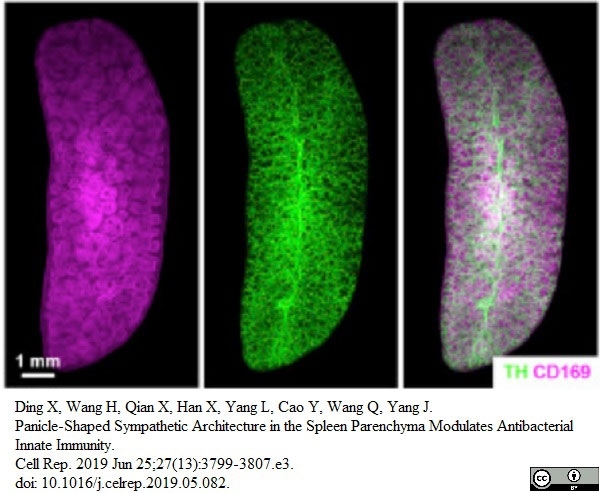
Ding, X. et al. (2019) Panicle-Shaped Sympathetic Architecture in the Spleen Parenchyma Modulates Antibacterial Innate Immunity.
Cell Rep. 27 (13): 3799-3807.e3. -
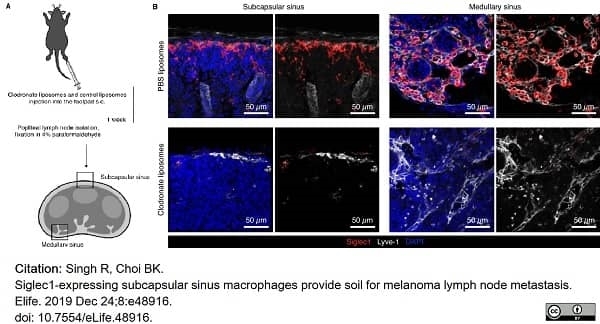
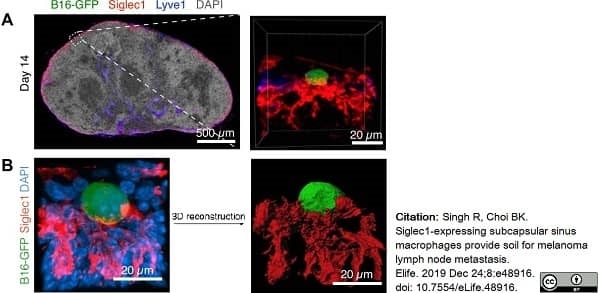
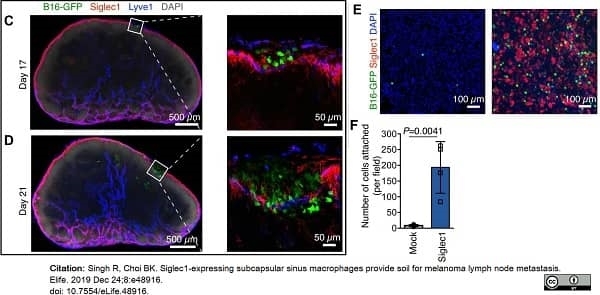
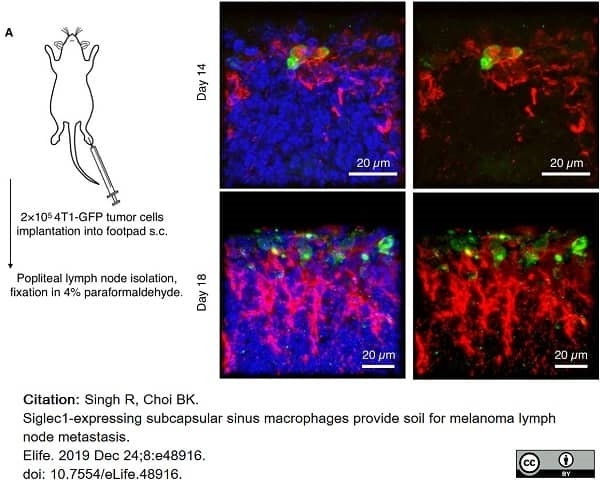
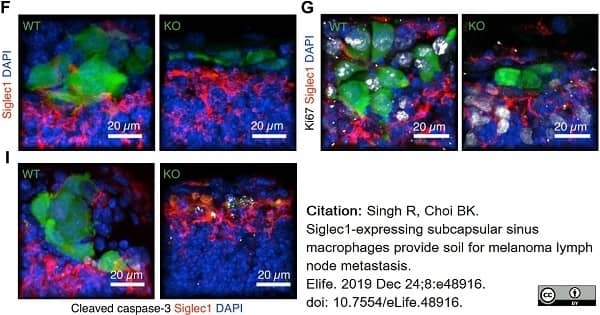
Singh, R. & Choi, B.K. (2019) Siglec1-expressing subcapsular sinus macrophages provide soil for melanoma lymph node metastasis.
Elife. 8: e48916. -
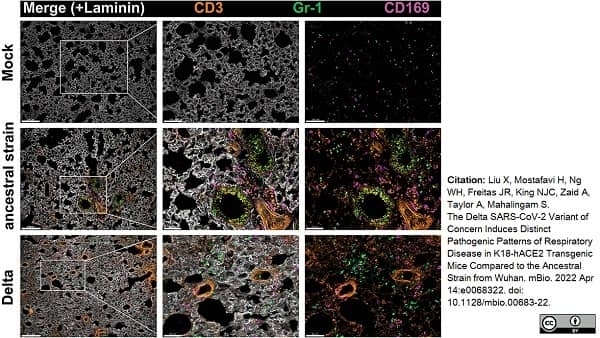
Liu, X. et al. (2022) The Delta SARS-CoV-2 Variant of Concern Induces Distinct Pathogenic Patterns of Respiratory Disease in K18-hACE2 Transgenic Mice Compared to the Ancestral Strain from Wuhan.
mBio. : e0068322. -
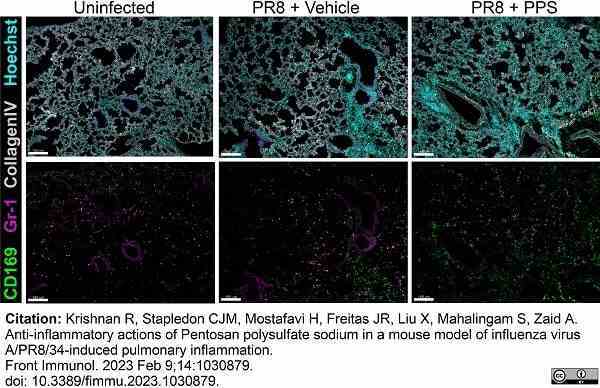
Krishnan, R. et al. (2023) Anti-inflammatory actions of Pentosan polysulfate sodium in a mouse model of influenza virus A/PR8/34-induced pulmonary inflammation.
Front Immunol. 14: 1030879. -
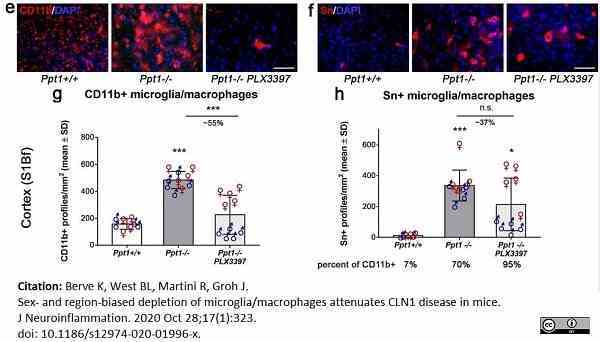
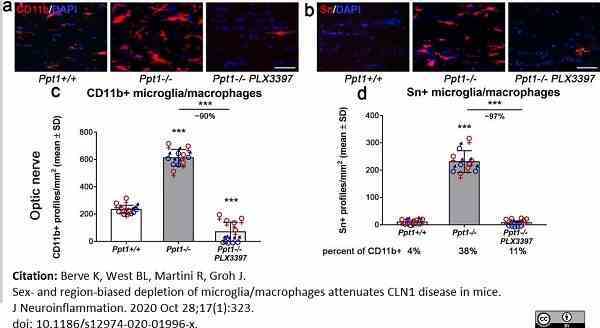
Berve, K. et al. (2020) Sex- and region-biased depletion of microglia/macrophages attenuates CLN1 disease in mice.
J Neuroinflammation. 17 (1): 323.
- Synonyms
- Sialoadhesin
- RRID
- AB_322416
- UniProt
- Q62230
- Entrez Gene
- Siglec1
- GO Terms
- GO:0005886 plasma membrane
- GO:0005515 protein binding
- GO:0007155 cell adhesion
- GO:0016021 integral to membrane
- GO:0005576 extracellular region
- GO:0005529 sugar binding
- GO:0006897 endocytosis
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Mouse ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up