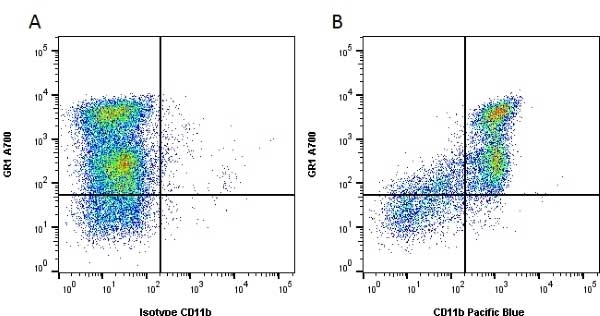
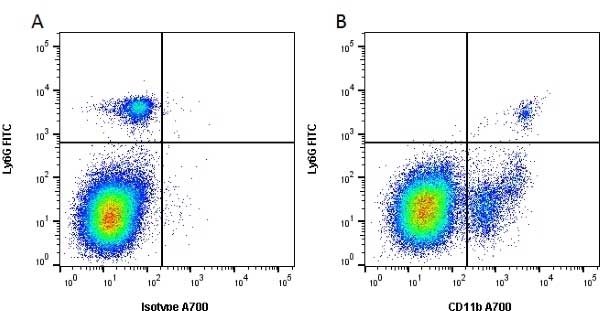
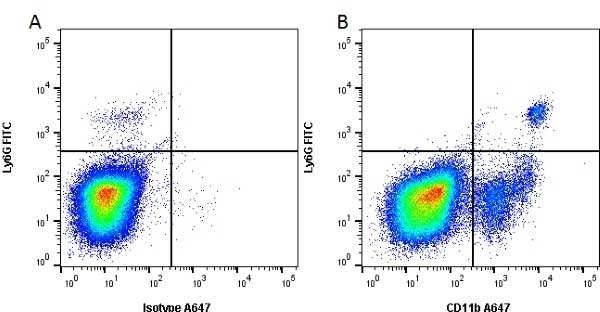
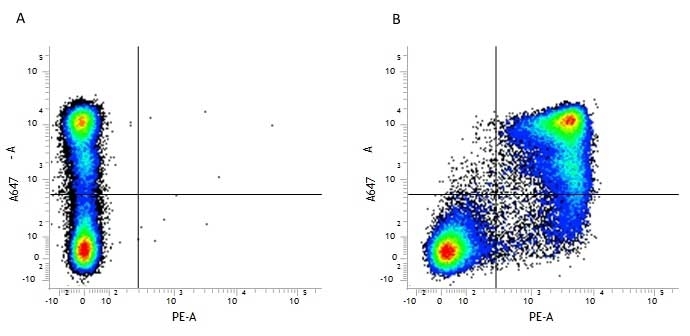
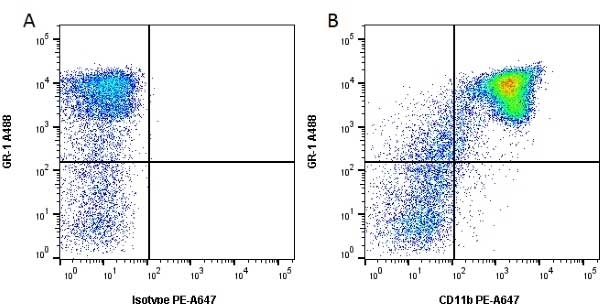
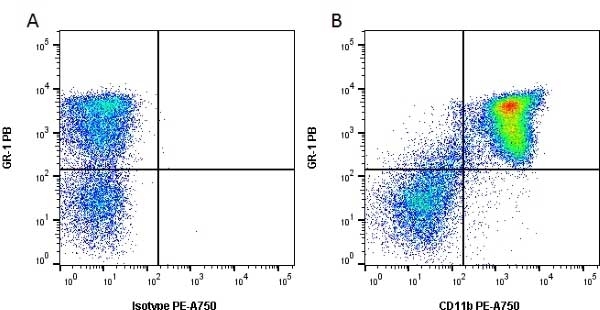
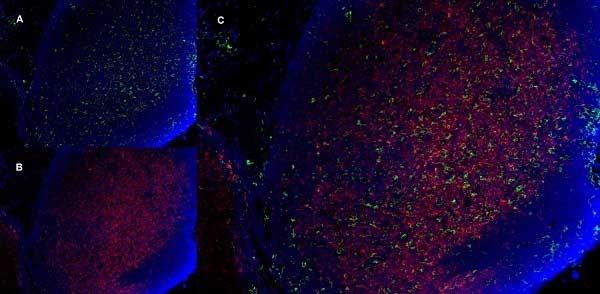
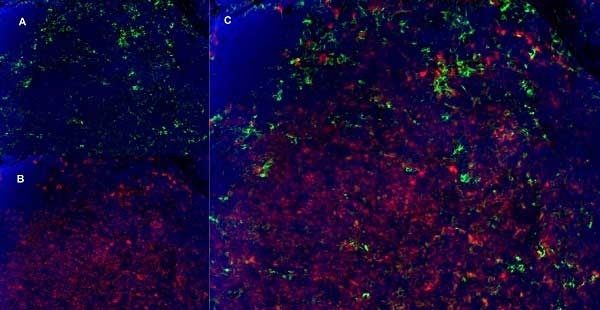
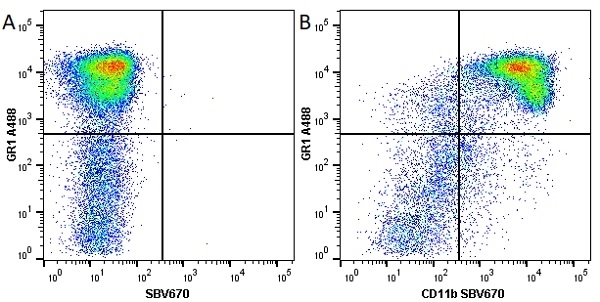
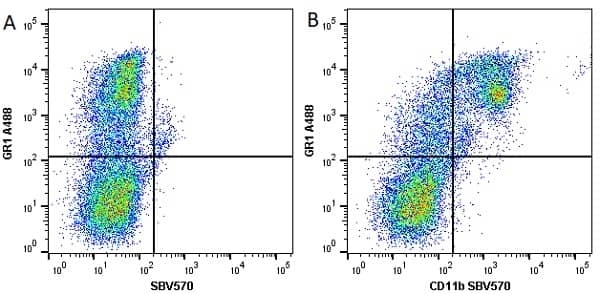
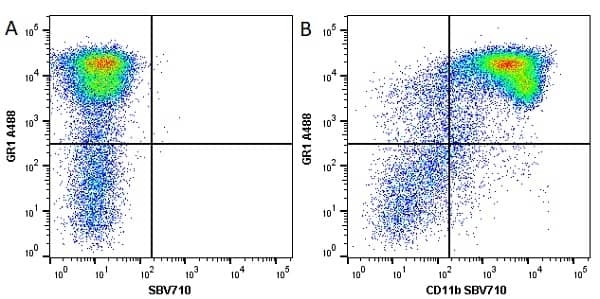
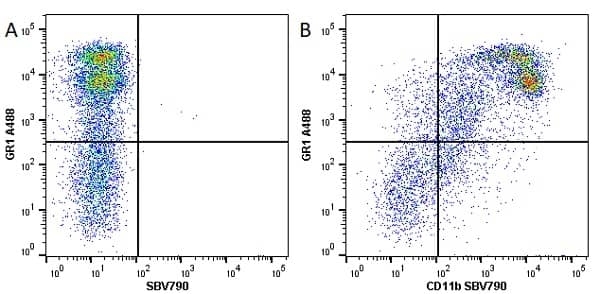
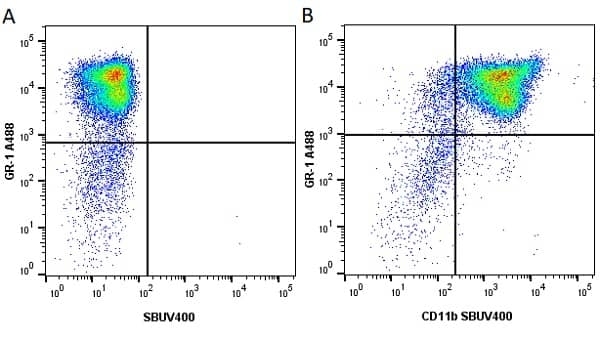
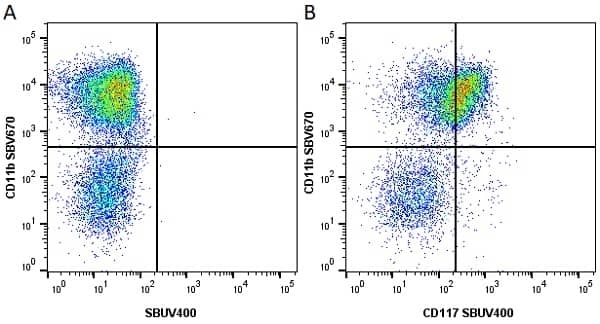
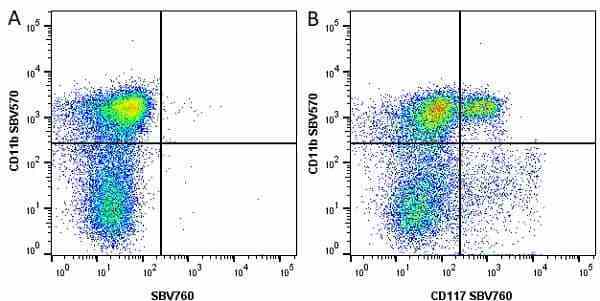
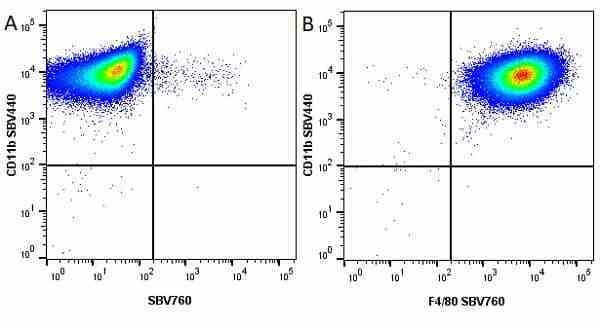
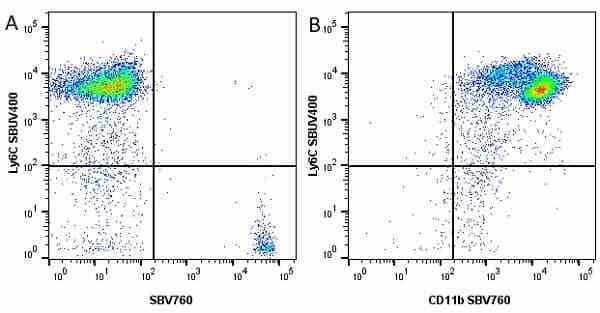
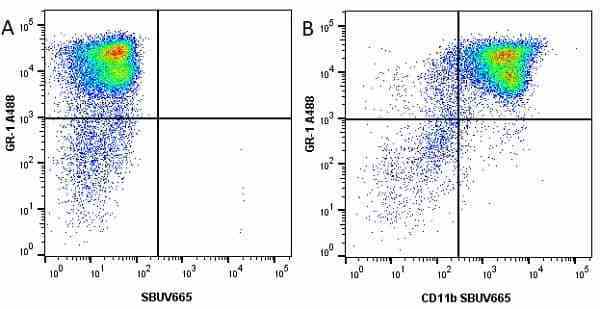
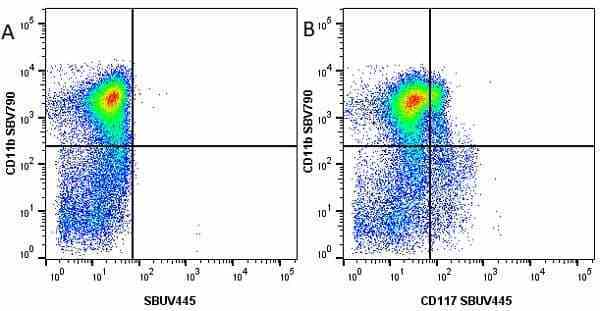
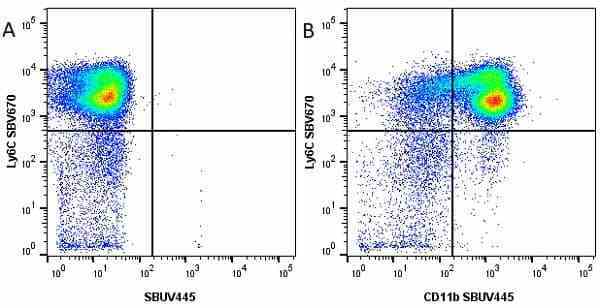
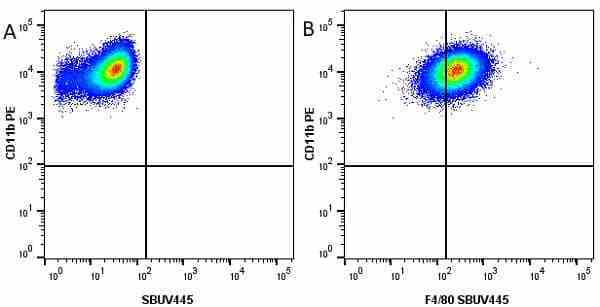
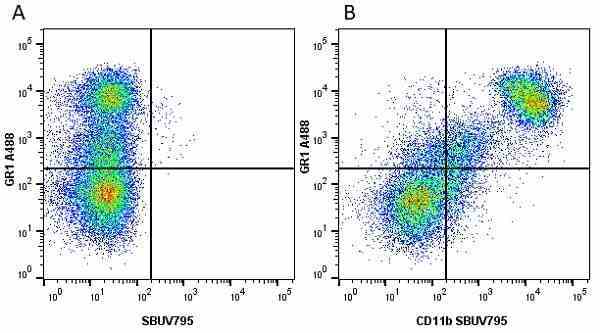
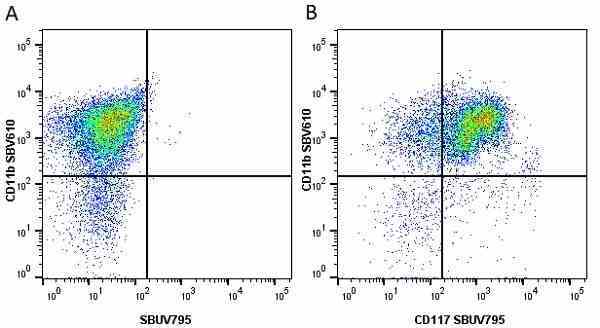
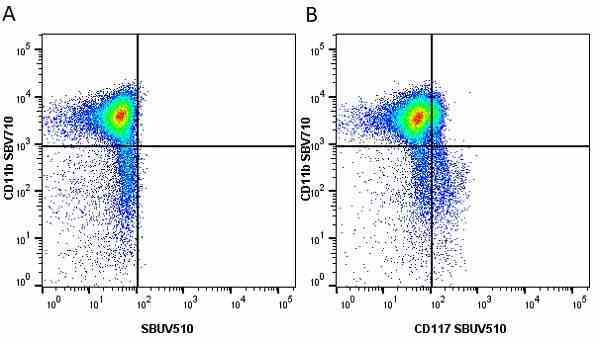
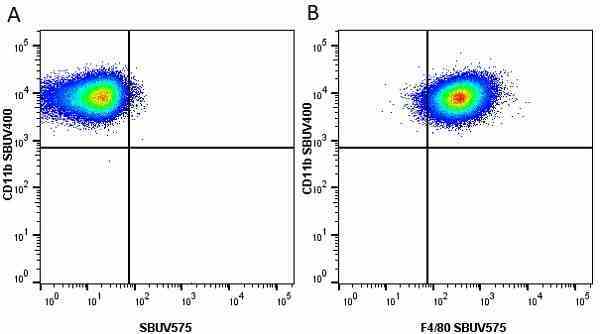
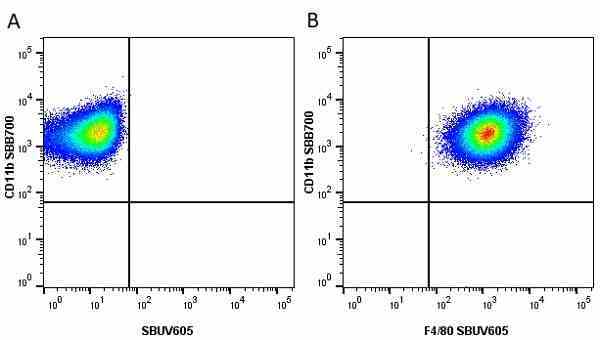
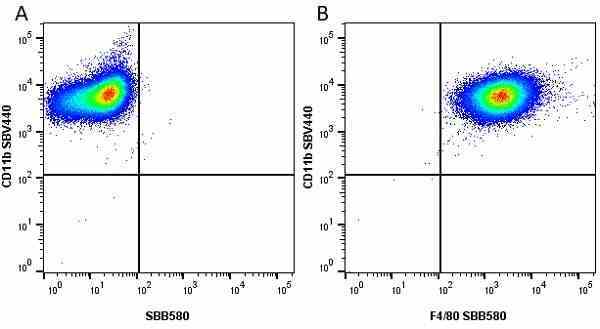
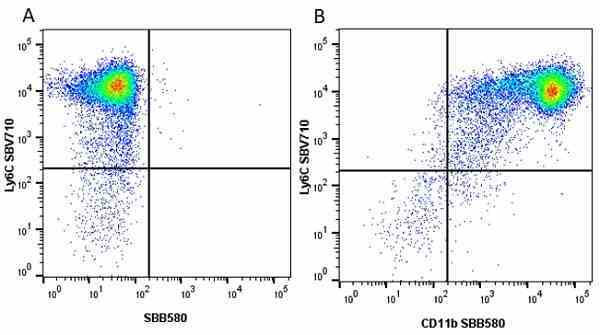
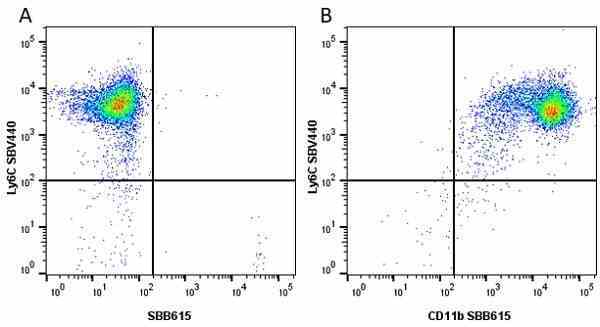
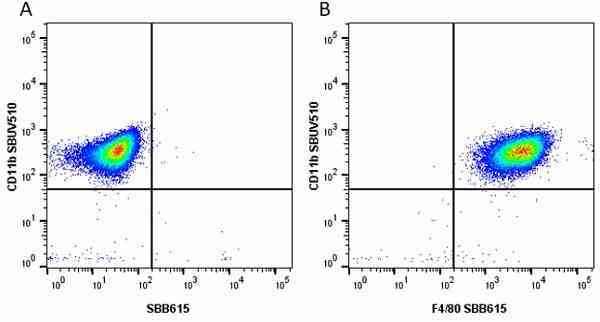
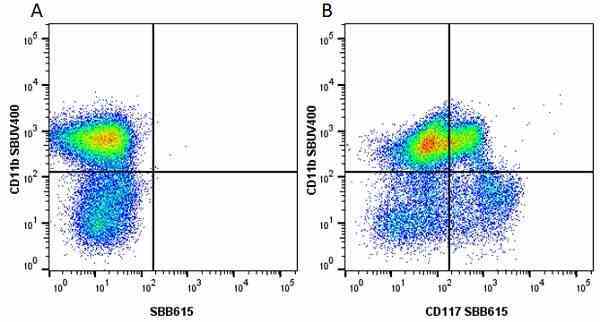
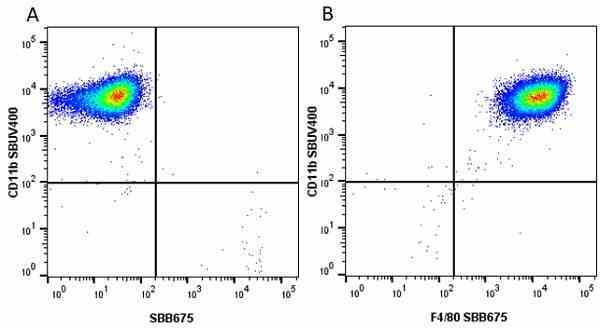
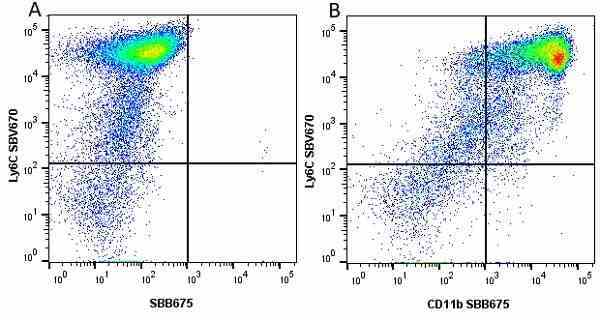
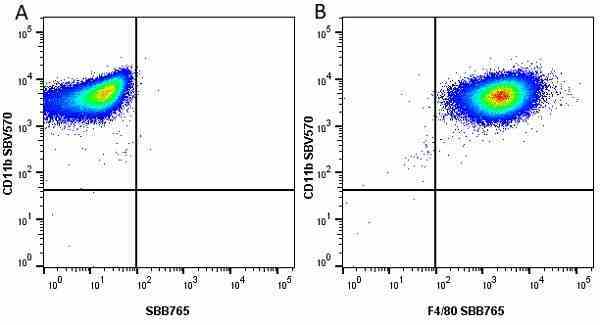
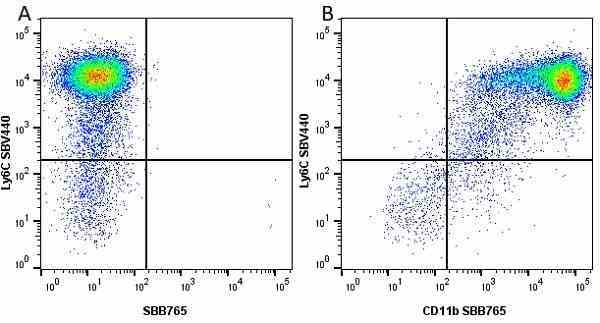
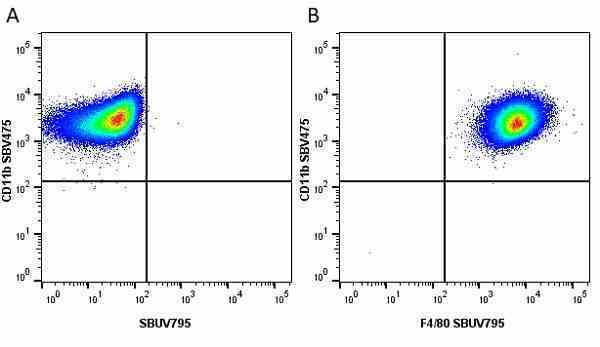
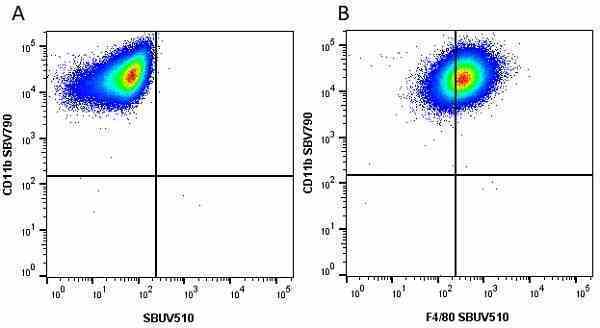
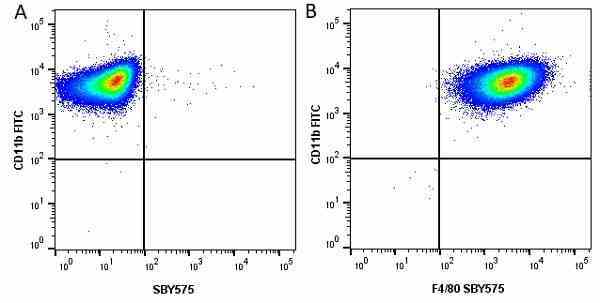
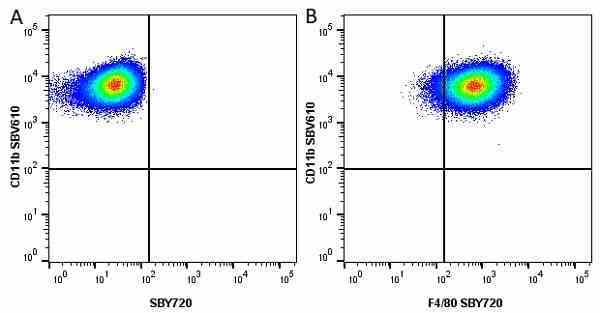
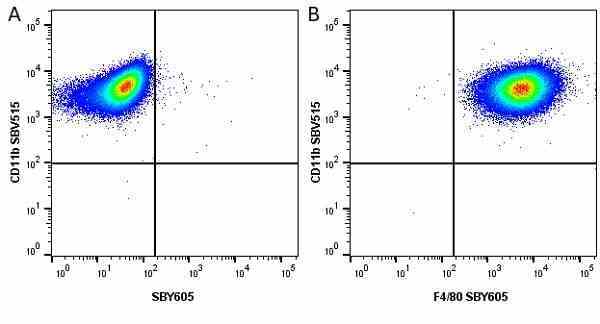
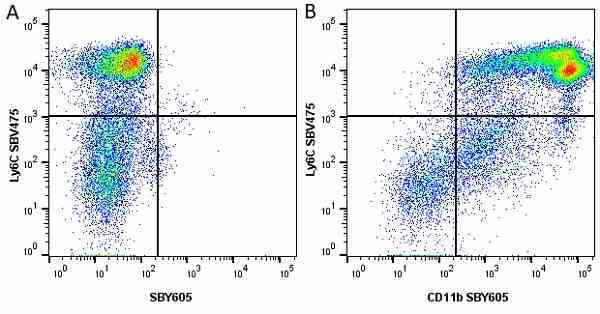
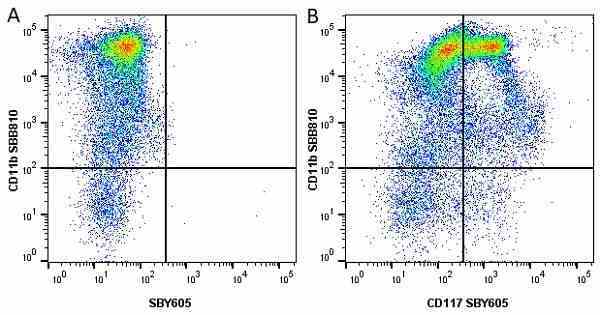
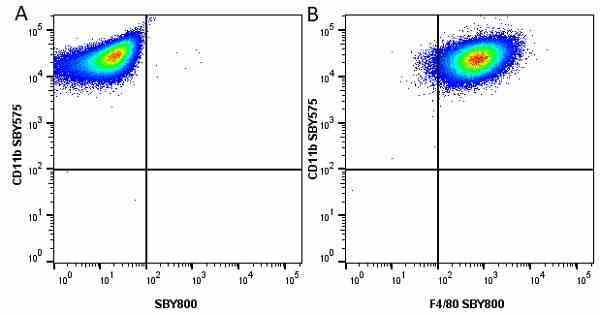
CD11b antibody | 5C6










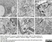


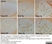



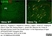
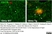


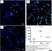


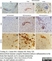
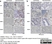










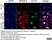













































Rat anti Mouse CD11b
- Product Type
- Monoclonal Antibody
- Clone
- 5C6
- Isotype
- IgG2b
- Specificity
- CD11b
| Rat anti Mouse CD11b antibody, clone 5C6 recognizes CD11b, also known as the integrin alpha M chain. CD11b is implicated in various adhesive interactions of monocytes, macrophages and granulocytes as well as in mediating the uptake of complement-coated particles. Rat anti Mouse CD11b antibody, clone 5C6 immunoprecipitates a heterodimer of ~165 and ~95 kDa. This clone also exhibits various functional properties, reportedly inhibiting adhesion in vitro and inflammatory recruitment in vivo. Rat anti Mouse CD11b antibody, clone 5C6 also inhibits delayed hypersensitivity, potentiates bacterial infections and inhibits type 1 diabetes. |

Our CD11b (5C6) Antibody has been referenced in >161 publications* *Based on June 2020 data from CiteAb's antibody search engine. |
- Target Species
- Mouse
- Species Cross-Reactivity
-
Target Species Cross Reactivity Human - N.B. Antibody reactivity and working conditions may vary between species.
- Product Form
- Purified IgG - liquid
- Preparation
- MCA711G, MCA711: Purified IgG prepared by affinity chromatography on Protein G from tissue culture supernatant
- MCA711GT: Purified IgG prepared by ion exchange chromatography from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
0.09% Sodium Azide - Carrier Free
- Yes
- Immunogen
- Thioglycollate-elicited peritoneal macrophages (TPM)
- Approx. Protein Concentrations
- IgG concentration 1 mg/ml
- Fusion Partners
- Spleen cells from AO rats were fused with cells of the Y3 rat myeloma cell line
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Flow Cytometry | 1/100 | ||
| Immunofluorescence | |||
| Immunohistology - Frozen | |||
| Immunoprecipitation |
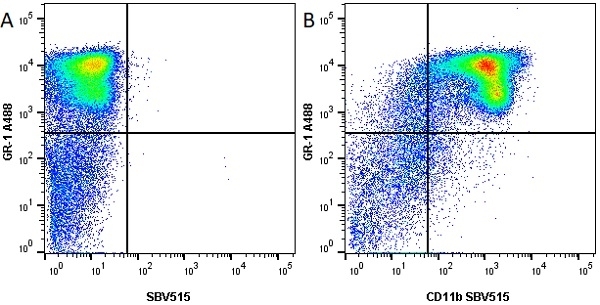
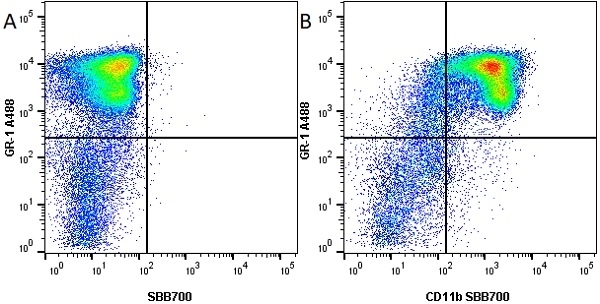
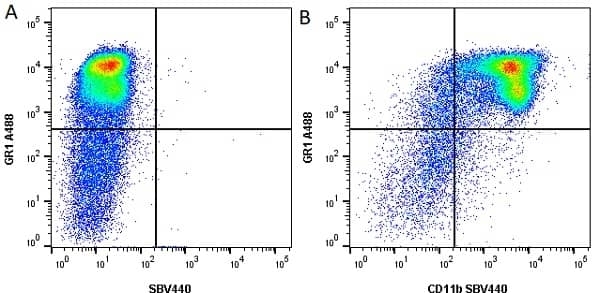
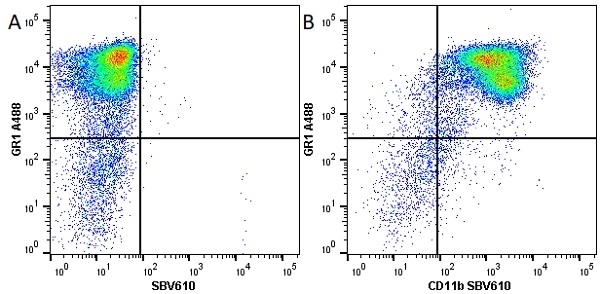
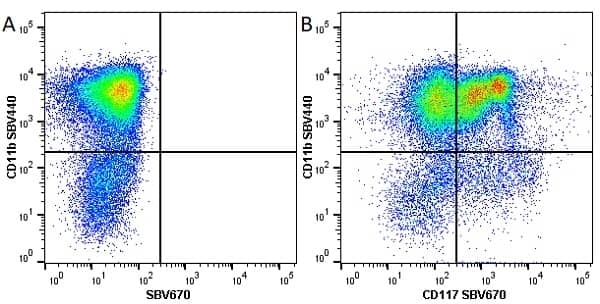
- Flow Cytometry
- Use 10ul of the suggested working dilution to label 106 cells in 100ul
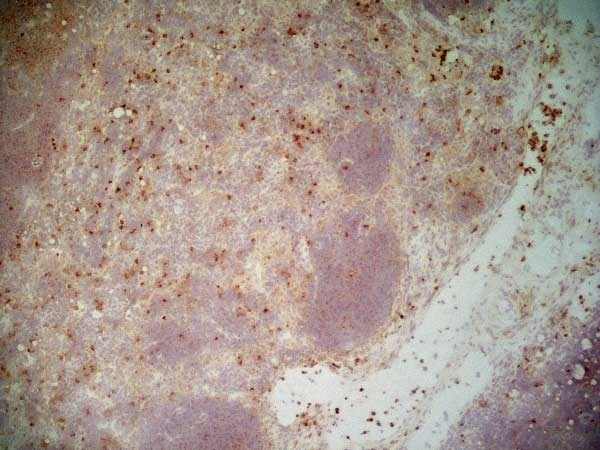
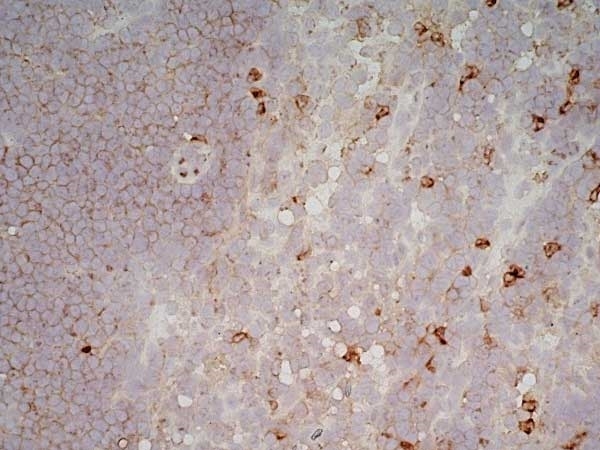
- Histology Positive Control Tissue
- Spleen
References for CD11b antibody
-
Rosen, H. and Gordon, S. (1987) Monoclonal antibody to the murine type 3 complement receptor inhibits adhesion of myelomonocytic cells in vitro and inflammatory cell recruitment in vivo.
J Exp Med. 166: 1685-701. -
Rosen, H. et al. (1989) Antibody to the murine type 3 complement receptor inhibits T lymphocyte-dependent recruitment of myelomonocytic cells in vivo.
J Exp Med. 169: 535-48. -
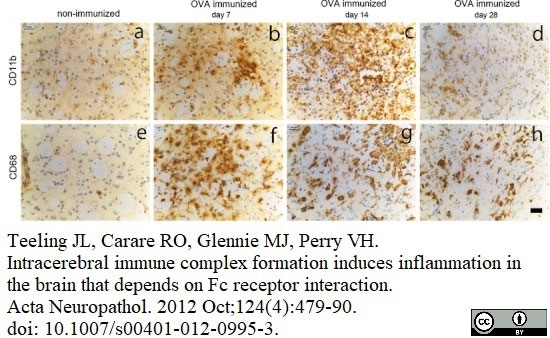
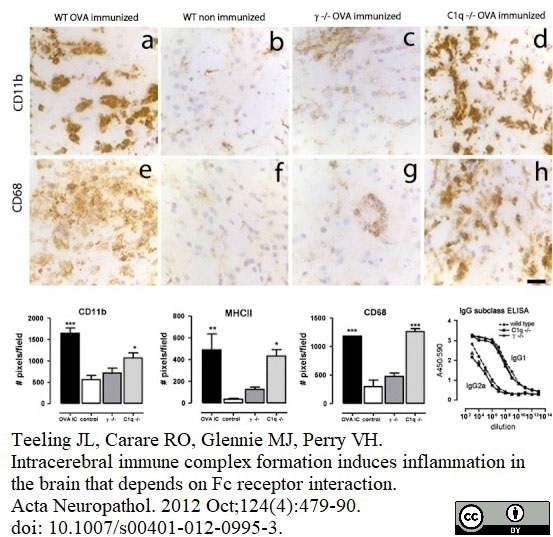
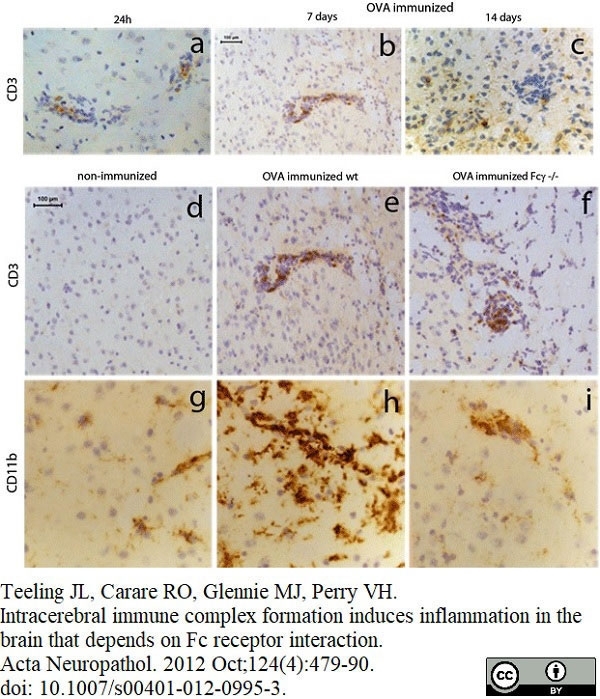
Serafini, B. et al. (2000) Intracerebral recruitment and maturation of dendritic cells in the onset and progression of experimental autoimmune encephalomyelitis.
Am J Pathol. 157: 1991-2002. -
Platt, N. et al. (2000) Apoptotic thymocyte clearance in scavenger receptor class A-deficient mice is apparently normal.
J Immunol. 164: 4861-7. -
Carenini, S. et al. (2001) The role of macrophages in demyelinating peripheral nervous system of mice heterozygously deficient in p0.
J Cell Biol. 152: 301-8. -
Engwerda, C.R. et al. (2002) Locally up-regulated lymphotoxin alpha, not systemic tumor necrosis factor alpha, is the principle mediator of murine cerebral malaria.
J Exp Med. 195: 1371-7. -
Mittal, A. et al. (2003) CD11b+ cells are the major source of oxidative stress in UV radiation-irradiated skin: possible role in photoaging and photocarcinogenesis.
Photochem Photobiol. 77 (3): 259-64. -
Lesnik, P. et al. (2003) Decreased atherosclerosis in CX3CR1-/- mice reveals a role for fractalkine in atherogenesis.
J Clin Invest. 111: 333-40.
View The Latest Product References
-
Di Filippo, C. et al. (2005) Cannabinoid CB2 receptor activation reduces mouse myocardial ischemia-reperfusion injury: involvement of cytokine/chemokines and PMN.
J Leukoc Biol. 75 (3): 453-9. -
Lin, H.H. et al. (2005) The macrophage F4/80 receptor is required for the induction of antigen-specific efferent regulatory T cells in peripheral tolerance.
J Exp Med. 201: 1615-25. -
Mennini, T. et al. (2006) Nonhematopoietic erythropoietin derivatives prevent motoneuron degeneration in vitro and in vivo.
Mol Med. 12: 153-60. -
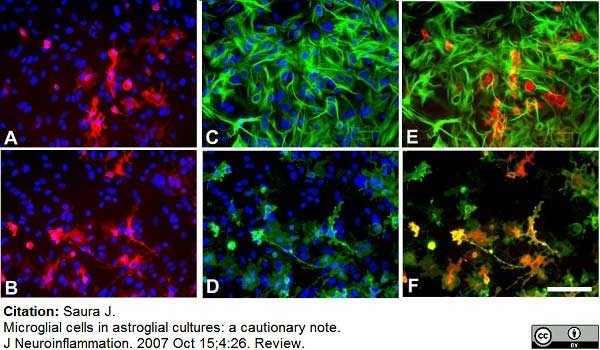
Saura, J. (2007) Microglial cells in astroglial cultures: a cautionary note.
J Neuroinflammation. 4: 26. -
Kondo, Y. et al. (2007) Osteopetrotic (op/op) mice have reduced microglia, no Abeta deposition, and no changes in dopaminergic neurons.
J Neuroinflammation. 4: 31. -
Halle, A. et al. (2008) The NALP3 inflammasome is involved in the innate immune response to amyloid-beta.
Nat Immunol. 9: 857-65. -
Devey, L. et al. (2008) Tissue-resident Macrophages protect the Liver From Ischemia Reperfusion Injury via a Heme Oxygenase-1-Dependent mechanism.
Mol Ther. 1: 65-72. -
Hickman, S.E. et al. (2008) Microglial dysfunction and defective beta-amyloid clearance pathways in aging Alzheimer's disease mice.
J Neurosci. 28 (33): 8354-60. -
Khorooshi, R. et al. (2008) NF-kappaB-driven STAT2 and CCL2 expression in astrocytes in response to brain injury.
J Immunol.181: 7284-91. -
Basso, A.S. et al. (2008) Reversal of axonal loss and disability in a mouse model of progressive multiple sclerosis.
J Clin Invest. 118: 1532-43. -
Weberpals, M. et al. (2009) NOS2 gene deficiency protects from sepsis-induced long-term cognitive deficits.
J Neurosci. 29: 14177-84. -
Valerio, A. et al. (2009) Leptin is induced in the ischemic cerebral cortex and exerts neuroprotection through NF-kappaB/c-Rel-dependent transcription.
Stroke. 40: 610-7. -
Traka, .M. et al (2010) A genetic mouse model of adult-onset, pervasive central nervous system demyelination with robust remyelination.
Brain. 133: 3017-29. -
Kim, D. et al. (2010) NADPH oxidase 2-derived reactive oxygen species in spinal cord microglia contribute to peripheral nerve injury-induced neuropathic pain.
Proc Natl Acad Sci U S A. 107: 14851-6. -
Lu, J. et al. (2010) Ursolic acid attenuates D-galactose-induced inflammatory response in mouse prefrontal cortex through inhibiting AGEs/RAGE/NF-κB pathway activation.
Cereb Cortex. 20: 2540-8. -
Heneka, M.T. et al. (2010) Locus ceruleus controls Alzheimer's disease pathology by modulating microglial functions through norepinephrine.
Proc Natl Acad Sci U S A. 107: 6058-63. -
Dohi, K. et al. (2010) Gp91phox (NOX2) in classically activated microglia exacerbates traumatic brain injury.
J Neuroinflammation. 7: 41. -
Cui, Y.F. et al. (2010) Embryonic stem cell-derived L1 overexpressing neural aggregates enhance recovery in Parkinsonian mice.
Brain. 133: 189-204. -
Wu, T. et al. (2011) Expression and cellular localization of cyclooxygenases and prostaglandin E synthases in the hemorrhagic brain.
J Neuroinflammation. 8:22. -
Tysseling, V.M.et al. (2011) SDF1 in the dorsal corticospinal tract promotes CXCR4+ cell migration after spinal cord injury.
J Neuroinflammation. 8:16. -
McDonald, J.U. et al. (2011) In vivo functional analysis and genetic modification of in vitro-derived mouse neutrophils.
FASEB J. 25: 1972-82. -
Terwel, D. et al. (2011) Critical Role of Astroglial Apolipoprotein E and Liver X Receptor-{alpha} Expression for Microglial A{beta} Phagocytosis.
J Neurosci. 31: 7049-59. -
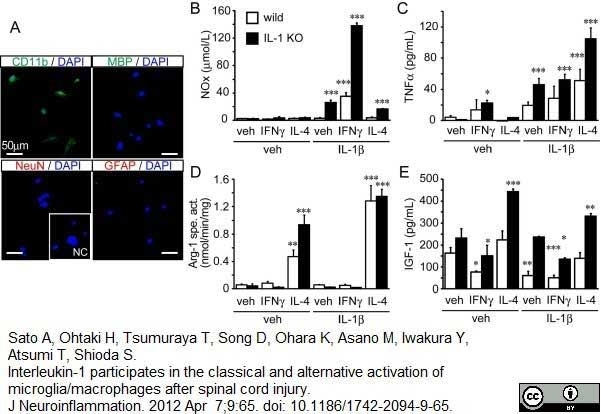
Sato, A. et al. (2012) Interleukin-1 participates in the classical and alternative activation of microglia/macrophages after spinal cord injury.
J Neuroinflammation. 9: 65. -
Heydenreich, N. et al. (2012) C1-inhibitor protects from brain ischemia-reperfusion injury by combined antiinflammatory and antithrombotic mechanisms.
Stroke. 43 (9): 2457-67. -
Yamanaka M et al. (2012) PPARγ/RXRα-induced and CD36-mediated microglial amyloid-β phagocytosis results in cognitive improvement in amyloid precursor protein/presenilin 1 mice.
J Neurosci. 32 (48): 17321-31. -
Mencl, S. et al. (2014) FTY720 does not protect from traumatic brain injury in mice despite reducing posttraumatic inflammation.
J Neuroimmunol. 274 (1-2): 125-31. -
Natrajan, M.S. et al. (2015) Retinoid X receptor activation reverses age-related deficiencies in myelin debris phagocytosis and remyelination.
Brain. 138 (Pt 12): 3581-97. -
Kan, M.J. et al. (2015) Arginine deprivation and immune suppression in a mouse model of Alzheimer's disease.
J Neurosci. 35 (15): 5969-82. -
Babcock, A.A. et al. (2015) Cytokine-producing microglia have an altered beta-amyloid load in aged APP/PS1 Tg mice.
Brain Behav Immun. 48: 86-101. -
Kaindlstorfer, C. et al. (2015) Failure of Neuroprotection Despite Microglial Suppression by Delayed-Start Myeloperoxidase Inhibition in a Model of Advanced Multiple System Atrophy: Clinical Implications.
Neurotox Res. 28 (3): 185-94. -
Liu, Z. et al. (2016) Transforming growth factor-β1 acts via TβR-I on microglia to protect against MPP(+)-induced dopaminergic neuronal loss.
Brain Behav Immun. 51: 131-43. -
Garcia-Mesa, Y. et al. (2017) Immortalization of primary microglia: a new platform to study HIV regulation in the central nervous system.
J Neurovirol. 23 (1): 47-66. -
Lu, Y. et al. (2016) Annexin A10 is involved in the development and maintenance of neuropathic pain in mice.
Neurosci Lett. 631: 1-6. -
Hristova M et al. (2016) Inhibition of Signal Transducer and Activator of Transcription 3 (STAT3) reduces neonatal hypoxic-ischaemic brain damage.
J Neurochem. 136 (5): 981-994. -
Ye, M. et al. (2016) Neuroprotective effects of bee venom phospholipase A2 in the 3xTg AD mouse model of Alzheimer's disease.
J Neuroinflammation. 13 (1): 10. -
Nagai, J. et al. (2016) Inhibition of CRMP2 phosphorylation repairs CNS by regulating neurotrophic and inhibitory responses.
Exp Neurol. 277: 283-95. -
Tachibana, M. et al. (2016) Rescuing effects of RXR agonist bexarotene on aging-related synapse loss depend on neuronal LRP1.
Exp Neurol. 277: 1-9. -
Sun, H. et al. (2016) Aquaporin-4 mediates communication between astrocyte and microglia: Implications of neuroinflammation in experimental Parkinson's disease.
Neuroscience. 317: 65-75. -
Shinohara, M. et al. (2016) APOE2 eases cognitive decline during Aging: Clinical and preclinical evaluations.
Ann Neurol. 79 (5): 758-74. -
Kami, K. et al. (2016) Histone Acetylation in Microglia Contributes to Exercise-Induced Hypoalgesia in Neuropathic Pain Model Mice.
J Pain. 17 (5): 588-99. -
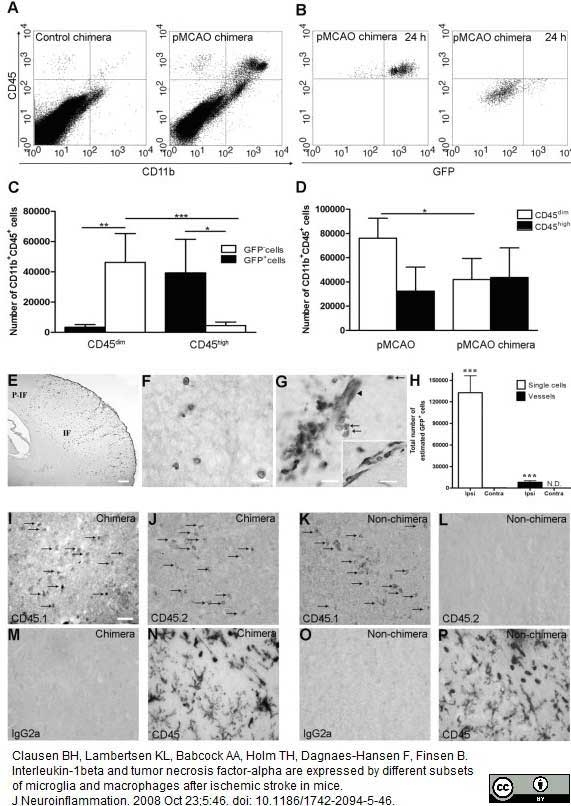
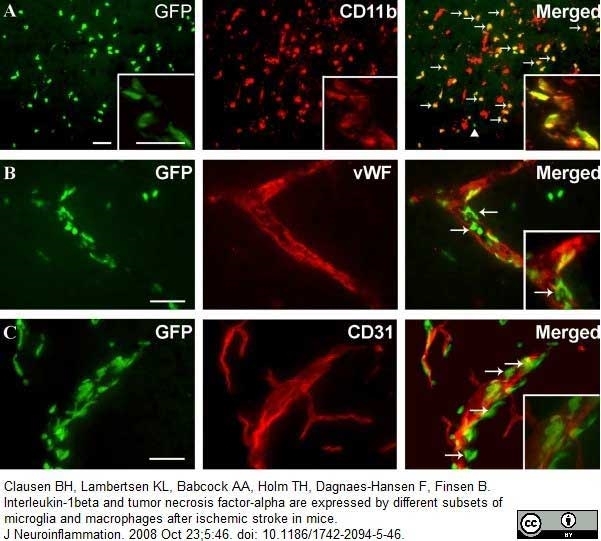
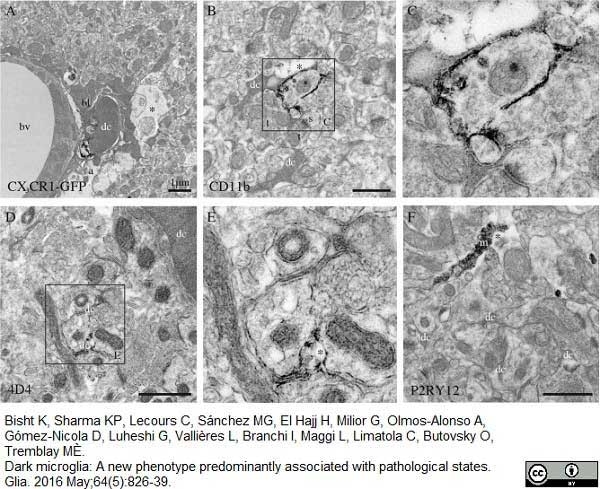
Bisht, K. et al. (2016) Dark microglia: A new phenotype predominantly associated with pathological states.
Glia. 64 (5): 826-39. -
Hilla, A.M. et al. (2017) Microglia Are Irrelevant for Neuronal Degeneration and Axon Regeneration after Acute Injury.
J Neurosci. 37 (25): 6113-24. -
Paizs, M. et al. (2017) Axotomy Leads to Reduced Calcium Increase and Earlier Termination of CCL2 Release in Spinal Motoneurons with Upregulated Parvalbumin Followed by Decreased Neighboring Microglial Activation.
CNS Neurol Disord Drug Targets. 16 (3): 356-67. -
Pulido-Salgado, M. et al. (2017) Myeloid C/EBPβ deficiency reshapes microglial gene expression and is protective in experimental autoimmune encephalomyelitis.
J Neuroinflammation. 14 (1): 54. -
Thomsen, M.S. et al. (2017) Synthesis and deposition of basement membrane proteins by primary brain capillary endothelial cells in a murine model of the blood-brain barrier.
J Neurochem. 140 (5): 741-754. -
Laurent, C. et al. (2017) Hippocampal T cell infiltration promotes neuroinflammation and cognitive decline in a mouse model of tauopathy.
Brain. 140 (Pt 1): 184-200. -
Schuhmann, M.K. et al. (2017) Blocking of platelet glycoprotein receptor Ib reduces "thrombo-inflammation" in mice with acute ischemic stroke.
J Neuroinflammation. 14 (1): 18. -
Rabl, R. et al. (2017) Early start of progressive motor deficits in Line 61 α-synuclein transgenic mice.
BMC Neurosci. 18 (1): 22. -
Crépeaux, G. et al. (2017) Non-linear dose-response of aluminium hydroxide adjuvant particles: Selective low dose neurotoxicity.
Toxicology. 375: 48-57. -
Da Ros, F. et al. (2017) Targeting Interleukin-1β Protects from Aortic Aneurysms Induced by Disrupted Transforming Growth Factor β Signaling.
Immunity. 47 (5): 959-973.e9. -
Hou, L. et al. (2018) Taurine protects noradrenergic locus coeruleus neurons in a mouse Parkinson's disease model by inhibiting microglial M1 polarization.
Amino Acids. 50 (5): 547-556. -
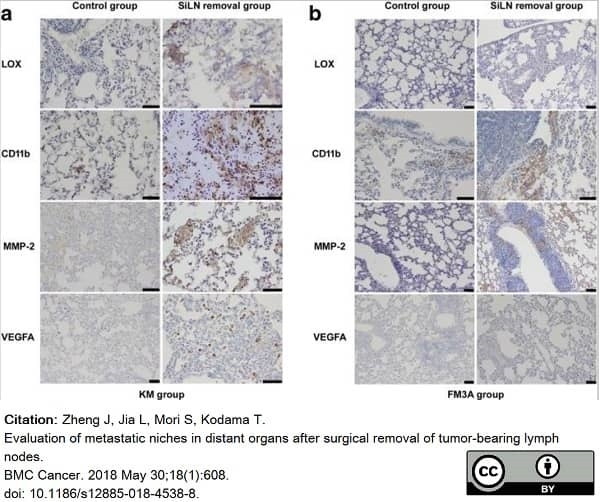
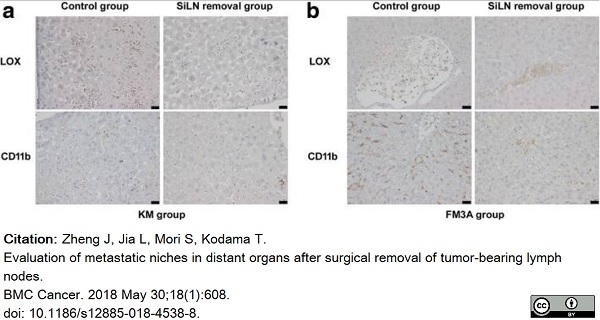
Zheng, J. et al. (2018) Evaluation of metastatic niches in distant organs after surgical removal of tumor-bearing lymph nodes.
BMC Cancer. 18 (1): 608. -
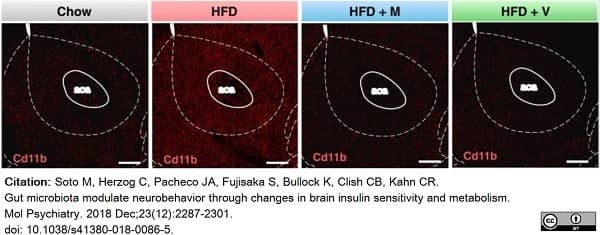
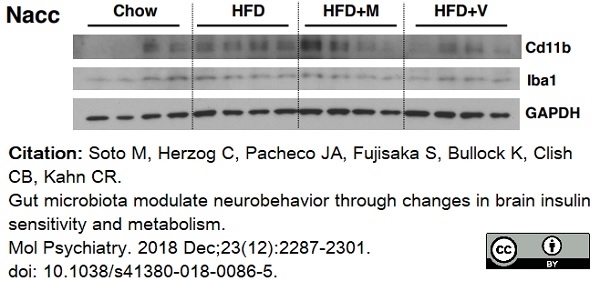
Soto, M. et al. (2018) Gut microbiota modulate neurobehavior through changes in brain insulin sensitivity and metabolism.
Mol Psychiatry. 23 (12): 2287-301. -
Mouton-Liger, F. et al. (2018) Parkin deficiency modulates NLRP3 inflammasome activation by attenuating an A20-dependent negative feedback loop.
Glia. 66 (8): 1736-51. -
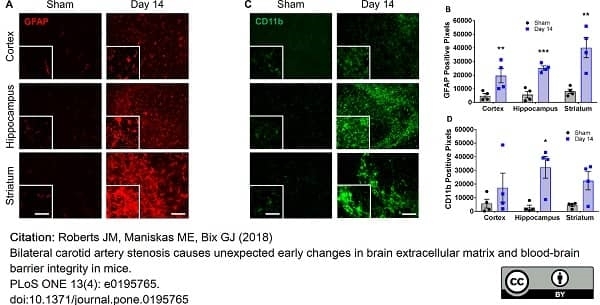
Roberts, J.M. et al. (2018) Bilateral carotid artery stenosis causes unexpected early changes in brain extracellular matrix and blood-brain barrier integrity in mice.
PLoS One. 13 (4): e0195765. -
Cope, E.C. et al. (2018) Microglia Play an Active Role in Obesity-Associated Cognitive Decline.
J Neurosci. 38 (41): 8889-904. -
Song, S. et al. (2019) Noradrenergic dysfunction accelerates LPS-elicited inflammation-related ascending sequential neurodegeneration and deficits in non-motor/motor functions.
Brain Behav Immun. 81: 374-87. -
Liu, Z. et al. (2019) IL-17A exacerbates neuroinflammation and neurodegeneration by activating microglia in rodent models of Parkinson's disease.
Brain Behav Immun. 81: 630-45. -
Gomez-Nicola, D. et al. (2019) Measuring Microglial Turnover in the Adult Brain.
Methods Mol Biol. 2034: 207-15. -
Di Benedetto, G. et al. (2019) Beneficial effects of curtailing immune susceptibility in an Alzheimer's disease model.
J Neuroinflammation. 16 (1): 166. -
Bernier, L.P. et al. (2019) Nanoscale Surveillance of the Brain by Microglia via cAMP-Regulated Filopodia.
Cell Rep. 27 (10): 2895-2908.e4. -
Tunesi, M. et al. (2019) Hydrogel-based delivery of Tat-fused protein Hsp70 protects dopaminergic cells in vitro and in a mouse model of Parkinson’s disease
NPG Asia Materials. 11: 28. -
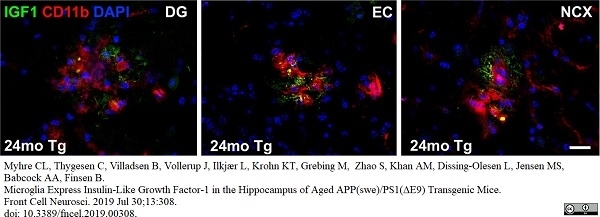
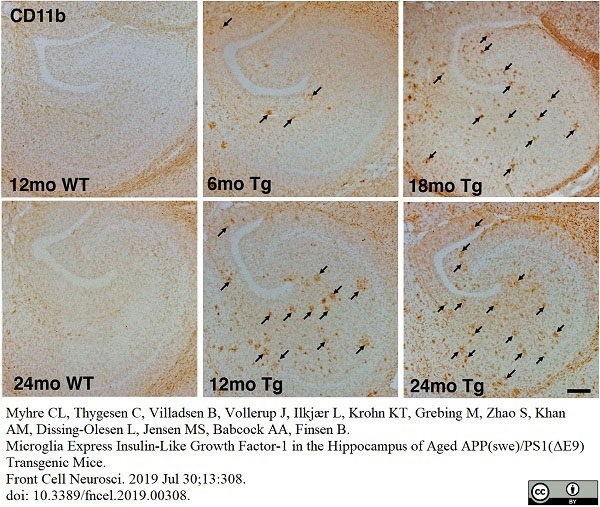
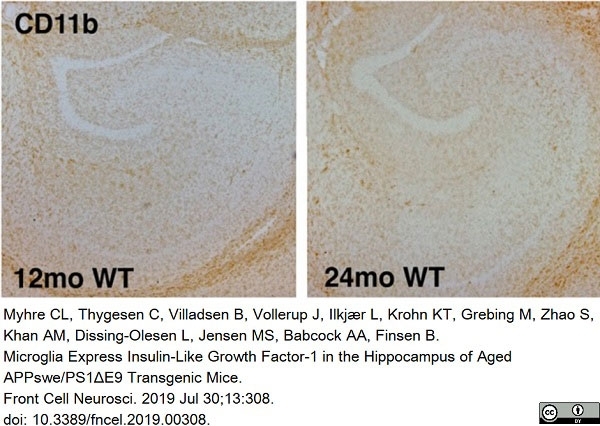
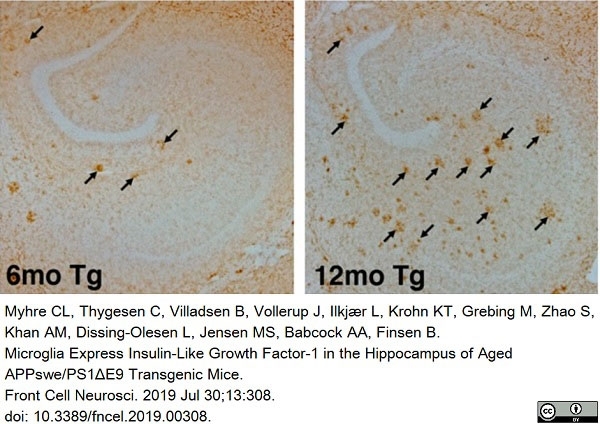
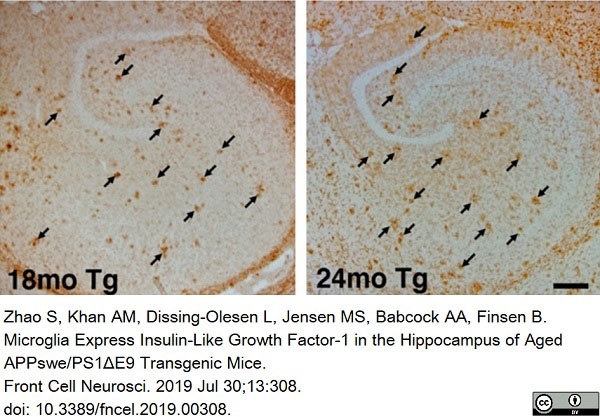
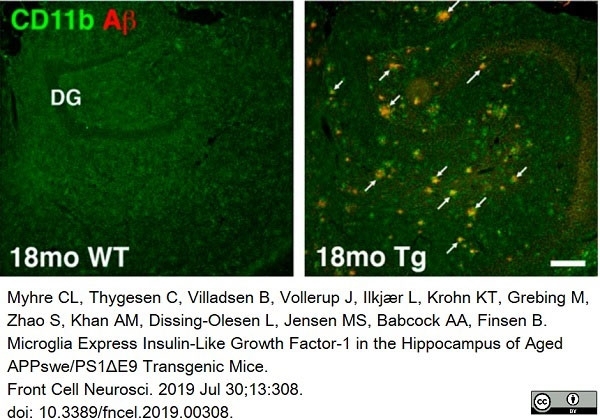
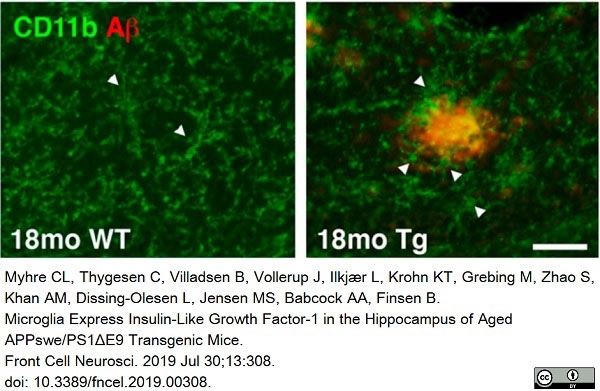
Myhre, C.L. et al. (2019) Microglia Express Insulin-Like Growth Factor-1 in the Hippocampus of Aged APPswe/PS1ΔE9 Transgenic Mice.
Front Cell Neurosci. 13: 308. -
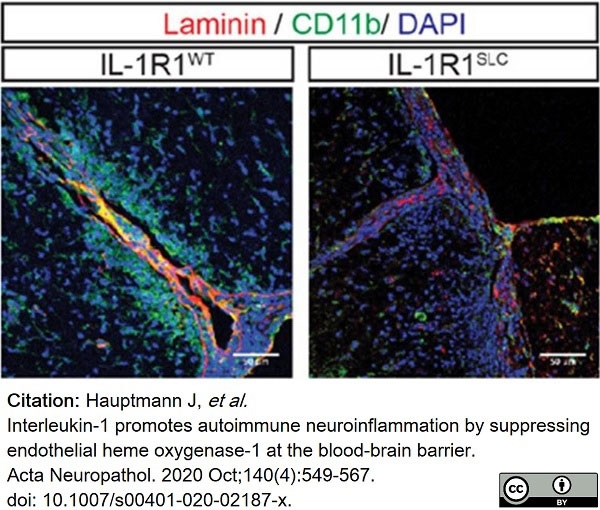
Hauptmann, J. et al. (2020) Interleukin-1 promotes autoimmune neuroinflammation by suppressing endothelial heme oxygenase-1 at the blood-brain barrier.
Acta Neuropathol. 140 (4): 549-67. -
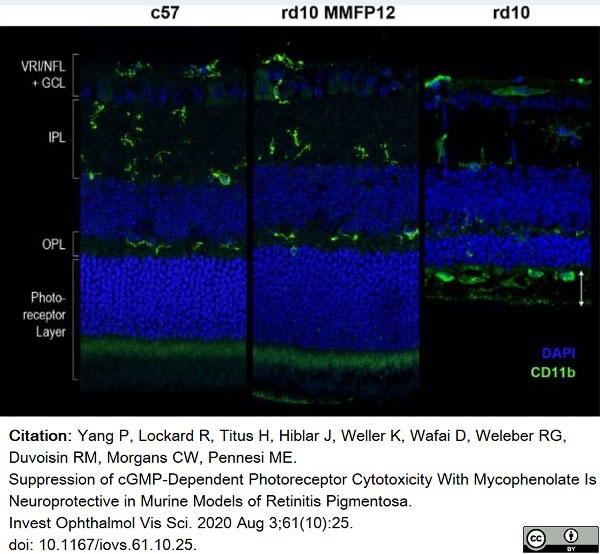
Yang, P. et al. (2020) Suppression of cGMP-Dependent Photoreceptor Cytotoxicity With Mycophenolate Is Neuroprotective in Murine Models of Retinitis Pigmentosa.
Invest Ophthalmol Vis Sci. 61 (10): 25. -
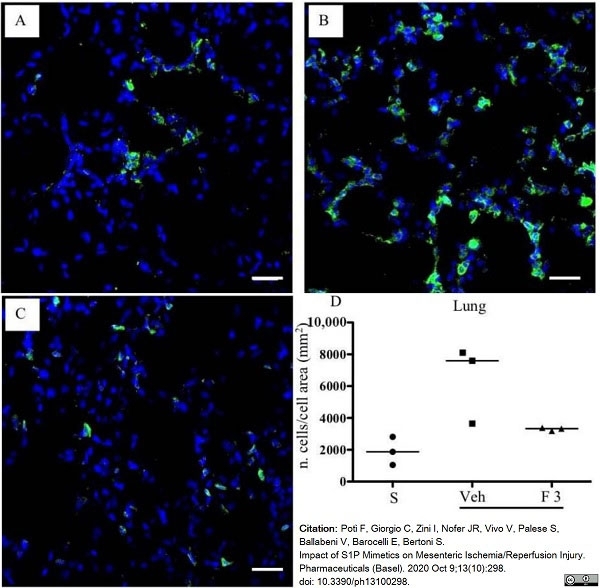
Potì, F. et al. (2020) Impact of S1P Mimetics on Mesenteric Ischemia/Reperfusion Injury.
Pharmaceuticals (Basel). 13 (10) 298. -
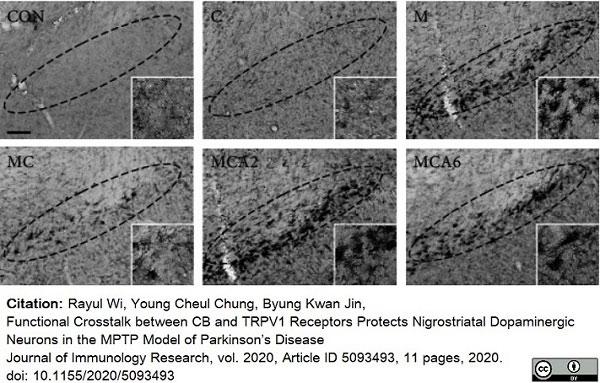
Wi, R. et al. (2020) Functional Crosstalk between CB and TRPV1 Receptors Protects Nigrostriatal Dopaminergic Neurons in the MPTP Model of Parkinson's Disease.
J Immunol Res. 2020: 5093493. -
Madore, C. et al. (2020) Essential omega-3 fatty acids tune microglial phagocytosis of synaptic elements in the mouse developing brain.
Nat Commun. 11 (1): 6133. -
Ellman, D.G. et al. (2020) Conditional Ablation of Myeloid TNF Improves Functional Outcome and Decreases Lesion Size after Spinal Cord Injury in Mice.
Cells. 9 (11): 2407. -
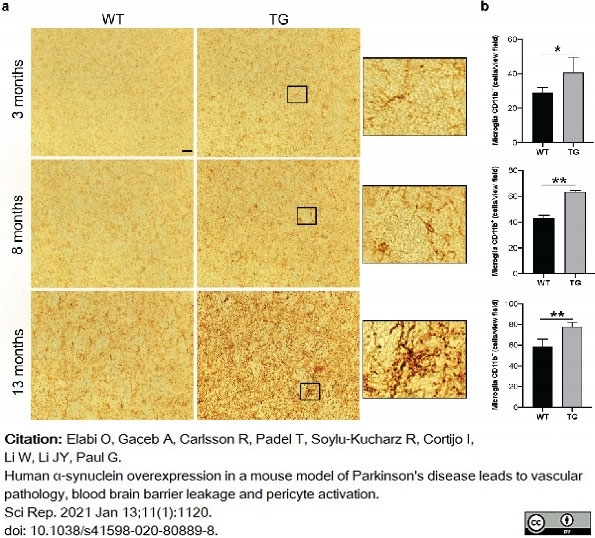
Elabi, O. et al. (2021) Human α-synuclein overexpression in a mouse model of Parkinson's disease leads to vascular pathology, blood brain barrier leakage and pericyte activation.
Sci Rep. 11 (1): 1120. -
Yoshizaki, S. et al. (2021) Microglial inflammation after chronic spinal cord injury is enhanced by reactive astrocytes via the fibronectin/β1 integrin pathway.
J Neuroinflammation. 18 (1): 12. -
Mañucat-Tan, N. et al. (2021) Hypochlorite-induced aggregation of fibrinogen underlies a novel antioxidant role in blood plasma.
Redox Biol. 40: 101847. -
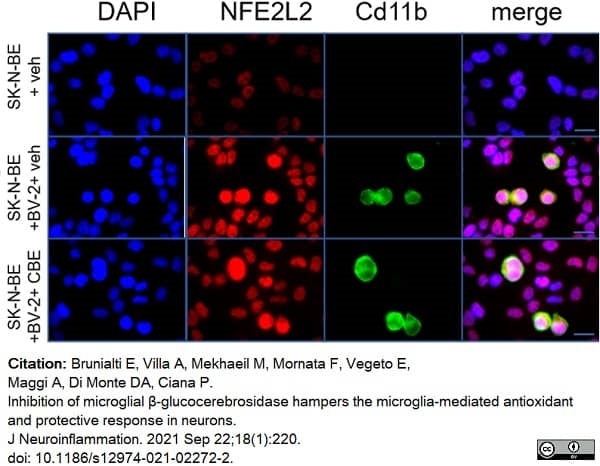
Brunialti, E. et al. (2021) Inhibition of microglial β-glucocerebrosidase hampers the microglia-mediated antioxidant and protective response in neurons.
J Neuroinflammation. 18 (1): 220. -
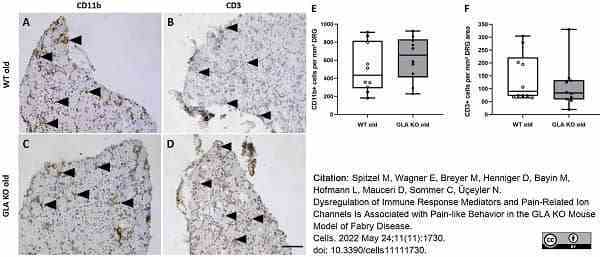
Spitzel, M. et al. (2022) Dysregulation of Immune Response Mediators and Pain-Related Ion Channels Is Associated with Pain-like Behavior in the GLA KO Mouse Model of Fabry Disease.
Cells. 11 (11): 1730. -
Ji, N. et al. (2022) VSIG4 Attenuates NLRP3 and Ameliorates Neuroinflammation via JAK2-STAT3-A20 Pathway after Intracerebral Hemorrhage in Mice.
Neurotox Res. 40 (1): 78-88. -
Shanaki-Bavarsad, M. et al. (2022) Astrocyte-targeted Overproduction of IL-10 Reduces Neurodegeneration after TBI.
Exp Neurobiol. 31 (3): 173-95. -
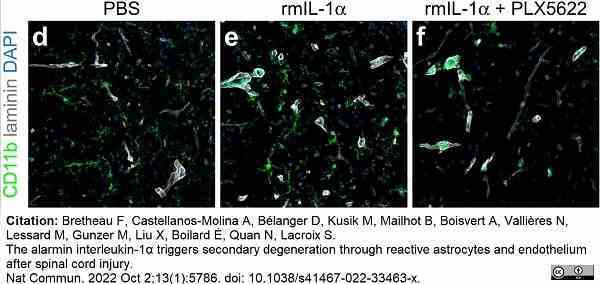
Bretheau, F. et al. (2022) The alarmin interleukin-1α triggers secondary degeneration through reactive astrocytes and endothelium after spinal cord injury.
Nat Commun. 13 (1): 5786. -
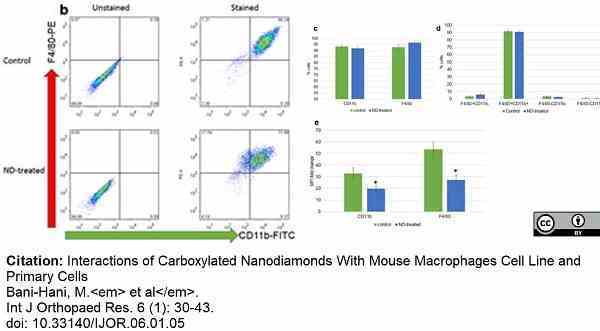
Bani-Hani, M. et al. (2023) Interactions of Carboxylated Nanodiamonds With Mouse Macrophages Cell Line and Primary Cells
Int J Orthopaed Res. 6 (1): 30-43. -
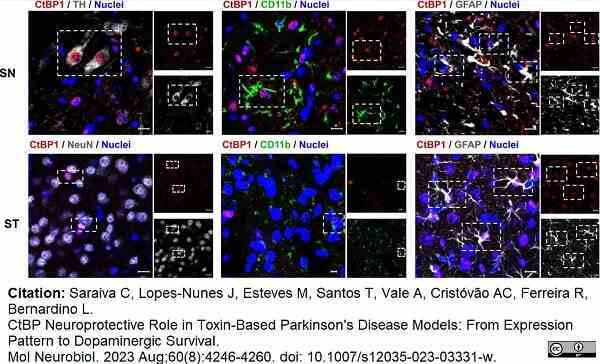
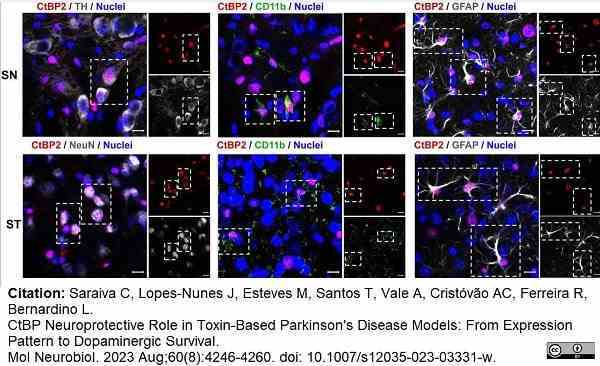
Saraiva, C. et al. (2023) CtBP Neuroprotective Role in Toxin-Based Parkinson's Disease Models: From Expression Pattern to Dopaminergic Survival.
Mol Neurobiol. 60 (8): 4246-60. -
Nelke, C. et al. (2023) K(2P)2.1 is a regulator of inflammatory cell responses in idiopathic inflammatory myopathies.
J Autoimmun. 142: 103136. -
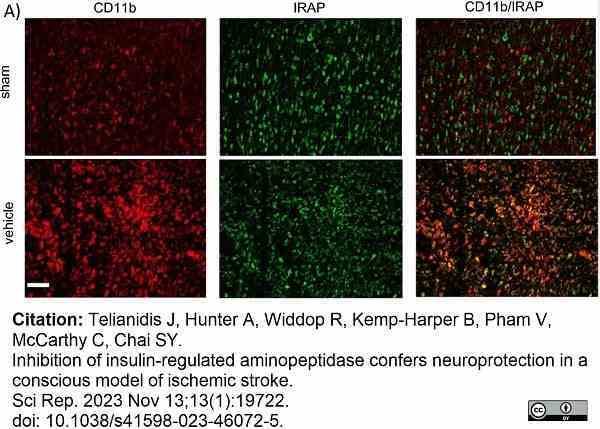
Telianidis, J. et al. (2023) Inhibition of insulin-regulated aminopeptidase confers neuroprotection in a conscious model of ischemic stroke.
Sci Rep. 13 (1): 19722. -
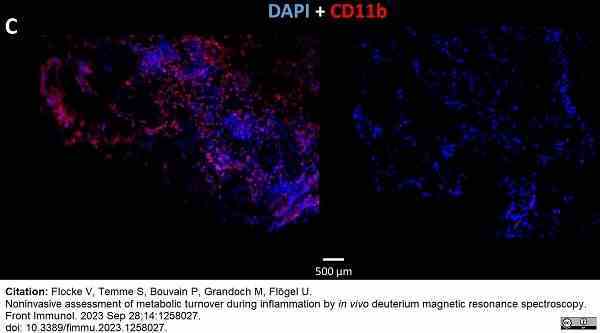
Flocke, V. et al. (2023) Noninvasive assessment of metabolic turnover during inflammation by in vivo deuterium magnetic resonance spectroscopy.
Front Immunol. 14: 1258027. -
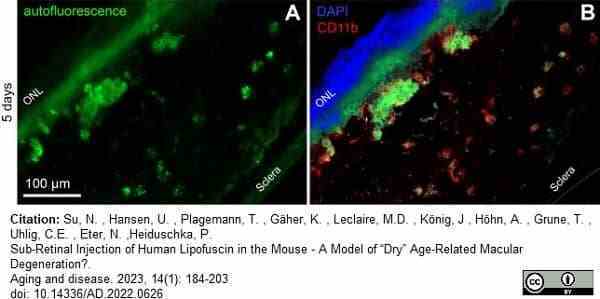
Heiduschka, P. et al. (2023) Sub-Retinal Injection of Human Lipofuscin in the Mouse - A Model of “Dry” Age-Related Macular Degeneration?
Aging and disease. 14 (1): 184. -
Lim, J. et al. (2018) Characterizing the Mechanisms of Nonopsonic Uptake of Cryptococci by Macrophages.
J Immunol. 200 (10): 3539-46. -
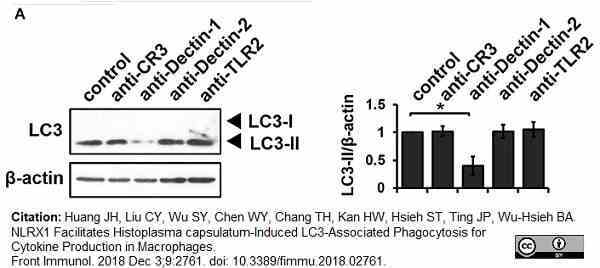
Huang, J.H. et al. (2018) NLRX1 Facilitates Histoplasma capsulatum-Induced LC3-Associated Phagocytosis for Cytokine Production in Macrophages.
Front Immunol. 9: 2761. -
Frey, O. et al. (2018) Induction of chronic destructive arthritis in SCID mice by arthritogenic fibroblast-like synoviocytes derived from mice with antigen-induced arthritis.
Arthritis Res Ther. 20 (1): 261. -
Friker, L.L. et al. (2020) β-Amyloid Clustering around ASC Fibrils Boosts Its Toxicity in Microglia.
Cell Rep. 30 (11): 3743-54.e6. -
Hickman, S.E. et al. (2019) Heterozygous CX3CR1 Deficiency in Microglia Restores Neuronal β-Amyloid Clearance Pathways and Slows Progression of Alzheimer's Like-Disease in PS1-APP Mice.
Front Immunol. 10: 2780. -
Kim, D.H. et al. (2018) Effect of growth differentiation factor-15 secreted by human umbilical cord blood-derived mesenchymal stem cells on amyloid beta levels in in vitro and in vivo models of Alzheimer's disease.
Biochem Biophys Res Commun. 504 (4): 933-40. -
Maezawa, I. et al. (2018) Kv1.3 inhibition as a potential microglia-targeted therapy for Alzheimer's disease: preclinical proof of concept.
Brain. 141 (2): 596-612. -
Price, B.R. et al. (2020) Therapeutic Trem2 activation ameliorates amyloid-beta deposition and improves cognition in the 5XFAD model of amyloid deposition.
J Neuroinflammation. 17 (1): 238. -
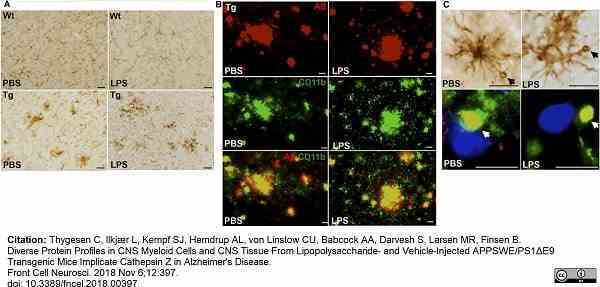
Thygesen, C. et al. (2018) Diverse Protein Profiles in CNS Myeloid Cells and CNS Tissue From Lipopolysaccharide- and Vehicle-Injected APPSWE/PS1ΔE9 Transgenic Mice Implicate Cathepsin Z in Alzheimer's Disease.
Front Cell Neurosci. 12: 397. -
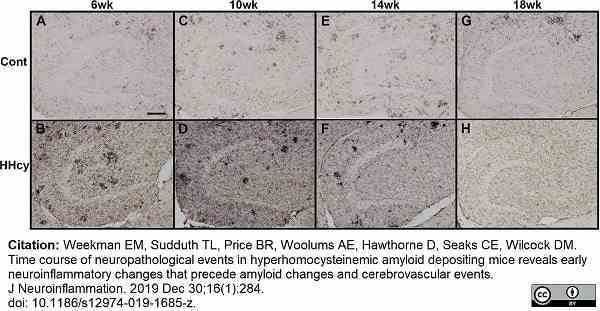
Weekman, E.M. et al. (2019) Time course of neuropathological events in hyperhomocysteinemic amyloid depositing mice reveals early neuroinflammatory changes that precede amyloid changes and cerebrovascular events.
J Neuroinflammation. 16 (1): 284. -
Thei, L. et al. (2018) Extracellular signal-regulated kinase 2 has duality in function between neuronal and astrocyte expression following neonatal hypoxic-ischaemic cerebral injury.
J Physiol. 596 (23): 6043-62. -
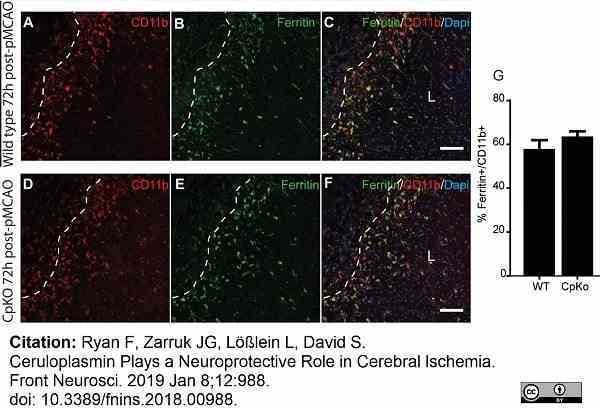
Ryan, F. et al. (2018) Ceruloplasmin Plays a Neuroprotective Role in Cerebral Ischemia.
Front Neurosci. 12: 988. -
Rizzi, C. et al. (2018) NGF steers microglia toward a neuroprotective phenotype.
Glia. 66 (7): 1395-416. -
Haight, E.S. et al. (2020) Of mice, microglia, and (wo)men: a case series and mechanistic investigation of hydroxychloroquine for complex regional pain syndrome.
Pain Rep. 5 (5): e841.
- Synonyms
- Integrin Alpha M Chain
- MAC-1
- RRID
- AB_321292
- UniProt
- P05555
- Entrez Gene
- Itgam
- GO Terms
- GO:0001846 opsonin binding
- GO:0004872 receptor activity
- GO:0008305 integrin complex
- GO:0005634 nucleus
- GO:0007159 leukocyte cell-cell adhesion
- GO:0007229 integrin-mediated signaling pathway
- GO:0008201 heparin binding
- GO:0009897 external side of plasma membrane
- GO:0030593 neutrophil chemotaxis
- View More GO Terms
- GO:0043395 heparan sulfate proteoglycan binding
- GO:0045123 cellular extravasation
- GO:0050798 activated T cell proliferation
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Mouse ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up