ER-TR7 antibody | ER-TR7



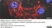
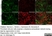
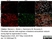
Rat anti Mouse ER-TR7
- Product Type
- Monoclonal Antibody
- Clone
- ER-TR7
- Isotype
- IgG2a
- Specificity
- ER-TR7
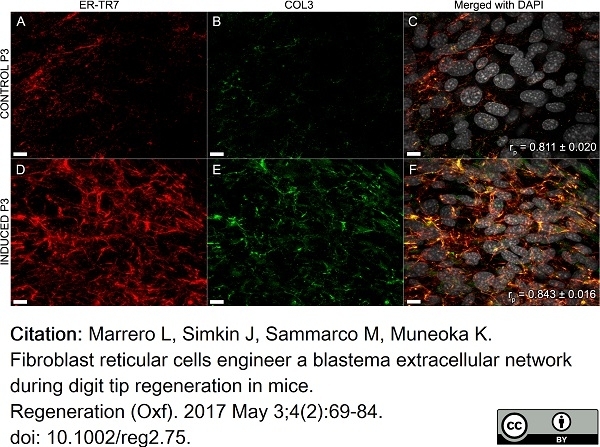
| Rat anti Mouse ER-TR7 antibody, clone ER-TR7 recognizes ER-TR7, an antigen located in the cytoplasm of reticular fibroblasts, a component of the extracellular matrix of lymphoid and non-lymphoid organs. Rat anti Mouse ER-TR7 antibody, clone ER-TR7 recognizes collagen type VI (Schiavinato et al. 2021) and has been used to stain the microanatomy of various organs and also stains subendothelial deposits in atherosclerotic plaques. |
- Target Species
- Mouse
- Product Form
- Purified IgG - liquid
- Preparation
- Purified IgG prepared by affinity chromatography on Protein G from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- 0.09% sodium azide (NaN3)
- Carrier Free
- Yes
- Immunogen
- Isolated C3H thymic stromal cells.
- Approx. Protein Concentrations
- IgG concentration 1.0 mg/ml
- Fusion Partners
- Cells from immunized rats were fused with cells of the mouse P3-X63-Ag8.563 myeloma cell line.
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Flow Cytometry 1 | |||
| Immunofluorescence | |||
| Immunohistology - Frozen | 1/50 | 1/100 |
- 1 Membrane permeabilization is required for this application. The use of Leucoperm (Product Code BUF09) is recommended for this purpose.
- Flow Cytometry
- Use 10μl of the suggested working dilution to label 1x106 cells in 100μl
- Histology Positive Control Tissue
- Mouse spleen
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Rat IgG2a Negative Control | MCA1212 | E F | 1 ml |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Rat IgG2a Negative Control | ||||||
Source Reference
-
VanVliet, E. et al. (1984) Monoclonal antibodies to stromal cell types of the mouse thymus.
Eur J Immunol. 14 (6): 524-9.
References for ER-TR7 antibody
-
Van Vliet, E. et al. (1986) Reticular fibroblasts in peripheral lymphoid organs identified by a monoclonal antibody.
J Histochem Cytochem. 34 (7): 883-90. -
Kalled, S.L. et al. (1998) Anti-CD40 ligand antibody treatment of SNF1 mice with established nephritis: preservation of kidney function.
J Immunol. 160: 2158-65. -
Katakai, T. et al. (2003) Th1-biased tertiary lymphoid tissue supported by CXC chemokine ligand 13-producing stromal network in chronic lesions of autoimmune gastritis.
J Immunol. 171: 4359-68. -
Fujii, N. et al. (2006) Targeting of interstitial cells using a simple gene-transfer strategy.
Nephrol Dial Transplant. 21: 2745-53. -
Mueller, S.N. et al. (2007) Viral targeting of fibroblastic reticular cells contributes to immunosuppression and persistence during chronic infection.
Proc Natl Acad Sci U S A. 104:15430-5. -
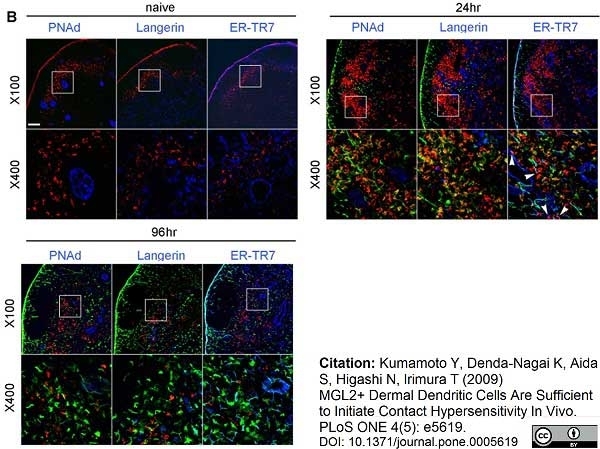
Kumamoto, Y. et al. (2009) MGL2 Dermal dendritic cells are sufficient to initiate contact hypersensitivity in vivo.
PLoS One. 4: e5619. -
Tumanov, A.V. et al. (2010) Cellular source and molecular form of TNF specify its distinct functions in organization of secondary lymphoid organs.
Blood. 116: 3456-64. -
Köhler, C.N. (2010) The actin-binding protein caldesmon is in spleen and lymph nodes predominately expressed by smooth-muscle cells, reticular cells, and follicular dendritic cells.
J Histochem Cytochem. 58 (2): 183-93.
View The Latest Product References
-
Umemoto, E. et al. (2012) Constitutive plasmacytoid dendritic cell migration to the splenic white pulp is cooperatively regulated by CCR7- and CXCR4-mediated signaling.
J Immunol. 189 (1): 191-9. -
Burrell, B.E. et al. (2015) Lymph Node Stromal Fiber ER-TR7 Modulates CD4+ T Cell Lymph Node Trafficking and Transplant Tolerance.
Transplantation. 99 (6): 1119-25. -
Bennett, K.M. et al. (2016) Induction of Colonic M Cells during Intestinal Inflammation.
Am J Pathol. 186 (5): 1166-79. -
Watanabe, R. et al. (2016) Formation of fibroblastic reticular network in the brain after infection with neurovirulent murine coronavirus.
Neuropathology. 36 (6): 513-26. -
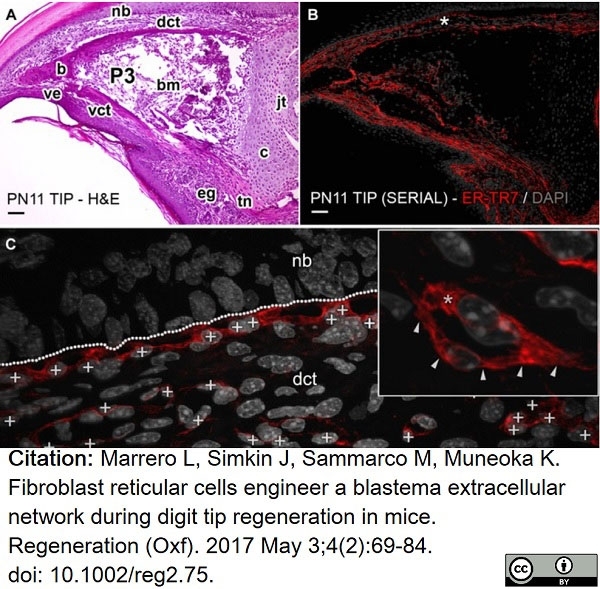
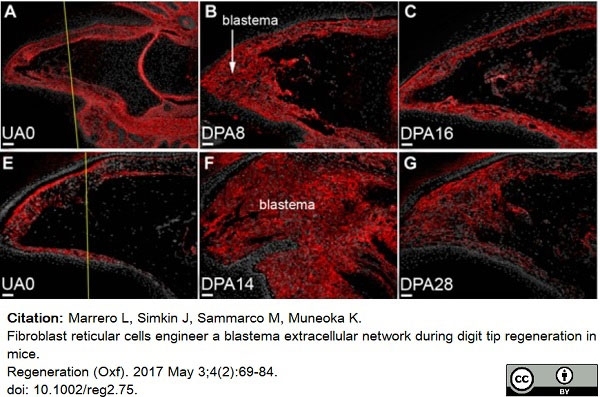
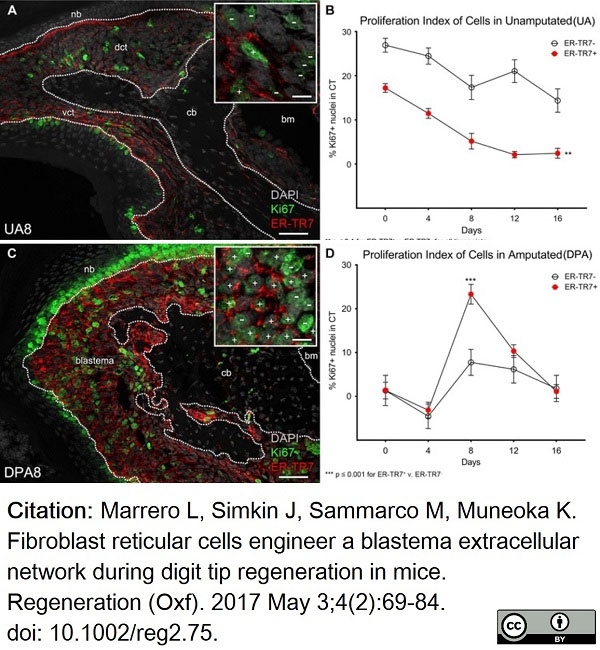
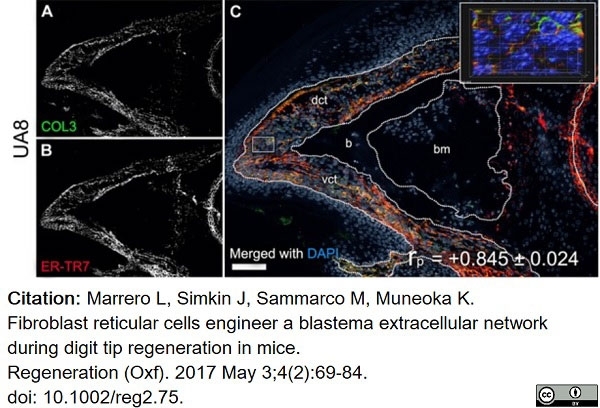
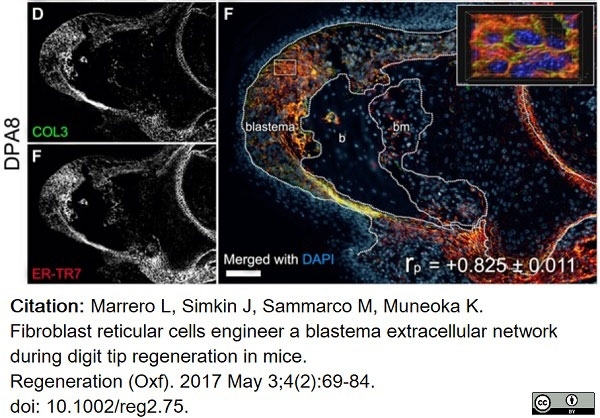
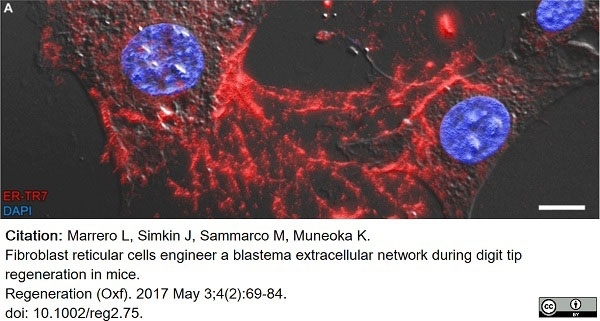
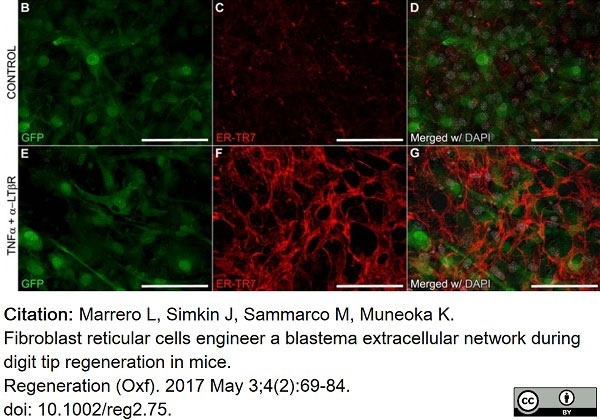
Marrero, L. et al. (2017) Fibroblast reticular cells engineer a blastema extracellular network during digit tip regeneration in mice.
Regeneration (Oxf). 4 (2): 69-84. -
Hwang, I.Y. et al. (2017) Normal Thymocyte Egress, T Cell Trafficking, and CD4+ T Cell Homeostasis Require Interactions between RGS Proteins and Gαi2.
J Immunol. 198 (7): 2721-2734. -
Dawson, L.A. et al. (2021) Proximal digit tip amputation initiates simultaneous blastema and transient fibrosis formation and results in partial regeneration.
Wound Repair Regen. 29 (1): 196-205.
Further Reading
-
Lokmic, Z. et al. (2008) The extracellular matrix of the spleen as a potential organizer of immune cell compartments.
Semin Immunol. 20: 4-13. -
Schiavinato, A. et al. (2021) Collagen type VI is the antigen recognized by the ER-TR7 antibody.
Eur J Immunol. 51 (9): 2345-7.
- Synonyms
- Collagen alpha-1(VI) Chain
- RRID
- AB_915429
- UniProt
- Q04857
- Entrez Gene
- Col6a1
- GO Terms
- GO:0007155 cell adhesion
- GO:0005578 proteinaceous extracellular matrix
- GO:0042383 sarcolemma
MCA2402
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Mouse ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up