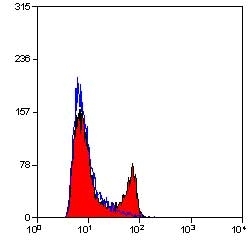
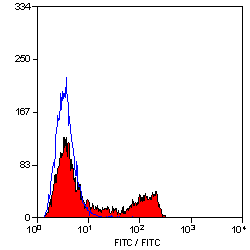
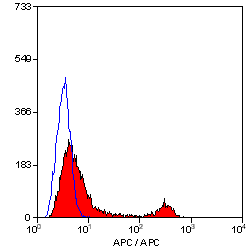
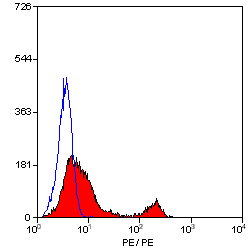
CD4 antibody | YTS191.1
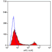
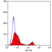
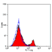




Rat anti Mouse CD4
- Product Type
- Monoclonal Antibody
- Clone
- YTS191.1
- Isotype
- IgG2b
- Specificity
- CD4
| Rat anti Mouse CD4 antibody, clone YTS191.1 recognizes the murine CD4 cell surface antigen, expressed by a subset of T lymphocytes. Rat anti Mouse CD4 antibody, clone YTS191.1 exhibits depleting activity when used in vivo ( |
- Target Species
- Mouse
- Product Form
- Purified IgG - liquid
- Preparation
- Purified IgG prepared by affinity chromatography on Protein G from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- 0.09% sodium azide (NaN3)
- Carrier Free
- Yes
- Approx. Protein Concentrations
- MCA1767GA: IgG concentration 1 mg/ml
- MCA1767T, MCA1767: IgG concentration 1.0 mg/ml
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Flow Cytometry | 1/50 | 1/200 Pack Size: 0.1 mg 1/100 Pack Size: 25 µg, 0.25 mg |
|
| Immunohistology - Frozen 1 | |||
| Immunohistology - Paraffin |
- 1The epitope recognised by this antibody is reported to be sensitive to formaldehyde fixation and tissue processing. Bio-Rad recommends the use of acetone fixation for frozen sections.
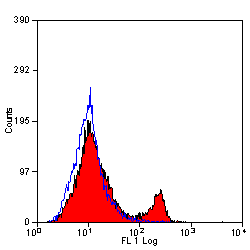
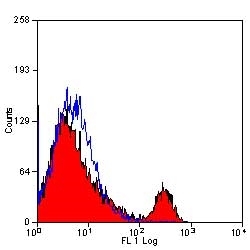
- Flow Cytometry
- Use 10μl of the suggested working dilution to label 106 cells in 100μl
References for CD4 antibody
-
Cobbold, S.P. et al. (1990) The induction of skin graft tolerance in major histocompatibility complex-mismatched or primed recipients: primed T cells can be tolerized in the periphery with anti-CD4 and anti-CD8 antibodies.
Eur J Immunol. 20 (12): 2747-55. -
Bemelman, F. et al. (1998) Bone marrow transplantation induces either clonal deletion or infectious tolerance depending on the dose.
J Immunol. 160 (6): 2645-8. -
Higgins, L.M. et al. (1999) Regulation of T cell activation in vitro and in vivo by targeting the OX40-OX40 ligand interaction: amelioration of ongoing inflammatory bowel disease with an OX40-IgG fusion protein, but not with an OX40 ligand-IgG fusion protein.
J Immunol. 162 (1): 486-93. -
Croxford, J.L. et al. (2001) Different therapeutic outcomes in experimental allergic encephalomyelitis dependent upon the mode of delivery of IL-10: a comparison of the effects of protein, adenoviral or retroviral IL-10 delivery into the central nervous system.
J Immunol. 166: 4124-30. -
Eller, K. et al. (2011) IL-9 production by regulatory T cells recruits mast cells that are essential for regulatory T cell-induced immune suppression.
J Immunol. 186: 83-91. -
Gaupp, S. et al. (2008) Amelioration of experimental autoimmune encephalomyelitis in IL-4Ralpha-/- mice implicates compensatory up-regulation of Th2-type cytokines.
Am J Pathol. 173: 119-29. -
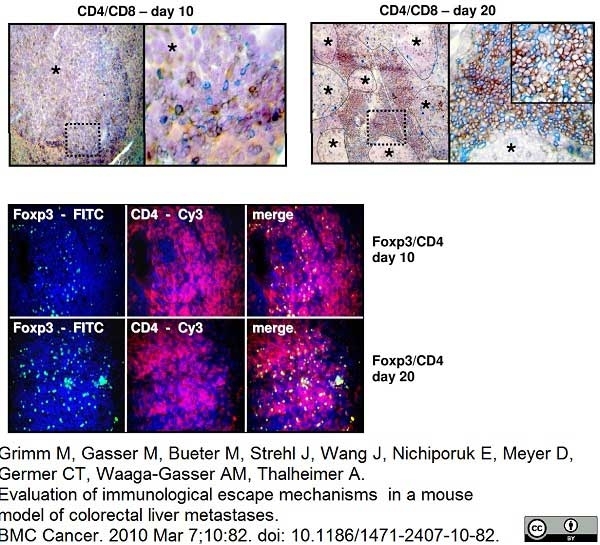
Grimm, M.et al. (2010) Evaluation of immunological escape mechanisms in a mouse model of colorectal liver metastases.
BMC Cancer. 10: 82. -
Jégou, J.F. et al. (2007) C3d Binding to the Myelin Oligodendrocyte Glycoprotein Results in an Exacerbated Experimental Autoimmune Encephalomyelitis
J Immunol. 178: 3323-31.
View The Latest Product References
-
Huber, J.M. et al. (2009) The proteasome inhibitor bortezomib aggravates renal ischemia-reperfusion injury.
Am J Physiol Renal Physiol. 297: F451-60. -
Wolf, D. et al. (2005) CD4+CD25+ regulatory T cells inhibit experimental anti-glomerular basement membrane glomerulonephritis in mice.
J Am Soc Nephrol. 16: 1360-70. -
Abdulreda, M.H. et al. (2011) High-resolution, noninvasive longitudinal live imaging of immune responses.
Proc Natl Acad Sci U S A. 108: 12863-8. -
Nakashima, H. et al. (2011) A Novel Combination Immunotherapy for Cancer by IL-13Rα2-Targeted DNA Vaccine and Immunotoxin in Murine Tumor Models.
J Immunol. 187: 4935-46. -
Scotland, R.S. et al. (2011) Sex-differences in resident immune cell phenotype underlies more efficient acute inflammatory responses in female mice.
Blood. 118: 5918-27. -
Zitt, E. et al. (2011) The selective mineralocorticoid receptor antagonist eplerenone is protective in mild anti-GBM glomeru-lonephritis.
Int J Clin Exp Pathol. 4:606-15. -
Nelvagal, H.R. et al. (2020) Comparative proteomic profiling reveals mechanisms for early spinal cord vulnerability in CLN1 disease.
Sci Rep. 10 (1): 15157. -
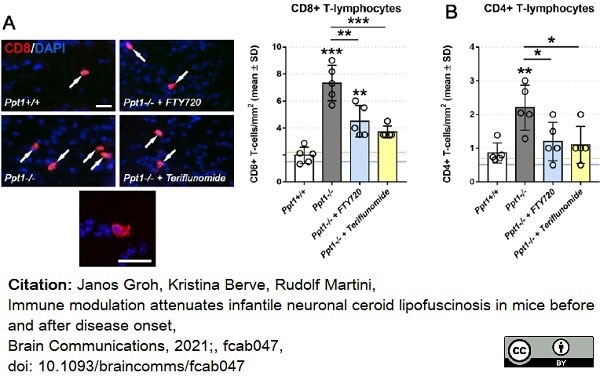
Groh, J. et al. (2021) Immune modulation attenuates infantile neuronal ceroid lipofuscinosis in mice before and after disease onset
Brain Communications. fcab047. -
Karikari, A.A. et al. (2022) Neurodegeneration by α-synuclein-specific T cells in AAV-A53T-α-synuclein Parkinson's disease mice.
Brain Behav Immun. 101: 194-210. -
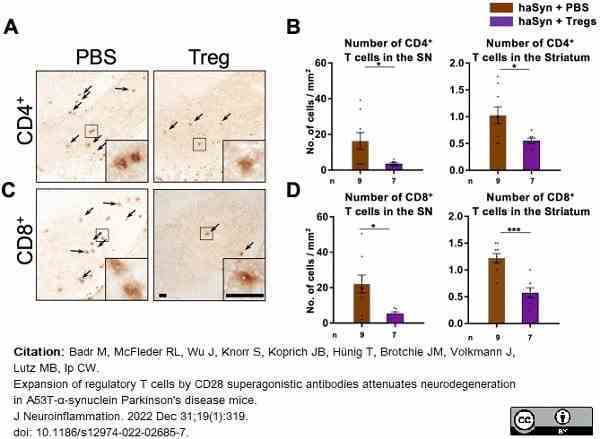
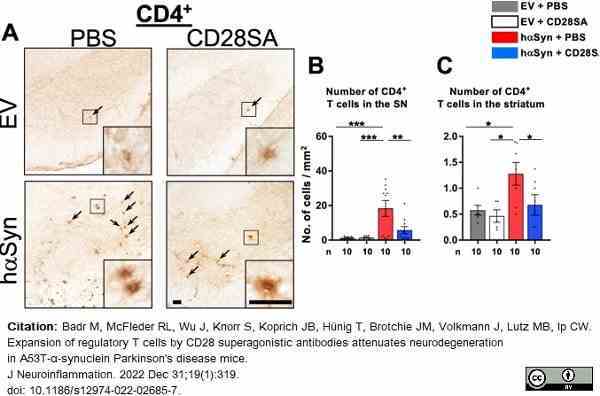
Badr, M. et al. (2022) Expansion of regulatory T cells by CD28 superagonistic antibodies attenuates neurodegeneration in A53T-α-synuclein Parkinson's disease mice.
J Neuroinflammation. 19 (1): 319. -
McFleder, R.L. et al. (2023) Brain-to-gut trafficking of alpha-synuclein by CD11c(+) cells in a mouse model of Parkinson's disease.
Nat Commun. 14 (1): 7529. -
Griffiths, M.R. et al. (2018) CD93 regulates central nervous system inflammation in two mouse models of autoimmune encephalomyelitis.
Immunology. 155 (3): 346-55. -
Mahadevan, K.K. et al. (2023) Antigen-presenting type-I conventional dendritic cells facilitate curative checkpoint blockade immunotherapy in pancreatic cancer.
bioRχiv. Mar 06 [Epub ahead of print]. -
Aringer, I. et al. (2021) Agonism of Prostaglandin E2 Receptor 4 Ameliorates Tubulointerstitial Injury in Nephrotoxic Serum Nephritis in Mice.
J Clin Med. 10 (4): 832.
- Synonyms
- L3T4 Antigen
- LY-4
- RRID
- AB_322769
- UniProt
- P06332
- Entrez Gene
- Cd4
- GO Terms
- GO:0001816 cytokine production
- GO:0007155 cell adhesion
- GO:0016021 integral to membrane
- GO:0005788 endoplasmic reticulum lumen
- GO:0005789 endoplasmic reticulum membrane
- GO:0006955 immune response
- GO:0007166 cell surface receptor linked signaling pathway
- GO:0009897 external side of plasma membrane
- GO:0045058 T cell selection
- View More GO Terms
- GO:0045121 membrane raft
- GO:0050731 positive regulation of peptidyl-tyrosine phosphorylation
- GO:0050850 positive regulation of calcium-mediated signaling
- GO:0050870 positive regulation of T cell activation
- GO:0051789 response to protein stimulus
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Mouse ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up