CD52 antibody | YTH34.5





Rat anti Human CD52
- Product Type
- Monoclonal Antibody
- Clone
- YTH34.5
- Isotype
- IgG2b
- Specificity
- CD52
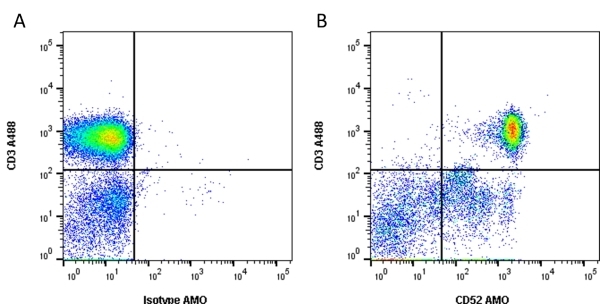
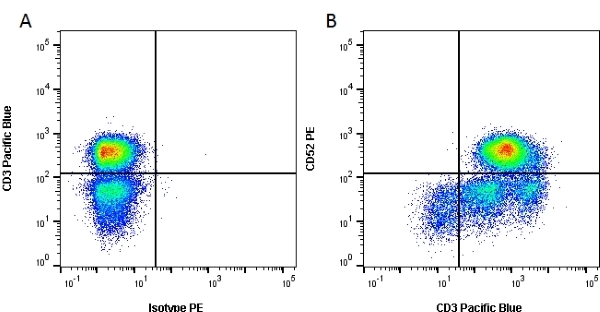
| Rat anti Human CD52 antibody, clone YTH34.5 recognizes the human CD52 antigen, also known as CAMPATH-1. The CD52 antigen is a remarkably small but heavily glycosylated peptide attached to the cell surface membrane via a GPI link (Xia et al. 1991). The apparent molecular mass of the native antigen on SDS-PAGE is 25-29 kDa, considerably reduced following N-glycanase treatment (Rowan et al. 1998). CD52 is expressed at high density by lymphocytes, monocytes, eosinophils, thymocytes and macrophages. It is expressed by most lymphoid derived malignancies, although expression on myeloma cells is variable. Humanized versions of CAMPATH-1 specific antibodies are currently in clinical trials for the treatment of a range of lymphoid malignancies (Dearden et al. 2002; Pettitt et al. 2012). |
- Target Species
- Human
- Species Cross-Reactivity
-
Target Species Cross Reactivity Rhesus Monkey - N.B. Antibody reactivity and working conditions may vary between species.
- Product Form
- Purified IgG - liquid
- Preparation
- Purified IgG prepared by affinity chromatography on Protein A from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- <0.1% sodium azide (NaN3)
- Immunogen
- Human lymphocytes
- Approx. Protein Concentrations
- IgG concentration 1.0 mg/ml
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Cytotoxic Assays | 50ug/ml (Use human serum as complement source) | ||
| ELISA | |||
| Flow Cytometry | 1/20 | ||
| Immunohistology - Frozen | 1/20 | ||
| Immunohistology - Paraffin | 1/20 | 1/20 Pack Size: 20 µg 1/40 Pack Size: 0.2 mg |
|
| Immunohistology - Resin | |||
| Immunoprecipitation | |||
| Western Blotting |
- Flow Cytometry
- use 10μl of the suggested working dilution to label 1 x 106 cells in 100μl
- Histology Positive Control Tissue
- Tonsil
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Rat IgG2b Negative Control | MCA6006GA | F | 0.1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Rat IgG2b Negative Control | ||||||
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Human anti Alemtuzumab | HCA174 | E | 0.1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Human anti Alemtuzumab | ||||||
| Human anti Alemtuzumab | HCA175 | E | 0.1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Human anti Alemtuzumab | ||||||
References for CD52 antibody
-
Klangsinsirikul, P. et al. (2002) Campath-1G causes rapid depletion of circulating host dendritic cells (DCs) before allogeneic transplantation but does not delay donor DC reconstitution.
Blood. 99: 2586-91. -
Ratzinger, G. et al. (2003) Differential CD52 expression by distinct myeloid dendritic cell subsets: implications for alemtuzumab activity at the level of antigen presentation in allogeneic graft-host interactions in transplantation.
Blood. 101: 1422-9. -
Zand, M.S. et al. (2005) A renewable source of donor cells for repetitive monitoring of T- and B-cell alloreactivity.
Am J Transplant. 5: 76-86. -
Westermann, J. et al. (2005) CD52 is not a promising immunotherapy target for most patients with multiple myeloma.
Int J Hematol. 82 (3): 248-50. -
Gopcsa, L. et al. (2005) Extensive flow cytometric characterization of plasmacytoid dendritic cell leukemia cells.
Eur J Haematol. 75: 346-51. -
Rodig SJ et al. (2006) Heterogeneous CD52 expression among hematologic neoplasms: implications for the use of alemtuzumab (CAMPATH-1H).
Clin Cancer Res. 12 (23): 7174-9. -
Golay, J. et al. (2006) The sensitivity of acute lymphoblastic leukemia cells carrying the t(12;21) translocation to campath-1H-mediated cell lysis.
Haematologica. 91: 322-30. -
Miles, R.R. et al. (2007) Immunophenotypic identification of possible therapeutic targets in paediatric non-Hodgkin lymphomas: a children's oncology group report.
Br J Haematol. 138: 506-12.
View The Latest Product References
-
Chang, S.T. et al. (2007) CD52 expression in non-mycotic T- and NK/T-cell lymphomas.
Leuk Lymphoma. 48: 117-21. -
Piccaluga, P.P. et al. (2007) Expression of CD52 in peripheral T-cell lymphoma.
Haematologica. 92: 566-7. -
Reimer, P. et al. (2009) Autologous stem-cell transplantation as first-line therapy in peripheral T-cell lymphomas: results of a prospective multicenter study.
J Clin Oncol. 27: 106-13. -
Hu, Y. et al. (2009) Investigation of the mechanism of action of alemtuzumab in a human CD52 transgenic mouse model.
Immunology. 128: 260-70. -
Rizzo, K. et al. (2009) Novel CD19 expression in a peripheral T cell lymphoma: A flow cytometry case report with morphologic correlation.
Cytometry B Clin Cytom. 76: 142-9. -
Haniffa, M. et al. (2009) Differential rates of replacement of human dermal dendritic cells and macrophages during hematopoietic stem cell transplantation.
J Exp Med. 206: 371-85. -
Bisig, B. et al. (2013) CD30-positive peripheral T-cell lymphomas share molecular and phenotypic features.
Haematologica. 98 (8): 1250-8. -
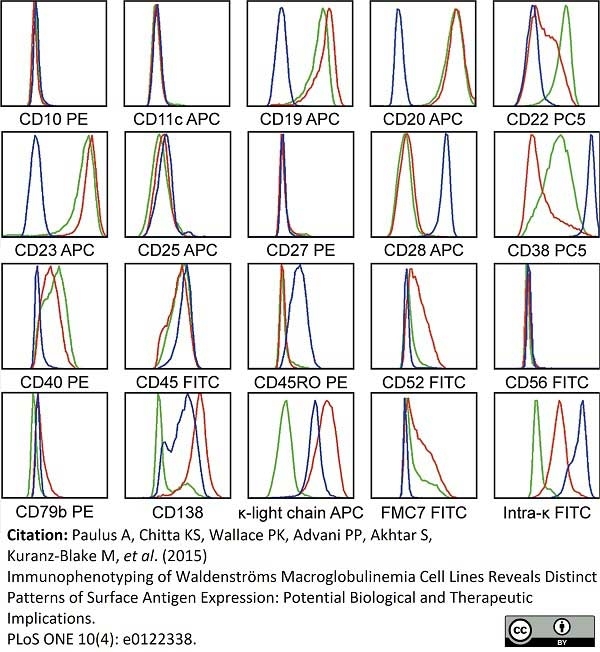
Paulus, A. et al. (2015) Immunophenotyping of Waldenströms macroglobulinemia cell lines reveals distinct patterns of surface antigen expression: potential biological and therapeutic implications.
PLoS One. 10 (4): e0122338. -
Hotta, R. et al. (2016) CD52-Negative NK Cells Are Abundant in the Liver and Less Susceptible to Alemtuzumab Treatment.
PLoS One. 11 (8): e0161618. -
Buckstein, R. et al. (2016) Alemtuzumab and CHOP Chemotherapy for the Treatment of Aggressive Histology Peripheral T Cell Lymphomas: A Multi-Center Phase I Study.
Clin Lymphoma Myeloma Leuk. 16 (1): 18-28.e4. -
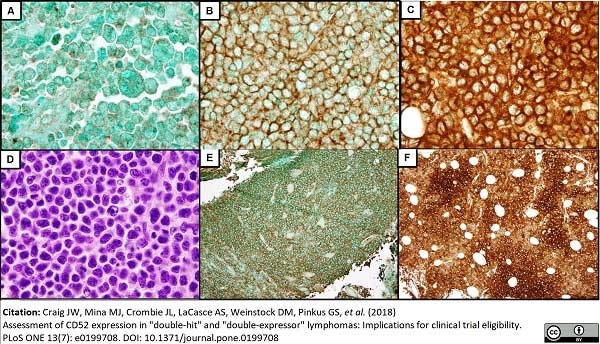
Craig, J.W. et al. (2018) Assessment of CD52 expression in "double-hit" and "double-expressor" lymphomas: Implications for clinical trial eligibility.
PLoS One. 13 (7): e0199708. -
Suwandi, J.S. et al. (2020) Multidimensional analyses of proinsulin peptide-specific regulatory T cells induced by tolerogenic dendritic cells.
J Autoimmun. 107: 102361.
Further Reading
-
Salisbury JR et al. (1994) Immunohistochemical analysis of CDw52 antigen expression in non-Hodgkin's lymphomas.
J Clin Pathol. 47 (4): 313-7. -
Hale G et al. (1998) Improving the outcome of bone marrow transplantation by using CD52 monoclonal antibodies to prevent graft-versus-host disease and graft rejection.
Blood. 92 (12): 4581-90.
- Synonyms
- CAMPATH-1
- RRID
- AB_321470
- UniProt
- P31358
- Entrez Gene
- CD52
- GO Terms
- GO:0005887 integral to plasma membrane
- GO:0005624 membrane fraction
- GO:0007204 elevation of cytosolic calcium ion concentration
- GO:0031225 anchored to membrane
- GO:0045730 respiratory burst
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Human ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up