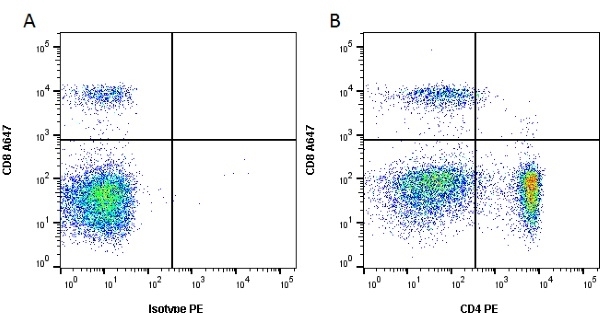
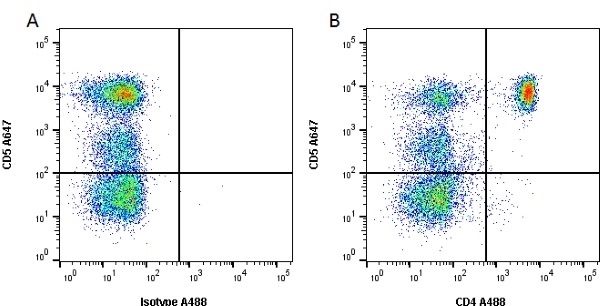
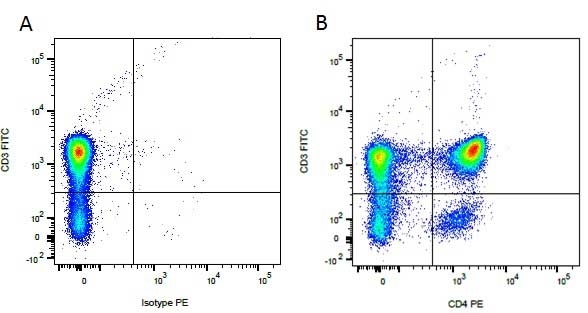
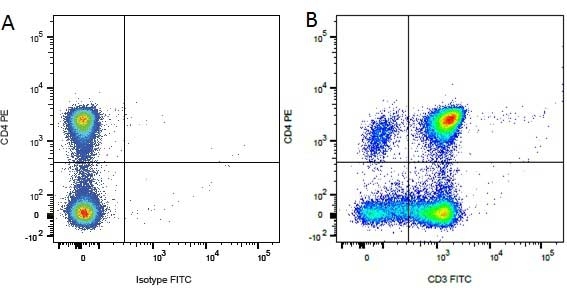
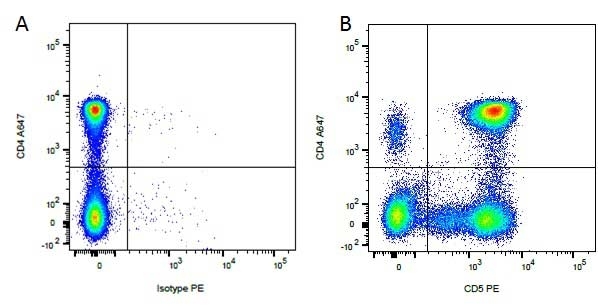
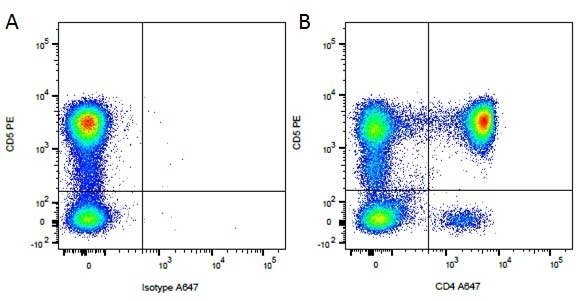
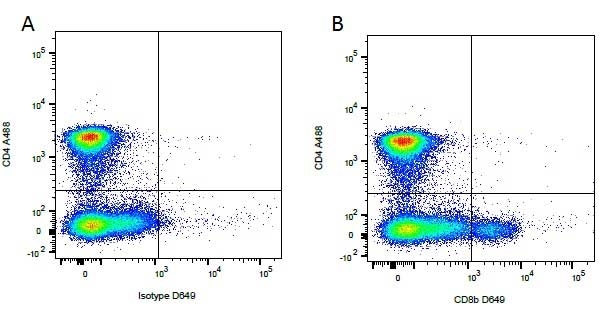
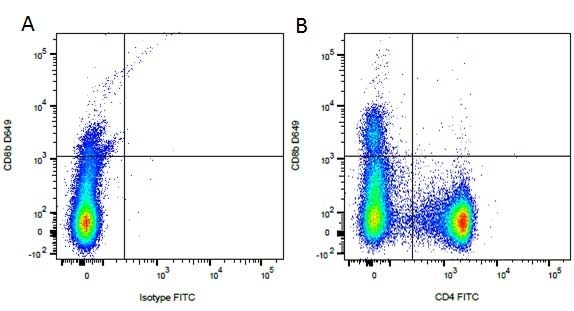
CD4 antibody | YKIX302.9














Rat anti Dog CD4
- Product Type
- Monoclonal Antibody
- Clone
- YKIX302.9
- Isotype
- IgG2a
- Specificity
- CD4
| Rat anti Dog CD4 antibody, clone YKIX302.9, is a monoclonal antibody specific for the canine CD4 cell surface antigen. Clone YKIX302.9 was clustered at the first Canine Leukocyte Antigen Workshop (CLAW) [Cobbold et al. 1992] along with clone CA13.1E4. Rat anti Dog CD4 antibody, clone YKIX302.9 partially depletes circulating T lymphocytes when administered in vivo, but alone is not sufficient to prolong allograft survival in a canine transplant model (Watson et al. 1993). Uniquely amongst mammals, canine CD4 is expressed by neutrophils as well as by lymphocyte subsets (Moore et al. 1992). |
- Target Species
- Dog
- Product Form
- Purified IgG - liquid
- Preparation
- Purified IgG prepared by affinity chromatography on Protein G from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- 0.09% sodium azide (NaN3)
- Carrier Free
- Yes
- Immunogen
- Canine concanavilin A activated T cell blasts.
- Approx. Protein Concentrations
- IgG concentration 1.0 mg/ml
- Fusion Partners
- Spleen cells from immunized DA rats were fused with cells of the Y3/Ag1.2.3 rat myeloma cell line.
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Flow Cytometry | 1/50 | 1/100 | |
| Immunohistology - Frozen |
- Flow Cytometry
- Use 10μl of the suggested working dilution to label 106 cells or 100μl whole blood
Source Reference
-
Cobbold, S. & Metcalfe, S. (1994) Monoclonal antibodies that define canine homologues of human CD antigens: summary of the First International Canine Leukocyte Antigen Workshop (CLAW).
Tissue Antigens. 43 (3): 137-54.
References for CD4 antibody
-
Watson, C.J. et al. (1993) CD4 and CD8 monoclonal antibody therapy: strategies to prolong renal allograft survival in the dog.
Br J Surg. 80 (11): 1389-92. -
Gorman, S.D. et al. (1994) Isolation and expression of cDNA encoding the canine CD4 and CD8 alpha antigens.
Tissue Antigens. 43 (3): 184-8. -
Out, T.A. et al. (2002) Local T-cell activation after segmental allergen challenge in the lungs of allergic dogs.
Immunology. 105: 499-508. -
Benyacoub, J. et al. (2003) Supplementation of food with Enterococcus faecium (SF68) stimulates immune functions in young dogs.
J Nutr. 133: 1158-62. -
Bauer. T.R. Jr. et al. (2006) Correction of the disease phenotype in canine leukocyte adhesion deficiency using ex vivo hematopoietic stem cell gene therapy.
Blood. 108: 3313-20. -
Reis, A.B. et al. (2006) Phenotypic features of circulating leucocytes as immunological markers for clinical status and bone marrow parasite density in dogs naturally infected by Leishmania chagasi.
Clin Exp Immunol. 146: 303-11. -
Miranda, S. et al. (2007) Characterization of circulating lymphocyte subpopulations in canine leishmaniasis throughout treatment with antimonials and allopurinol.
Vet Parasitol. 144 (3-4): 251-60. -
Yasuda, N. et al. (2008) CC chemokine receptor 4-positive CD4(+) lymphocytes in peripheral blood increases during maturation in healthy beagles.
J Vet Med Sci. 70 (9): 989-92.
View The Latest Product References
-
Papadogiannakis, E.I. et al. (2009) Determination of intracellular cytokines IFN-gamma and IL-4 in canine T lymphocytes by flow cytometry following whole-blood culture.
Can J Vet Res. 73 (2): 137-43. -
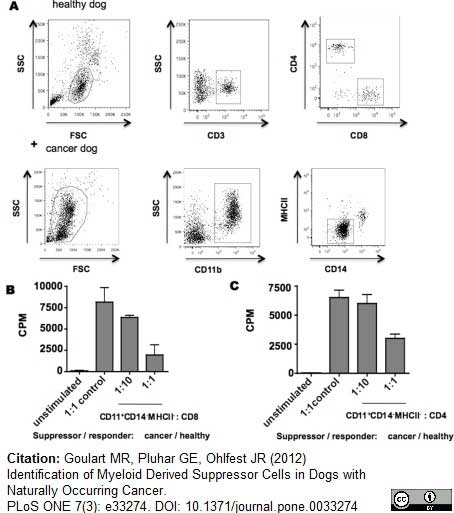
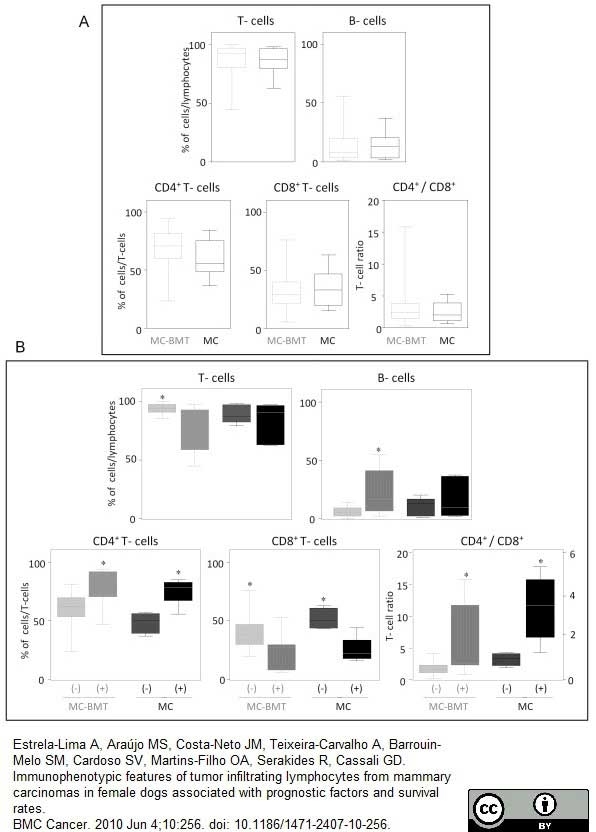
Estrela-Lima, A. et al. (2010) Immunophenotypic features of tumor infiltrating lymphocytes from mammary carcinomas in female dogs associated with prognostic factors and survival rates.
BMC Cancer. 10: 256. -
Boggiatto, P.M. et al. (2010) Immunologic indicators of clinical progression during canine Leishmania infantum infection.
Clin Vaccine Immunol. 17: 267-73. -
Tominaga, M. et al. (2010) Flow cytometric analysis of peripheral blood and tumor-infiltrating regulatory T cells in dogs with oral malignant melanoma.
J Vet Diagn Invest. 22: 438-41. -
Bund, D. et al. (2010) Canine-DCs using different serum-free methods as an approach to provide an animal-model for immunotherapeutic strategies.
Cell Immunol. 263: 88-98. -
Pinheiro, D. (2011) Phenotypic and functional characterization of a CD4(+) CD25(high) FOXP3(high) regulatory T-cell population in the dog.
Immunology. 132: 111-22. -
Araújo, M.S. et al. (2011) Immunological changes in canine peripheral blood leukocytes triggered by immunization with first or second generation vaccines against canine visceral leishmaniasis.
Vet Immunol Immunopathol. 141: 64-75. -
Mitchell, L. et al. (2012) Induction of remission results in spontaneous enhancement of anti-tumor cytotoxic T-lymphocyte activity in dogs with B cell lymphoma.
Vet Immunol Immunopathol. 145 (3-4): 597-603. -
Mitchell, L. et al. (2012) Clinical and immunomodulatory effects of toceranib combined with low-dose cyclophosphamide in dogs with cancer.
J Vet Intern Med. 26: 355-62. -
Aricò, A. et al. (2013) The role of vascular endothelial growth factor and matrix metalloproteinases in canine lymphoma: in vivo and in vitro study.
BMC Vet Res. 9: 94. -
Michael HT et al. (2013) Isolation and characterization of canine natural killer cells.
Vet Immunol Immunopathol. 155 (3): 211-7. -
Figueiredo, M.M. et al. (2014) Expression of Regulatory T Cells in Jejunum, Colon, and Cervical and Mesenteric Lymph Nodes of Dogs Naturally Infected with Leishmania infantum.
Infect Immun. 82: 3704-12. -
Aresu, L. et al. (2014) VEGF and MMP-9: biomarkers for canine lymphoma.
Vet Comp Oncol. 12: 29-36. -
Duz AL et al. (2014) The TcI and TcII Trypanosoma cruzi experimental infections induce distinct immune responses and cardiac fibrosis in dogs.
Mem Inst Oswaldo Cruz. 109 (8): 1005-13. -
Gelain, M.E. et al. (2014) CD44 in canine leukemia: analysis of mRNA and protein expression in peripheral blood.
Vet Immunol Immunopathol. 159 (1-2): 91-6. -
Yamaya, Y. & Watari, T. (2015) Increased proportions of CCR4(+) cells among peripheral blood CD4(+) cells and serum levels of allergen-specific IgE antibody in canine chronic rhinitis and bronchitis.
J Vet Med Sci. 77 (4): 421-5. -
Miller, J. et al. (2015) Humoral and Cellular Immune Response in Canine Hypothyroidism.
J Comp Pathol. 153 (1): 28-37. -
Viana, K.F. et al. (2015) Setting the proportion of CD4+ and CD8+ T-cells co-cultured with canine macrophages infected with Leishmania chagasi.
Vet Parasitol. 211 (3-4): 124-32. -
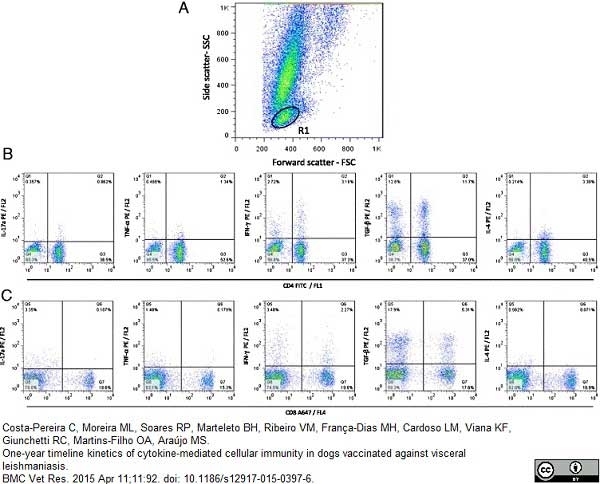
Costa-Pereira, C. et al. (2015) One-year timeline kinetics of cytokine-mediated cellular immunity in dogs vaccinated against visceral leishmaniasis.
BMC Vet Res. 11 (1): 92. -
Hauck, V. et al. (2016) Increased numbers of FoxP3-expressing CD4(+) CD25(+) regulatory T cells in peripheral blood from dogs with atopic dermatitis and its correlation with disease severity.
Vet Dermatol. 27 (1): 26-e9. -
Riondato, F. et al. (2016) Analytical and diagnostic validation of a flow cytometric strategy to quantify blood and marrow infiltration in dogs with large B-cell lymphoma.
Cytometry B Clin Cytom. 90 (6): 525-30. -
Bonnefont-Rebeix, C. et al. (2016) Characterization of a novel canine T-cell line established from a spontaneously occurring aggressive T-cell lymphoma with large granular cell morphology.
Immunobiology. 221 (1): 12-22. -
Viana, K.F. et al. (2016) Application of rapid in vitro co-culture system of macrophages and T-cell subsets to assess the immunogenicity of dogs vaccinated with live attenuated Leishmania donovani centrin deleted parasites (LdCen-/-).
Parasit Vectors. 9: 250. -
Munhoz.T.D. et al. (2016) Regulatory T cells in dogs with multicentric lymphoma: peripheral blood quantification at diagnosis and after initial stage chemotherapy.
Arq. Bras. Med. Vet. Zootec. 68 (1): 1-9. -
Tagawa, M. et al. (2016) Evaluation of Costimulatory Molecules in Peripheral Blood Lymphocytes of Canine Patients with Histiocytic Sarcoma.
PLoS One. 11 (2): e0150030. -
Schaut, R.G. et al. (2016) Regulatory IgDhi B Cells Suppress T Cell Function via IL-10 and PD-L1 during Progressive Visceral Leishmaniasis.
J Immunol. 196 (10): 4100-9. -
Schaut, R.G. et al. (2016) Recovery of antigen-specific T cell responses from dogs infected with Leishmania (L.) infantum by use of vaccine associated TLR-agonist adjuvant.
Vaccine. 34 (44): 5225-34. -
Deravi, N. et al. (2017) Specific immunotypes of canine T cell lymphoma are associated with different outcomes.
Vet Immunol Immunopathol. 191: 5-13. -
Bahamondes, F. et al. (2017) Omental adipose tissue is a more suitable source of canine Mesenchymal stem cells.
BMC Vet Res. 13 (1): 166. -
Roatt, B.M. et al. (2017) A Vaccine Therapy for Canine Visceral Leishmaniasis Promoted Significant Improvement of Clinical and Immune Status with Reduction in Parasite Burden.
Front Immunol. 8: 217. -
Anai, L.A. et al. (2017) Quantification of Treg cells in peripheral blood and lymph nodes of dogs with multicentric lymphoma
Arq Bras Med Vet Zootec. 69 (6): 1496-502. -
Pellin, M.A. et al. (2017) Safety evaluation of combination doxorubicin and toceranib phosphate (Palladia®) in tumour bearing dogs: a phase I dose-finding study.
Vet Comp Oncol. 15 (3): 919-31. -
Martins, G.C. et al. (2018) Clinical-pathological and immunological biomarkers in dogs with atopic dermatitis.
Vet Immunol Immunopathol. 205: 58-64. -
Withers, S.S. et al. (2018) Multi-color flow cytometry for evaluating age-related changes in memory lymphocyte subsets in dogs.
Dev Comp Immunol. 87: 64-74. -
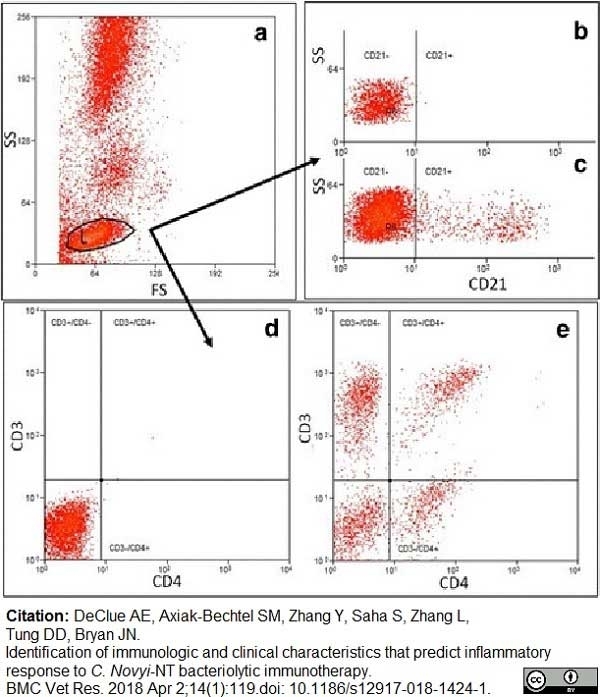
DeClue, A.E. et al. (2018) Identification of immunologic and clinical characteristics that predict inflammatory response to C. Novyi-NT bacteriolytic immunotherapy.
BMC Vet Res. 14 (1): 119. -
DaSilva, A.V.A. et al. (2018) Morphophysiological changes in the splenic extracellular matrix of Leishmania infantum-naturally infected dogs is associated with alterations in lymphoid niches and the CD4+ T cell frequency in spleens.
PLoS Negl Trop Dis. 12 (4): e0006445. -
Lisiecka. U. et al. (2019) Evaluation of T regulatory lymphocytes and serum concentration of selected cytokines in dogs with perianal tumors.
Vet Immunol Immunopathol. 207: 10-17. -
Akiyama, S. et al. (2019) Th17 cells increase during maturation in peripheral blood of healthy dogs.
Vet Immunol Immunopathol. 209: 17-21. -
Martini, V. et al. (2019) Prognostic role of non-neoplastic lymphocytes in lymph node aspirates from dogs with diffuse large B-cell lymphoma treated with chemo-immunotherapy.
Res Vet Sci. 125: 130-5. -
Aguiar-Soares, R.D.O. et al. (2020) Phase I and II Clinical Trial Comparing the LBSap, Leishmune®, and Leish-Tec® Vaccines against Canine Visceral Leishmaniasis.
Vaccines (Basel). 8 (4): 690. -
Wolf-Ringwall, A. et al. (2020) Prospective evaluation of flow cytometric characteristics, histopathologic diagnosis and clinical outcome in dogs with naïve B-cell lymphoma treated with a 19-week CHOP protocol.
Vet Comp Oncol. 18 (3): 342-52. -
Sayag, D. et al. (2020) Proof-of-concept study: Evaluation of plasma and urinary electrolytes as markers of response to L-asparaginase therapy in dogs with high-grade lymphoma.
Vet Clin Pathol. 49 (3): 476-83. -
Lee, J. et al. (2021) Canine Natural Killer Cell-Derived Exosomes Exhibit Antitumor Activity in a Mouse Model of Canine Mammary Tumor.
Biomed Res Int. 2021: 6690704. -
Grudzien, M. et al. (2021) A newly established canine NK-type cell line and its cytotoxic properties.
Vet Comp Oncol. 19 (3): 567-77. -
Lee, S.H. et al. (2021) Safety and immunological effects of recombinant canine IL-15 in dogs.
Cytokine. 148: 155599. -
Knebel, A. et al. (2021) Measurement of canine Th17 cells by flow cytometry.
Vet Immunol Immunopathol. 243: 110366. -
Konno, H. et al. (2022) An experimental challenge model for Leishmania donovani in beagle dogs, showing a similar pattern of parasite burden in the peripheral blood and liver.
Parasitol Res. 121 (12): 3569-79. -
Kanei, T. et al. (2022) Expression and functional analysis of chemokine receptor 7 in canine lymphoma cell lines.
J Vet Med Sci. 84 (1): 25-30. -
do Prado Duzanski, A. et al. (2022) Cell-mediated immunity and expression of MHC class I and class II molecules in dogs naturally infected by canine transmissible venereal tumor: Is there complete spontaneous regression outside the experimental CTVT?
Res Vet Sci. 145: 193-204. -
Karayannopoulou, M. et al. (2022) Effect of major versus minor mastectomy on host immunity in canine mammary cancer
Vet Immunol Immunopathol. Feb 24: 110403. -
Bragato, J.P. et al. (2022) miRNA-21 regulates CD69 and IL-10 expression in canine leishmaniasis.
PLoS One. 17 (3): e0265192. -
Riccardo, F. et al. (2022) Antigen mimicry as an effective strategy to induce CSPG4-targeted immunity in dogs with oral melanoma: a veterinary trial.
J Immunother Cancer. 10(5):e004007. -
Matralis, D.T. et al. (2023) Intracellular IFN-γ and IL-4 levels of CD4 + and CD8 + T cells in the peripheral blood of naturally infected (Leishmania infantum) symptomatic dogs before and following a 4-week treatment with miltefosine and allopurinol: a double-blinded, controlled and cross-sectional study.
Acta Vet Scand. 65 (1): 2. -
Hamouzová, P. et al. (2023) Lymphocyte immunophenotyping in dogs with lymphopenia of common causes.
Vet Immunol Immunopathol. 261: 110620. -
Tarone, L. et al. (2023) A chimeric human/dog-DNA vaccine against CSPG4 induces immunity with therapeutic potential in comparative preclinical models of osteosarcoma.
Mol Ther. 31 (8): 2342-59. -
Yamauchi, A. et al. (2023) Negative Influence of Aging on Differentiation and Proliferation of CD8(+) T-Cells in Dogs.
Vet Sci. 10 (9): 541. -
Bencze, M. et al. (2023) Receptor interacting protein kinase-3 mediates both myopathy and cardiomyopathy in preclinical animal models of Duchenne muscular dystrophy.
J Cachexia Sarcopenia Muscle. Nov 01 [Epub ahead of print]. -
Martini, V. et al. (2018) A retrospective study of flow cytometric characterization of suspected extranodal lymphomas in dogs.
J Vet Diagn Invest. 30 (6): 830-6. -
DeClue, A.E. et al. (2020) Transportation and Routine Veterinary Interventions Alter Immune Function in the Dog.
Top Companion Anim Med. 39: 100408. -
Lee, G.W. et al. (2021) Case Report: Long-Term Survival of a Dog With Chronic Lymphocytic Leukemia Treated With Chlorambucil, Prednisolone, and Imatinib.
Front Vet Sci. 8: 625527. -
Sainz, Á. et al. (2021) Effect of chemically modified tetracycline-8 (CMT-8) on hematology, blood chemistry, cytokines and peripheral blood lymphocyte subsets of healthy dogs.
Res Vet Sci. 136: 200-8. -
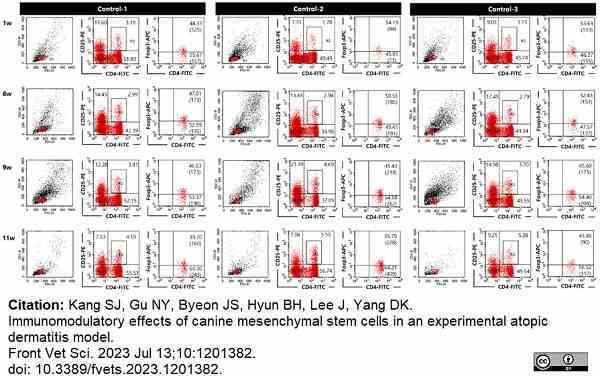
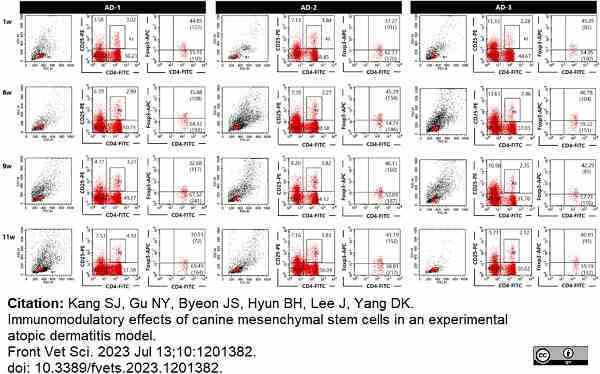
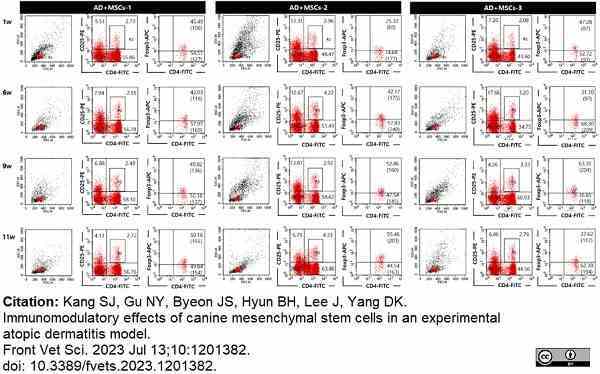
Kang, S.J. et al. (2023) Immunomodulatory effects of canine mesenchymal stem cells in an experimental atopic dermatitis model.
Front Vet Sci. 10: 1201382. -
Sheng, R. et al. (2023) Prognostic significance of CD25 expression in dogs with a noninvasive diagnosis of B-cell lymphoma treated with CHOP chemotherapy.
Vet Comp Oncol. 21 (1): 28-35. -
Miguelena Chamorro, B. et al. (2023) Characterization of Canine Peyer's Patches by Multidimensional Analysis: Insights from Immunofluorescence, Flow Cytometry, and Single-Cell RNA Sequencing.
Immunohorizons. 7 (11): 788-805.
- UniProt
- P33705
- Entrez Gene
- CD4
- GO Terms
- GO:0007155 cell adhesion
- GO:0016021 integral to membrane
- GO:0006955 immune response
- GO:0045058 T cell selection
MCA1038GA
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Dog ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up