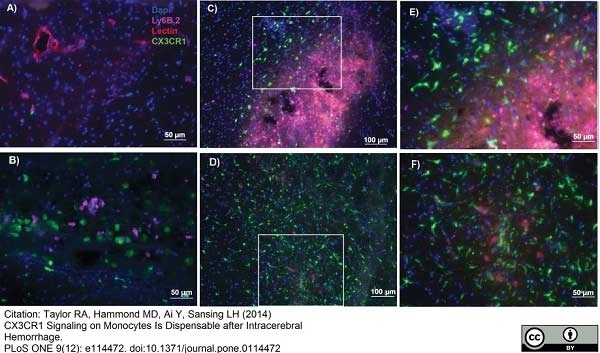
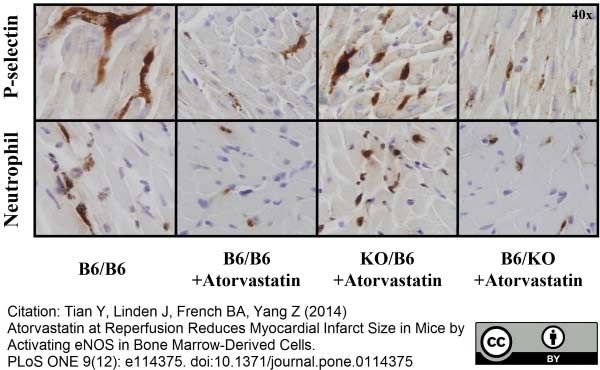
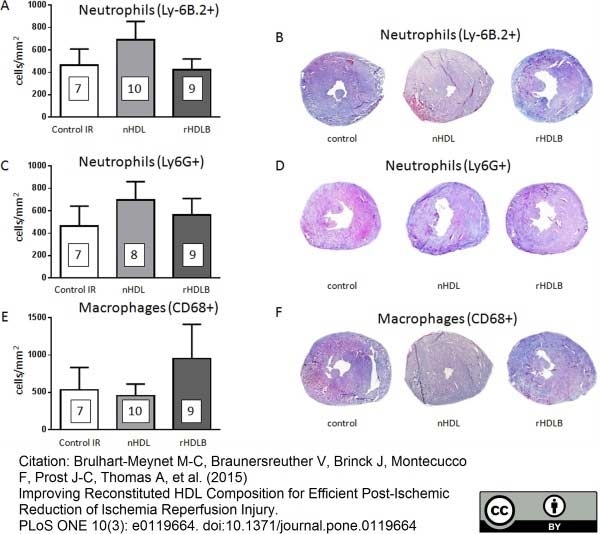
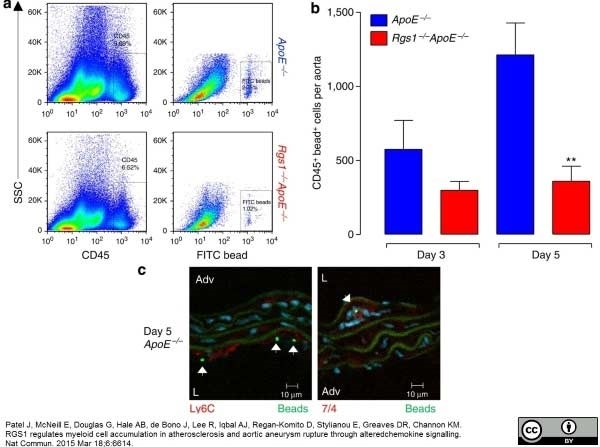
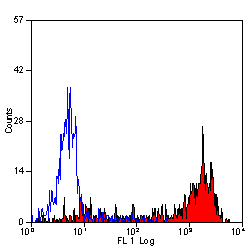
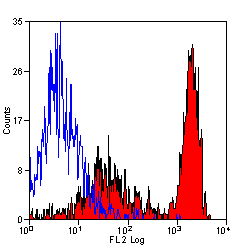
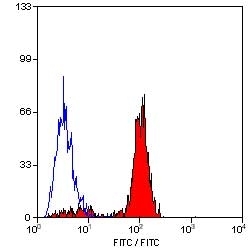
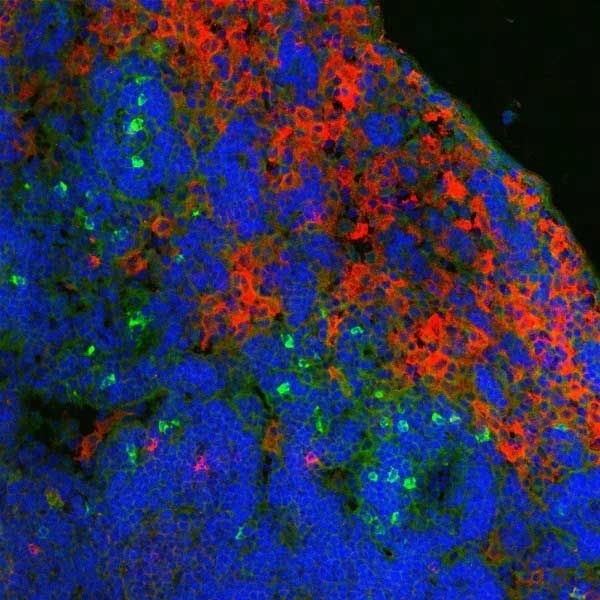
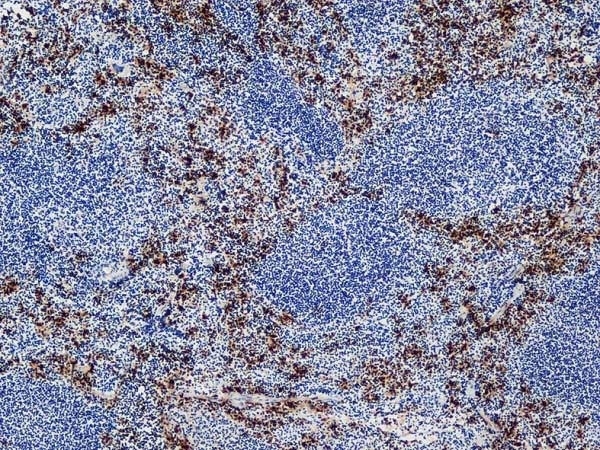
Ly-6B.2 Alloantigen antibody | 7/4











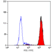
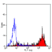
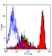


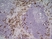
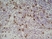





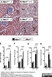

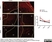
























Rat anti Mouse Ly-6B.2 Alloantigen
- Product Type
- Monoclonal Antibody
- Clone
- 7/4
- Isotype
- IgG2a
- Specificity
- Ly-6B.2 Alloantigen
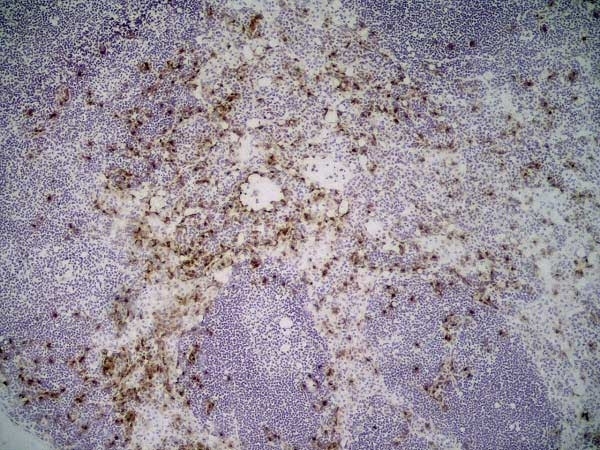
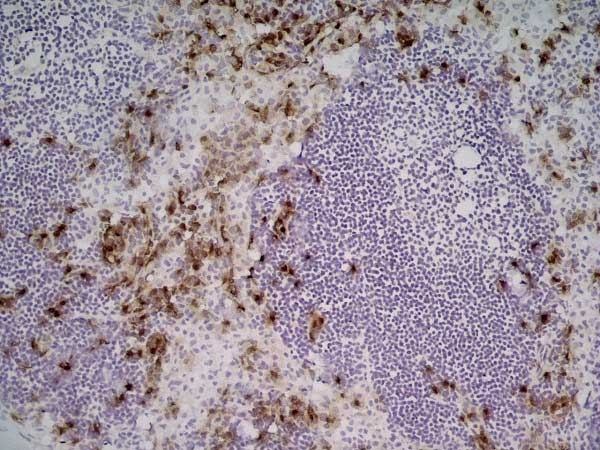
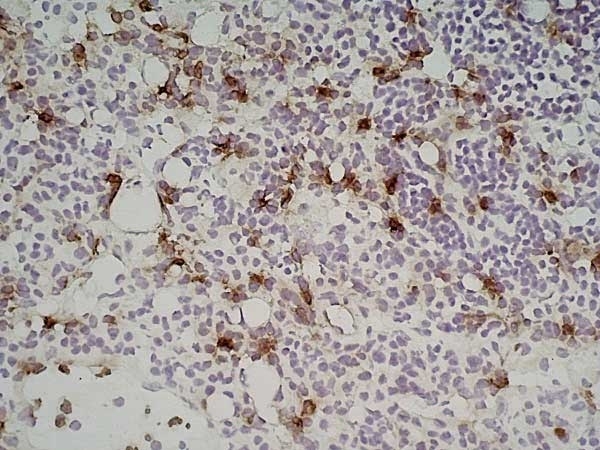
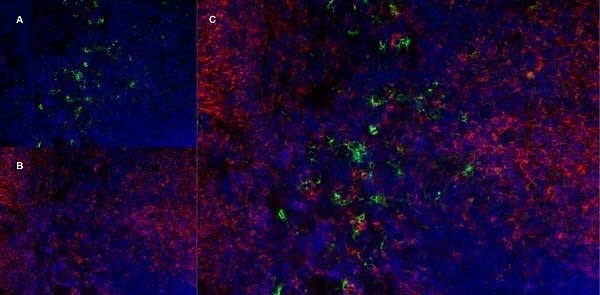
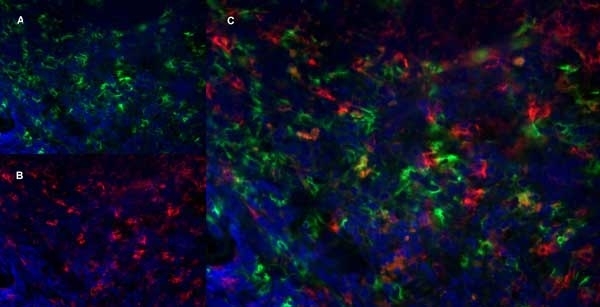
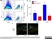
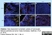
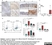
| Rat anti Mouse Ly-6B.2 monoclonal antibody, clone 7/4 recognizes the Ly-6B.2 antigen. Ly-6B.2 is a ~25-30 kDa GPI-anchored, heavily glycosylated protein expressed on neutrophils, inflammatory monocytes and some activated macrophages (Rosas et al. 2010). High levels of expression are seen in bone marrow, spleen, lung and lymph nodes. N-glycanase treatment of thioglycollate elicited peritoneal neutrophil lysates lowers the apparent molecular weight of Ly-6B.2 to ~15 kDa (Rosas et al.2010). In common with other Ly-6 antigens Ly-6B.2 demonstrates a polymorphic expression on inbred mouse strains (Kimura et al. 1984). Rat anti mouse Ly-6B.2, clone 7/4 recognizes the Ly-6B.2 antigen in 129J; AKR; C57BL/6; C57BL/10; C58; DBA/2; NZB; NZW; SJL; MFI; Swiss (PO) Strains whilst A2G; A/Sn; ASW; BALB/c; C3H/HEH: CBA.T6T6 are negative or demonstrate very weak reactivity (Hirsch and Gordon 1982). Rat anti mouse Ly-6B.2 has been successfully used for the immunomagnetic depletion of neutrophils during the enrichment of primitive hematopoietic cells from bone marrow (Bertoncello et al. 1991) and the depletion of myeloid cells in vivo (Rosas et al. 2010). |
- Target Species
- Mouse
- Product Form
- Purified IgG - liquid
- Preparation
- MCA771GT: Purified IgG prepared by affinity chromatography on Protein G from tissue culture supernatant.
- MCA771GA, MCA771G: Purified IgG prepared by affinity chromatography on Protein G from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
0.09% Sodium Azide - Carrier Free
- Yes
- Immunogen
- Cultured bone marrow cells
- Approx. Protein Concentrations
- MCA771GT, MCA771G: IgG concentration 1.0 mg/ml
- MCA771GA: IgG concentration 1 mg/ml
- Fusion Partners
- Spleen cells from AO rats were fused with cells from the Y3 Ag1.2.3 rat myeloma cell line.
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
This product should be stored undiluted. Should this product contain a precipitate we recommend microcentrifugation before use.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Flow Cytometry | 1/50 | 1/100 | |
| Immunofluorescence | |||
| Immunohistology - Frozen | |||
| Immunohistology - Paraffin | |||
| Western Blotting |
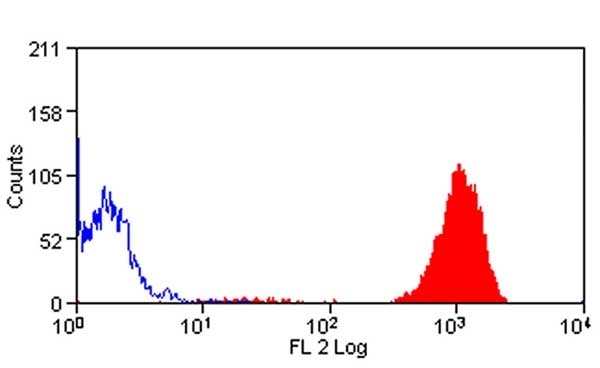
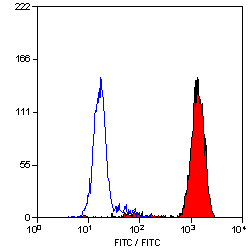
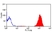
- Flow Cytometry
- Use 10ul of the suggested working dilution to label 106 cells in 100ul.
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Rat IgG2a Negative Control | MCA1212 | E F | 1 ml |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Rat IgG2a Negative Control | ||||||
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Antigen Retrieval Buffer, pH8.0 | BUF025A | P | 500 ml | Log in | |||
| List Price | Your Price | ||||||
| Log in | |||||||
| Description | Antigen Retrieval Buffer, pH8.0 | ||||||
Source Reference
-
Hirsch, S. & Gordon, S. (1983) Polymorphic expression of a neutrophil differentiation antigen revealed by monoclonal antibody 7/4.
Immunogenetics. 18 (3): 229-39.
References for Ly-6B.2 Alloantigen antibody
-
Gordon, S. et al. (1992) Antigen markers of macrophage differentiation in murine tissues.
Curr Top Microbiol Immunol. 181: 1-37. -
Horiba, M. et al. (2000) Neointima formation in a restenosis model is suppressed in midkine-deficient mice.
J Clin Invest. 105: 489-95. -
Pyo, R. et al. (2000) Targeted gene disruption of matrix metalloproteinase-9 (gelatinase B) suppresses development of experimental abdominal aortic aneurysms.
J Clin Invest. 105: 1641-9. -
Liao, C. et al. (2001) Altered myelopoiesis and the development of acute myeloid leukemia in transgenic mice overexpressing cyclin A1.
Proc Natl Acad Sci U S A. 98: 6853-8. -
Song, Y. et al. (2001) A low level of TNF-alpha mediates hemorrhage-induced acute lung injury via p55 TNF receptor.
Am J Physiol Lung Cell Mol Physiol. 281: L677-84. -
Singbartl, K. et al. (2001) Platelet, but not endothelial, P-selectin is critical for neutrophil-mediated acute postischemic renal failure.
FASEB J. 15: 2337-44. -
Endlich, B. et al. (2002) Distinct temporal patterns of macrophage-inflammatory protein-2 and KC chemokine gene expression in surgical injury.
J Immunol. 168: 3586-94. -
Lacroix-Lamandé, S. et al. (2002) Role of gamma interferon in chemokine expression in the ileum of mice and in a murine intestinal epithelial cell line after Cryptosporidium parvum infection.
Infect Immun. 70: 2090-9.
View The Latest Product References
-
Sato, J. et al. (2003) The fibrinolytic system in dissemination and matrix protein deposition during a mycobacterium infection.
Am J Pathol. 163: 517-31. -
Chen, Z. et al. (2004) Evidence for a role of macrophage migration inhibitory factor in vascular disease.
Arterioscler Thromb Vasc Biol. 24: 709-14. -
Lee, H.T. et al. (2004) A1 adenosine receptor activation inhibits inflammation, necrosis, and apoptosis after renal ischemia-reperfusion injury in mice.
J Am Soc Nephrol. 15: 102-11. -
Day, Y.J. et al. (2004) Protection from ischemic liver injury by activation of A2A adenosine receptors during reperfusion: inhibition of chemokine induction.
Am J Physiol Gastrointest Liver Physiol. 286: G285-93. -
Crockett, E.T. et al. (2004) Gene deletion of P-Selectin and ICAM-1 does not inhibit neutrophil infiltration into peritoneal cavity following cecal ligation-puncture.
BMC Clin Pathol. 4: 2 -
Kaur, S. et al. (2004) Acidic duodenal pH alters gene expression in the cystic fibrosis mouse pancreas.
Am J Physiol Gastrointest Liver Physiol. 287 (2): G480-90. -
Thatcher, T.H. et al. (2005) Role of CXCR2 in cigarette smoke-induced lung inflammation.
Am J Physiol Lung Cell Mol Physiol. 289: L322-8. -
Iwaki, T. et al. (2005) A cardioprotective role for the endothelial protein C receptor in lipopolysaccharide-induced endotoxemia in the mouse.
Blood. 105: 2364-71. -
Word, R.A. et al. (2005) Transgene insertion on mouse chromosome 6 impairs function of the uterine cervix and causes failure of parturition.
Biol Reprod. 73 (5): 1046-56. -
Sasaki, T. et al. (2006) A simple method of plaque rupture induction in apolipoprotein E-deficient mice.
Arterioscler Thromb Vasc Biol. 26: 1304-9. -
Chen, C.J. et al. (2006) MyD88-dependent IL-1 receptor signaling is essential for gouty inflammation stimulated by monosodium urate crystals.
J Clin Invest. 2006 Aug;116(8):2262-71. -
Timmons, B.C. & Mahendroo, M.S. (2006) Timing of neutrophil activation and expression of proinflammatory markers do not support a role for neutrophils in cervical ripening in the mouse.
Biol Reprod. 74: 236-45. -
Morison, N.B. et al. (2007) The long-term actions of etonogestrel and levonorgestrel on decidualized and non-decidualized endometrium in a mouse model mimic some effects of progestogen-only contraceptives in women.
Reproduction. 133: 309-21. -
Cheng, C.W. et al. (2007) Quantitative cellular and molecular analysis of the effect of progesterone withdrawal in a murine model of decidualization.
Biol Reprod. 76: 871-83. -
Tsou, C.L. et al. (2007) Critical roles for CCR2 and MCP-3 in monocyte mobilization from bone marrow and recruitment to inflammatory sites.
J Clin Invest. 117: 902-9. -
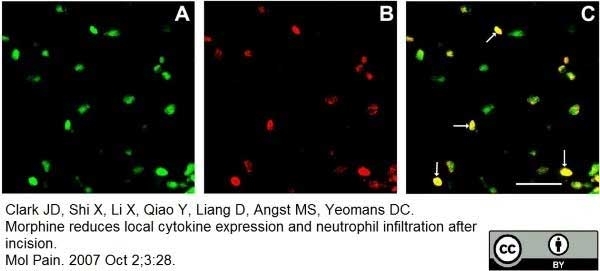
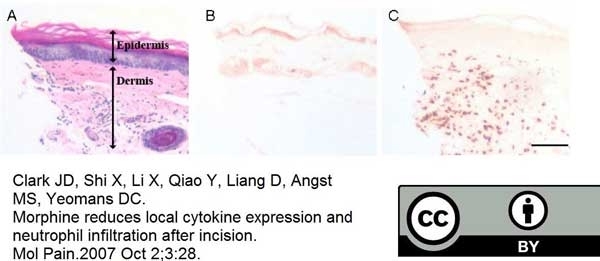
Clark, J.D. et al. (2007) Morphine reduces local cytokine expression and neutrophil infiltration after incision.
Mol Pain. 3:28. -
Laubitz, D. et al. (2008) Colonic gene expression profile in NHE3-deficient mice: evidence for spontaneous distal colitis.
Am J Physiol Gastrointest Liver Physiol. 295: G63-G77. -
Shornick, L.P. et al. (2008) Airway epithelial versus immune cell Stat1 function for innate defense against respiratory viral infection.
J Immunol. 180: 3319-28. -
Kato, N. et al. (2009) The E-selectin ligand basigin/CD147 is responsible for neutrophil recruitment in renal ischemia/reperfusion.
J Am Soc Nephrol. 20: 1565-76. -
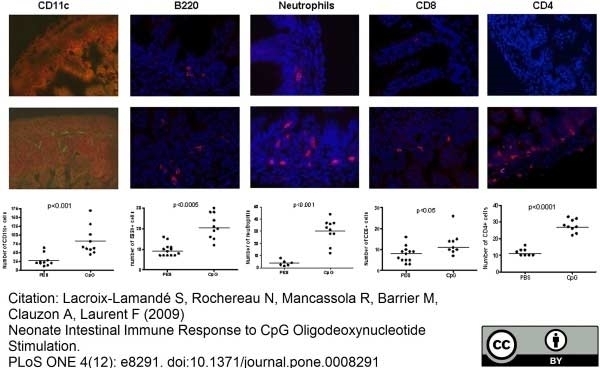
Lacroix-Lamandé, S. et al. (2009) Neonate intestinal immune response to CpG oligodeoxynucleotide stimulation.
PLoS One. 4: e8291. -
Park, S.W. et al. (2009) Human activated protein C attenuates both hepatic and renal injury caused by hepatic ischemia and reperfusion injury in mice.
Kidney Int. 76 (7): 739-50. -
Locke, L.W. (2009) A novel neutrophil-specific PET imaging agent: cFLFLFK-PEG-64Cu.
J Nucl Med. 50: 790-7. -
Omari, K.M. et al. (2009) Neuroprotection and remyelination after autoimmune demyelination in mice that inducibly overexpress CXCL1.
Am J Pathol. 174: 164-76. -
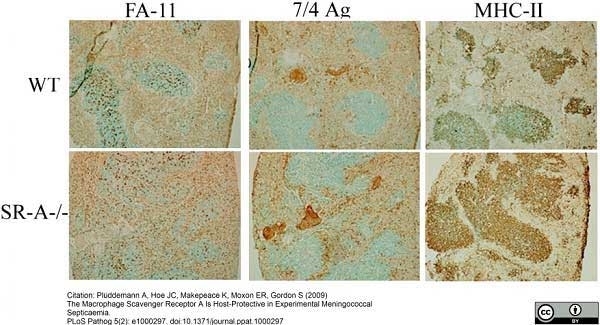
Plüddemann, A. (2009) The macrophage scavenger receptor A is host-protective in experimental meningococcal septicaemia.
PLoS Pathog. 5:e1000297. -
Lech, M. et al. (2009) Resident dendritic cells prevent postischemic acute renal failure by help of single Ig IL-1 receptor-related protein.
J Immunol. 183: 4109-18. -
Bombardelli, L. et al. (2010) Pancreas-specific ablation of beta1 integrin induces tissue degeneration by disrupting acinar cell polarity.
Gastroenterology. 138: 2531-40, -
Schneider, D.J. et al. (2010) Adenosine and osteopontin contribute to the development of chronic obstructive pulmonary disease.
FASEB J. 24: 70-80. -
Rosas, M. et al. (2010) The myeloid 7/4-antigen defines recently generated inflammatory macrophages and is synonymous with Ly-6B.
J Leukoc Biol. 88 (1): 169-80. -
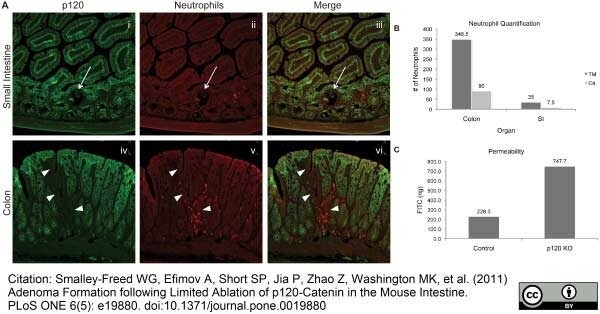
Smalley-Freed, W.G. et al. (2010) p120-catenin is essential for maintenance of barrier function and intestinal homeostasis in mice.
J Clin Invest. 120: 1824-35. -
Lassen, S. et al. (2010) Ischemia reperfusion induces IFN regulatory factor 4 in renal dendritic cells, which suppresses postischemic inflammation and prevents acute renal failure
J Immunol. 185: 1976-83. -
Kim, M. et al. (2010) Selective renal overexpression of human heat shock protein 27 reduces renal ischemia-reperfusion injury in mice.
Am J Physiol Renal Physiol. 299: F347-58. -
Shi, H. et al. (2010) MAP kinase phosphatase-1 deficiency impairs skeletal muscle regeneration and exacerbates muscular dystrophy.
FASEB J. 24: 2985-97. -
Nadeau, S. et al. (2011) Functional Recovery after Peripheral Nerve Injury is Dependent on the Pro-Inflammatory Cytokines IL-1{beta} and TNF: Implications for Neuropathic Pain.
J Neurosci. 31: 12533-12542. -
Guerriero, J.L. et al. (2011) DNA alkylating therapy induces tumor regression through an HMGB1-mediated activation of innate immunity.
J Immunol. 186: 3517-26. -
Yellon, S.M. et al. (2011) Remodeling of the cervix and parturition in mice lacking the progesterone receptor B isoform.
Biol Reprod. 85: 498-502. -
Frossard, J.L. et al. (2011) Role of CCL-2, CCR-2 and CCR-4 in cerulein-induced acute pancreatitis and pancreatitis-associated lung injury.
J Clin Pathol. 64: 387-93. -
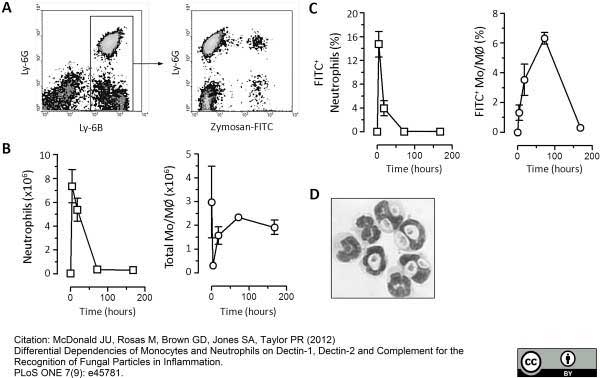
McDonald, J.U. et al. (2011) In vivo functional analysis and genetic modification of in vitro-derived mouse
neutrophils.
FASEB J. 25: 1972-82. -
Larmonier, C.B. et al. (2011) NHE3 modulates the severity of colitis in IL-10-deficient mice.
Am J Physiol Gastrointest Liver Physiol. 300: G998-G1009. -
Allam, R. et al. (2011) Cyclic Polypeptide and Aminoglycoside Antibiotics Trigger IL-1{beta} Secretion by Activating the NLRP3 Inflammasome.
J Immunol. 186: 2714-8. -
Holt, R. et al. (2011) The Molecular Mechanisms of Cervical Ripening Differ between Term and Preterm Birth.
Endocrinology. 152: 1036-46. -
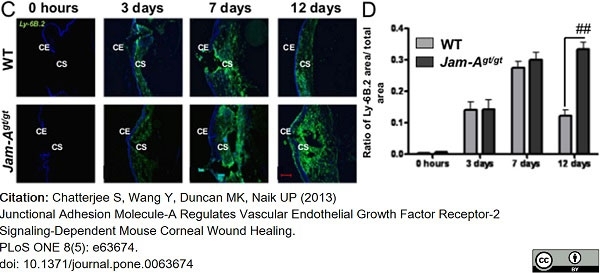
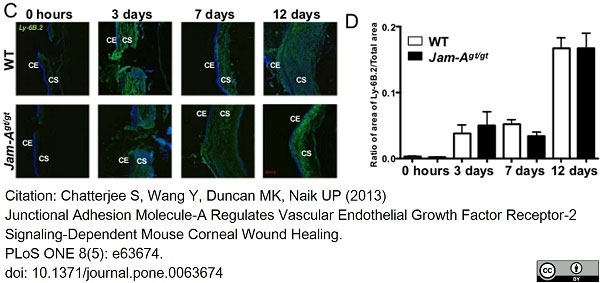
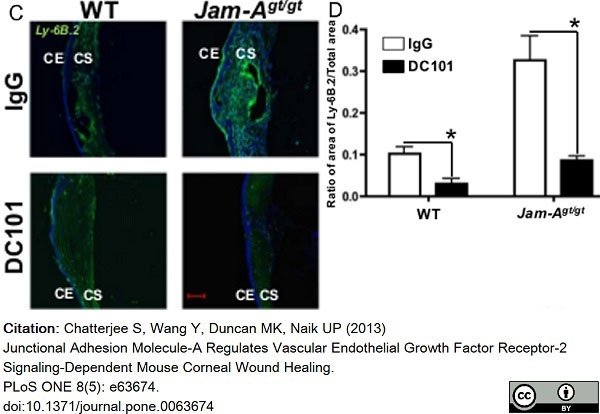
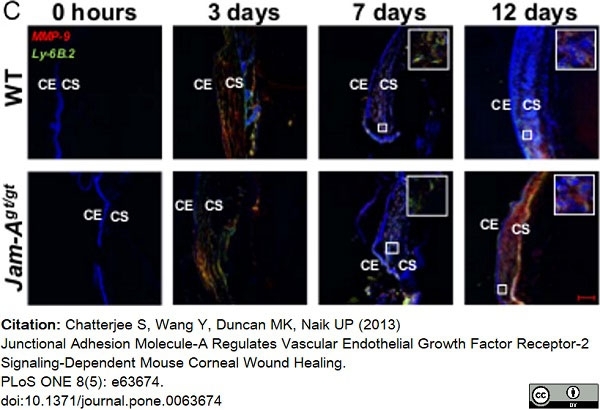
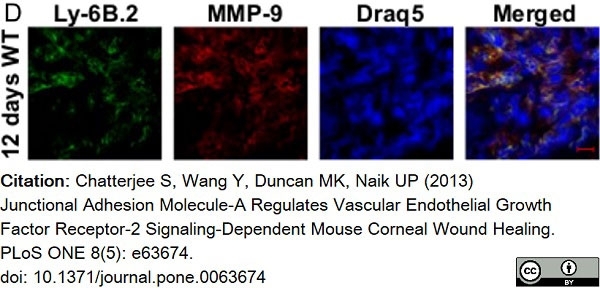
Chatterjee, S. et al. (2013) Junctional adhesion molecule-A regulates vascular endothelial growth factor receptor-2 signaling-dependent mouse corneal wound healing.
PLoS One. 8 (5): e63674. -
Brulhart-Meynet, M.C. et al. (2015) Improving Reconstituted HDL Composition for Efficient Post-Ischemic Reduction of Ischemia Reperfusion Injury.
PLoS One. 10 (3): e0119664. -
Patel, J. et al. (2015) RGS1 regulates myeloid cell accumulation in atherosclerosis and aortic aneurysm rupture through altered chemokine signalling.
Nat Commun. 6: 6614. -
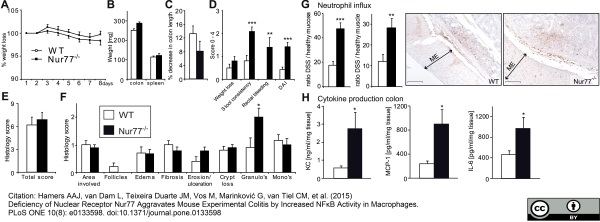
Hamers, A.A. et al. (2015) Deficiency of Nuclear Receptor Nur77 Aggravates Mouse Experimental Colitis by Increased NFκB Activity in Macrophages.
PLoS One. 10 (8): e0133598. -
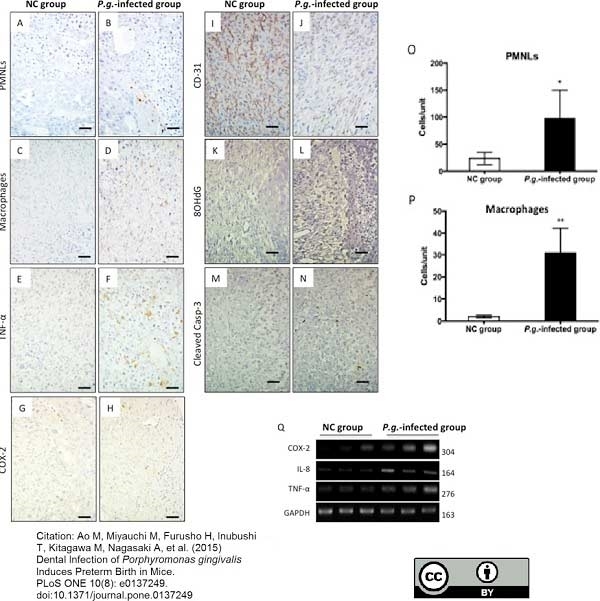
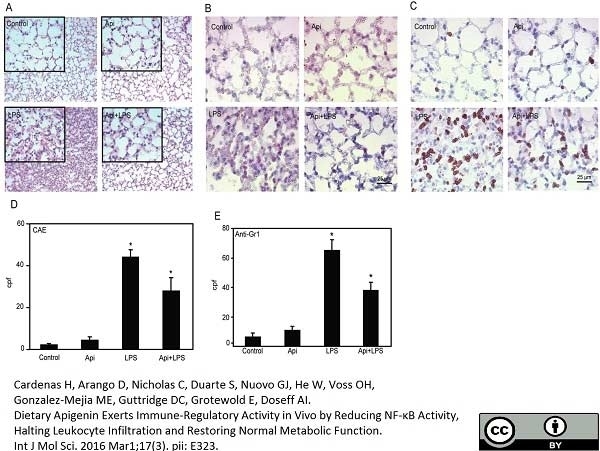
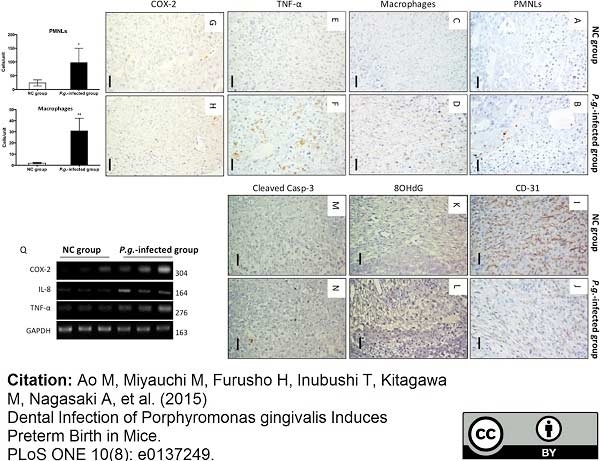
Ao, M. et al. (2015) Dental Infection of Porphyromonas gingivalis Induces Preterm Birth in Mice.
PLoS One. 10 (8): e0137249. -
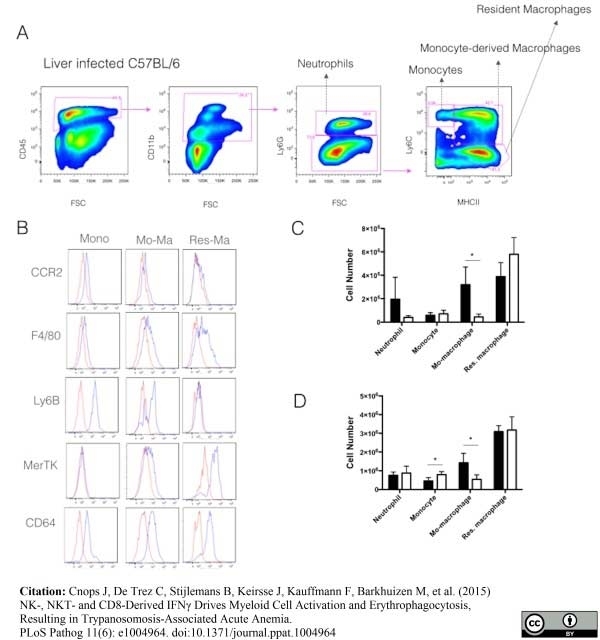
Cnops, J. et al. (2015) NK-, NKT- and CD8-Derived IFNγ Drives Myeloid Cell Activation and Erythrophagocytosis, Resulting in Trypanosomosis-Associated Acute Anemia.
PLoS Pathog. 11 (6): e1004964. -
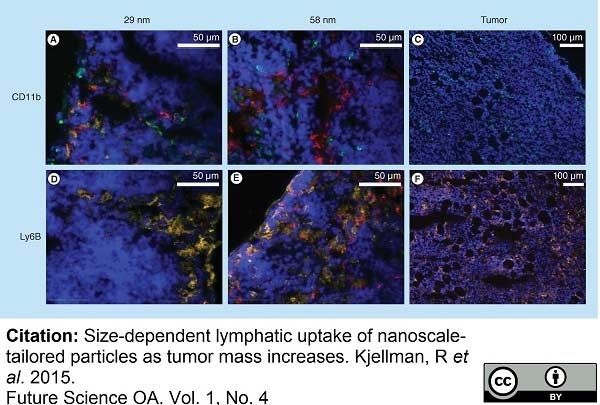
Kjellman, P. et al. (2015) Size-dependent lymphatic uptake of nanoscale-tailored particles as tumor mass increases.
Future Sci OA. 1 (4): FSO60. -
Wan, W. et al. (2015) Atypical chemokine receptor 1 deficiency reduces atherogenesis in ApoE-knockout mice.
Cardiovasc Res. 106 (3): 478-87. -
Kidoya, H. et al. (2015) APJ Regulates Parallel Alignment of Arteries and Veins in the Skin.
Dev Cell. 33 (3): 247-59. -
Brennan, F.H. et al. (2015) The Complement Receptor C5aR Controls Acute Inflammation and Astrogliosis following Spinal Cord Injury.
J Neurosci. 35 (16): 6517-31. -
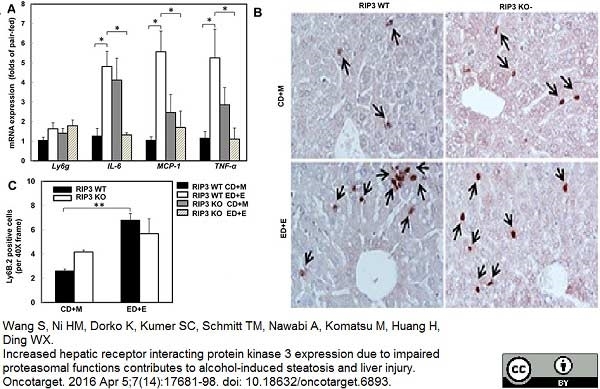
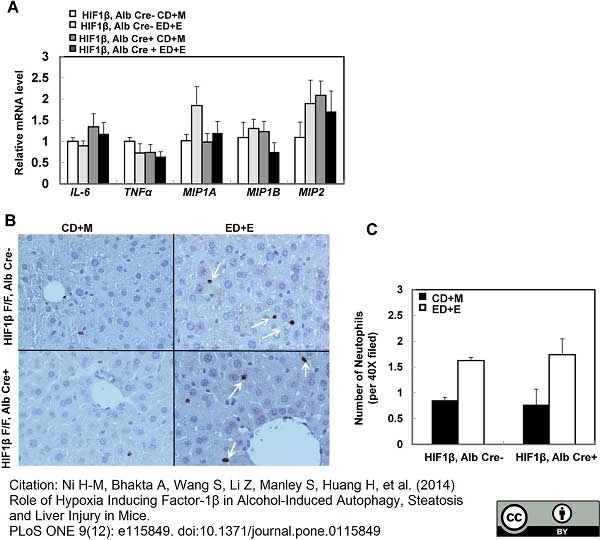
Wang, S. et al. (2016) Increased hepatic receptor interacting protein kinase 3 expression due to impaired proteasomal functions contributes to alcohol-induced steatosis and liver injury.
Oncotarget. 7 (14): 17681-98. -
Ni, H.M. et al. (2016) Caspase Inhibition Prevents Tumor Necrosis Factor-α-Induced Apoptosis and Promotes Necrotic Cell Death in Mouse Hepatocytes in Vivo and in Vitro.
Am J Pathol. 186 (10): 2623-36. -
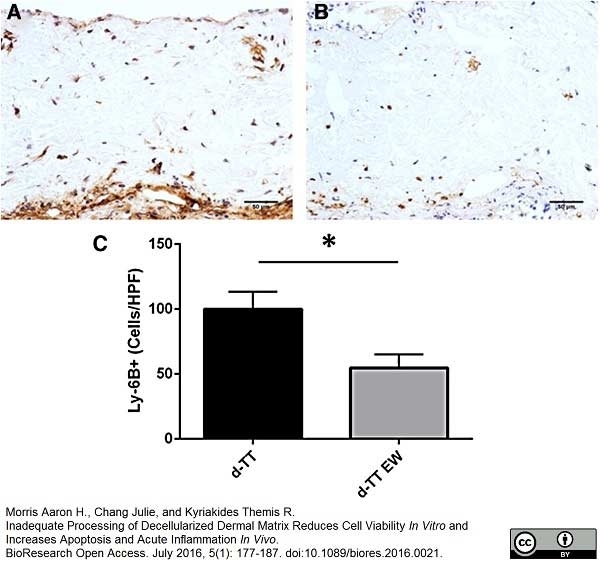
Morris, A.H. et al. (2016) Inadequate Processing of Decellularized Dermal Matrix Reduces Cell Viability In Vitro and Increases Apoptosis and Acute Inflammation In Vivo.
Biores Open Access. 5 (1): 177-87. -
Rabadi, M. et al. (2016) Peptidyl arginine deiminase-4-deficient mice are protected against kidney and liver injury after renal ischemia and reperfusion.
Am J Physiol Renal Physiol. 311 (2): F437-49. -
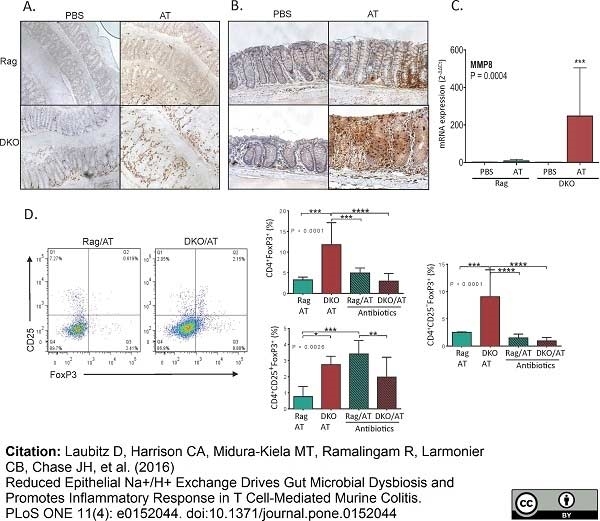
Laubitz, D. et al. (2016) Reduced Epithelial Na+/H+ Exchange Drives Gut Microbial Dysbiosis and Promotes Inflammatory Response in T Cell-Mediated Murine Colitis.
PLoS One. 11 (4): e0152044. -
Farrar, C.A. et al. (2016) Collectin-11 detects stress-induced L-fucose pattern to trigger renal epithelial injury.
J Clin Invest. 126 (5): 1911-25. -
Zhao, Y. et al. (2016) Rapamycin prevents bronchiolitis obliterans through increasing infiltration of regulatory B cells in a murine tracheal transplantation model.
J Thorac Cardiovasc Surg. 151 (2): 487-496.e3. -
Choi, E. et al. (2016) Expression of Activated Ras in Gastric Chief Cells of Mice Leads to the Full Spectrum of Metaplastic Lineage Transitions.
Gastroenterology. 150 (4): 918-30.e13. -
Nguyen, H.T. & Shen, H. (2016) The effect of PEGylation on the stimulation of IL-1β by gold (Au) nanoshell/silica core nanoparticles.
J Mater Chem B Mater Biol Med. 4 (9): 1650-9. -
Boal, F. et al. (2016) PI5P Triggers ICAM-1 Degradation in Shigella Infected Cells, Thus Dampening Immune Cell Recruitment.
Cell Rep. 14 (4): 750-9. -
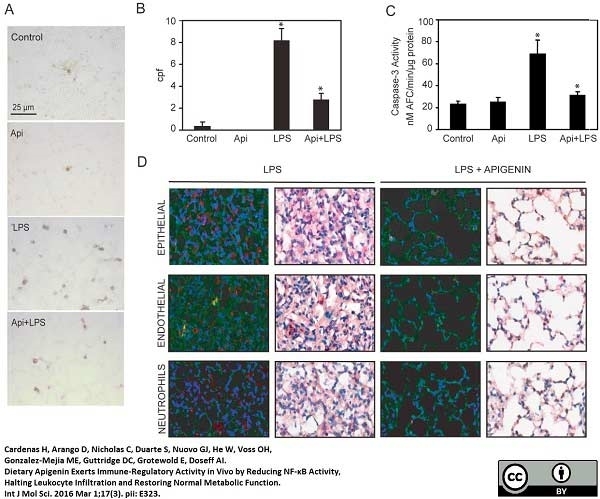
Cardenas, H. et al. (2016) Dietary Apigenin Exerts Immune-Regulatory Activity in Vivo by Reducing NF-κB Activity, Halting Leukocyte Infiltration and Restoring Normal Metabolic Function.
Int J Mol Sci. 17 (3): 323. -
Wieser, V. et al. (2016) Lipocalin 2 drives neutrophilic inflammation in alcoholic liver disease.
J Hepatol. 64 (4): 872-80. -
Konrad, F.M. et al. (2019) How Adhesion Molecule Patterns Change While Neutrophils Traffic through the Lung during Inflammation.
Mediators Inflamm. 2019: 1208086. -
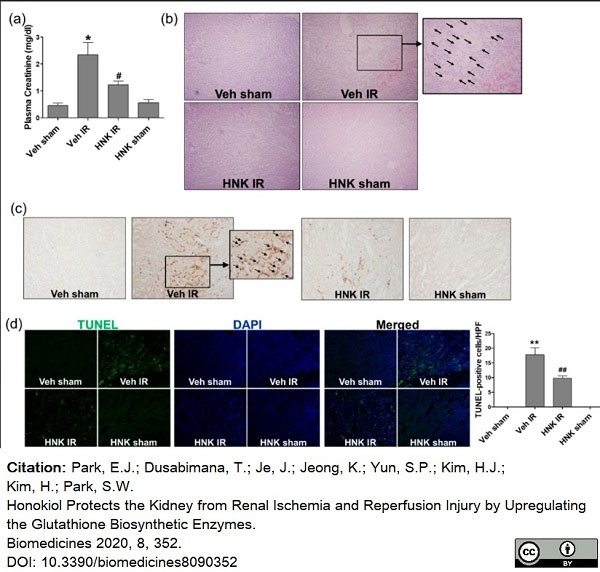
Jung, P.E. et al. (2020) Honokiol Protects the Kidney from Renal Ischemia and Reperfusion Injury by Upregulating the Glutathione Biosynthetic Enzymes.
Biomedicines. 8 (9): 352. -
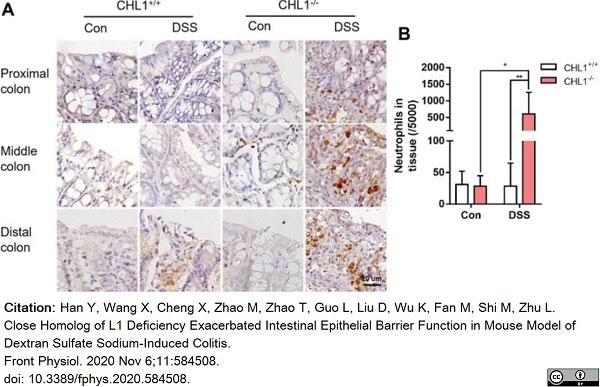
Han, Y. et al. (2020) Close Homolog of L1 Deficiency Exacerbated Intestinal Epithelial Barrier Function in Mouse Model of Dextran Sulfate Sodium-Induced Colitis.
Front Physiol. 11: 584508. -
Tilstra, J.S. et al. (2020) B cell-intrinsic TLR9 expression is protective in murine lupus.
J Clin Invest. 130 (6): 3172-3187. -
Gordon, R.A. et al. (2020) Murine lupus is neutrophil elastase-independent in the MRL.Faslpr model.
PLoS One. 15 (4): e0226396. -
Hawkins, R.B. et al. (2021) Mesenchymal Stem Cells Alter MicroRNA Expression and Attenuate Thoracic Aortic Aneurysm Formation.
J Surg Res. 268: 221-31. -
Leinweber, J. et al. (2021) Elastase inhibitor agaphelin protects from acute ischemic stroke in mice by reducing thrombosis, blood-brain barrier damage, and inflammation.
Brain Behav Immun. S0889-1591(20)32485-5. -
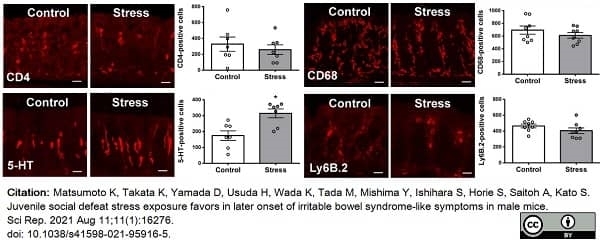
Matsumoto, K. et al. (2021) Juvenile social defeat stress exposure favors in later onset of irritable bowel syndrome-like symptoms in male mice.
Sci Rep. 11 (1): 16276. -
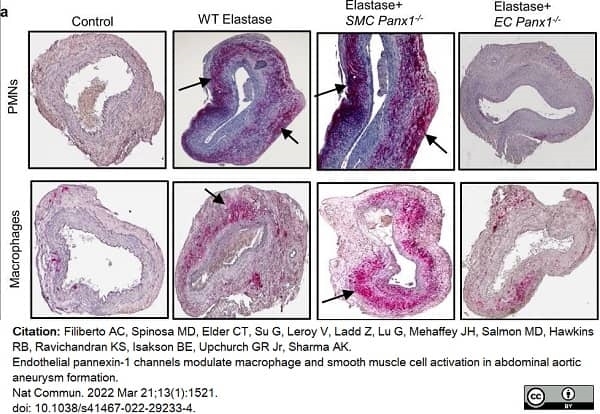
Filiberto, A.C. et al. (2022) Endothelial pannexin-1 channels modulate macrophage and smooth muscle cell activation in abdominal aortic aneurysm formation.
Nat Commun. 13 (1): 1521. -
Chute, M. et al. (2022) ADAM15 is required for optimal collagen cross-linking and scar formation following myocardial infarction.
Matrix Biol. 105: 127-43. -
Beuker, C. et al. (2022) Stroke induces disease-specific myeloid cells in the brain parenchyma and pia.
Nat Commun. 13 (1): 945. -
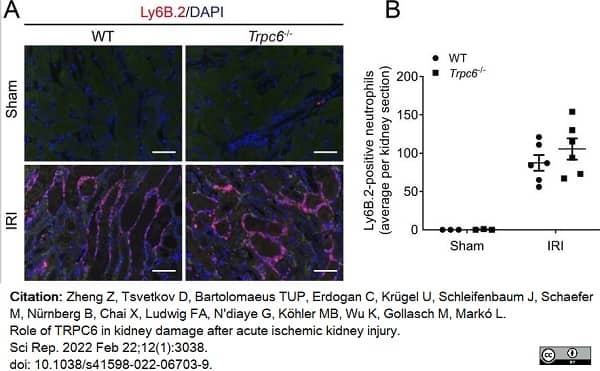
Zheng, Z. et al. (2022) Role of TRPC6 in kidney damage after acute ischemic kidney injury.
Sci Rep. 12 (1): 3038. -
Lyubenov, L. et al. (2022) Intravenous Glu-plasminogen attenuates cholesterol crystal embolism-induced thrombotic angiopathy, acute kidney injury and kidney infarction.
Nephrol Dial Transplant. gfac273. -
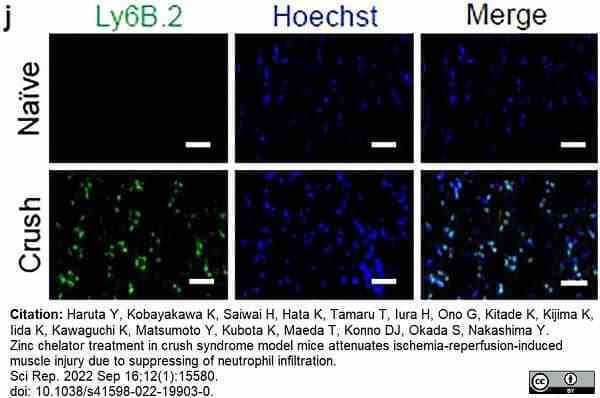
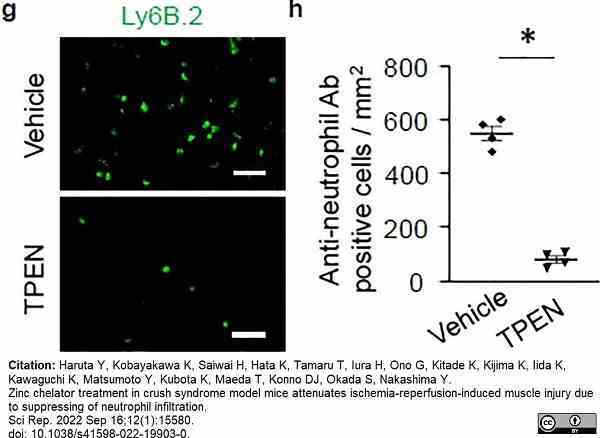
Haruta, Y. et al. (2022) Zinc chelator treatment in crush syndrome model mice attenuates ischemia-reperfusion-induced muscle injury due to suppressing of neutrophil infiltration.
Sci Rep. 12 (1): 15580. -
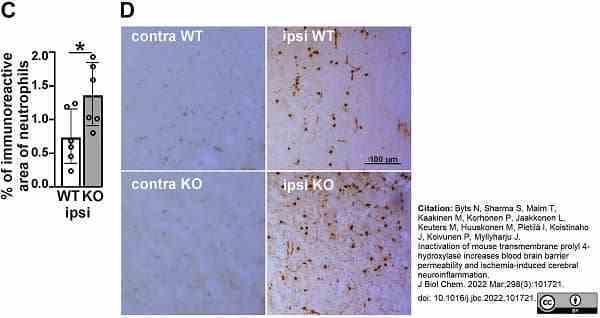
Byts, N. et al. (2022) Inactivation of mouse transmembrane prolyl 4-hydroxylase increases blood brain barrier permeability and ischemia-induced cerebral neuroinflammation.
J Biol Chem. 298 (3): 101721. -
Salmon, M. et al. (2022) Genetic and Pharmacological Disruption of Interleukin-1α Leads to Augmented Murine Aortic Aneurysm.
Ann Vasc Surg. 85: 358-370. -
Dardenne, C. et al. (2022) Topical Aspirin Administration Improves Cutaneous Wound Healing in Diabetic Mice Through a Phenotypic Switch of Wound Macrophages Toward an Anti-inflammatory and Proresolutive Profile Characterized by LXA4 Release.
Diabetes. 71 (10): 2181-96. -
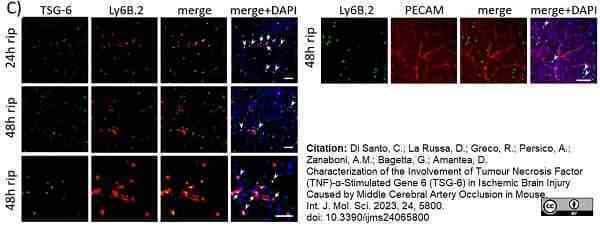
Di Santo, C.et al. (2023) Characterization of the Involvement of Tumour Necrosis Factor (TNF)-α-Stimulated Gene 6 (TSG-6) in Ischemic Brain Injury Caused by Middle Cerebral Artery Occlusion in Mouse
Int J Mol Sci. 24 (6): 5800. -
Yang, L. et al. (2023) Both hyperglycemia and hyperuricemia aggravate acute kidney injury during cholesterol embolism syndrome despite opposite effects on kidney infarct size.
Kidney Int. 104 (1): 139-150. -
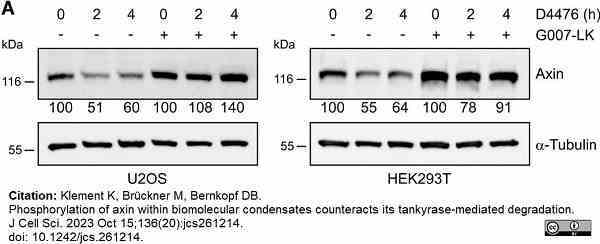
Klement, K. et al. (2023) Phosphorylation of axin within biomolecular condensates counteracts its tankyrase-mediated degradation.
J Cell Sci. 136 (20): jcs261214. -
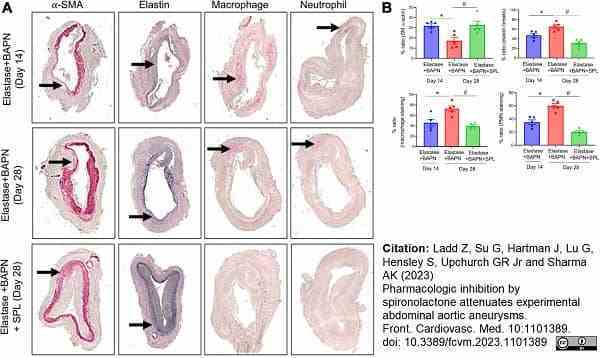
Ladd, Z. et al. (2023) Pharmacologic inhibition by spironolactone attenuates experimental abdominal aortic aneurysms.
Front Cardiovasc Med. 10: 1101389. -
Gilfillan, C.B. et al. (2018) Clec9A(+) Dendritic Cells Are Not Essential for Antitumor CD8(+) T Cell Responses Induced by Poly I:C Immunotherapy.
J Immunol. 200 (8): 2978-86. -
Lopes, J.P. et al. (2018) Evasion of Immune Surveillance in Low Oxygen Environments Enhances Candida albicans Virulence.
mBio. 9 (6): e02120-18. -
Howard, M.C. et al. (2020) l-Fucose prevention of renal ischaemia/reperfusion injury in Mice.
FASEB J. 34 (1): 822-34. -
Grist, J.J. et al. (2018) Induced CNS expression of CXCL1 augments neurologic disease in a murine model of multiple sclerosis via enhanced neutrophil recruitment.
Eur J Immunol. 48 (7): 1199-210. -
Urbschat, A. et al. (2018) Systemic TLR2 Antibody Application in Renal Ischaemia and Reperfusion Injury Decreases AKT Phosphorylation and Increases Apoptosis in the Mouse Kidney.
Basic Clin Pharmacol Toxicol. 122 (2): 223-32. -
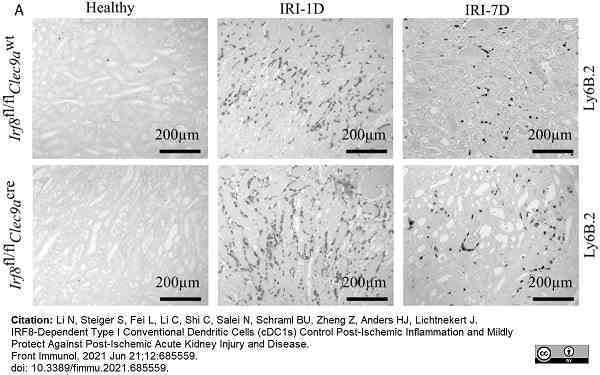
Li, N. et al. (2021) IRF8-Dependent Type I Conventional Dendritic Cells (cDC1s) Control Post-Ischemic Inflammation and Mildly Protect Against Post-Ischemic Acute Kidney Injury and Disease.
Front Immunol. 12: 685559. -
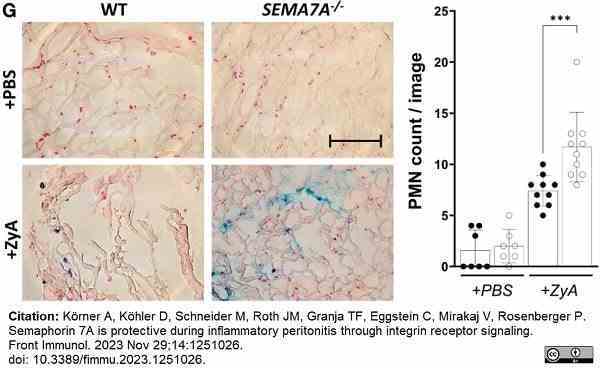
K&oumml;rner, A. et al. (2023) Semaphorin 7A is protective during inflammatory peritonitis through integrin receptor signaling.
Front Immunol. 14: 1251026. -
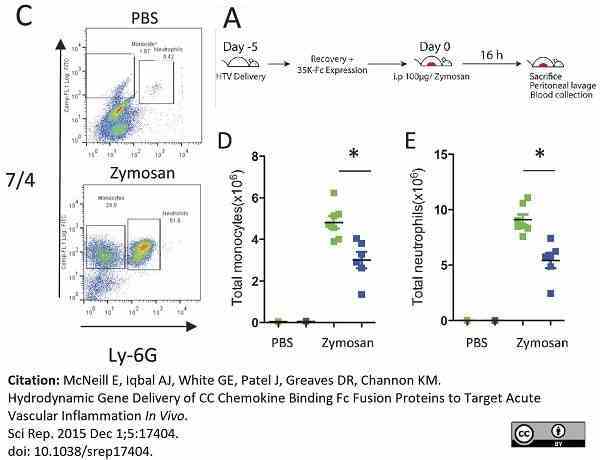
McNeill, E. et al. (2015) Hydrodynamic Gene Delivery of CC Chemokine Binding Fc Fusion Proteins to Target Acute Vascular Inflammation In Vivo.
Sci Rep. 5: 17404. -
Martin, K. et al. (2020) Requirement of Mucosa-Associated Lymphoid Tissue Lymphoma Translocation Protein 1 Protease Activity for Fcγ Receptor-Induced Arthritis, but Not Fcγ Receptor-Mediated Platelet Elimination, in Mice.
Arthritis Rheumatol. 72 (6): 919-30. -
Shannon, A.H. et al. (2020) Single-Photon Emission Computed Tomography Imaging Using Formyl Peptide Receptor 1 Ligand Can Diagnose Aortic Aneurysms in a Mouse Model.
J Surg Res. 251: 239-47. -
Markert, C. et al. (2021) Discovery of LYS006, a Potent and Highly Selective Inhibitor of Leukotriene A(4) Hydrolase.
J Med Chem. 64 (4): 1889-903. -
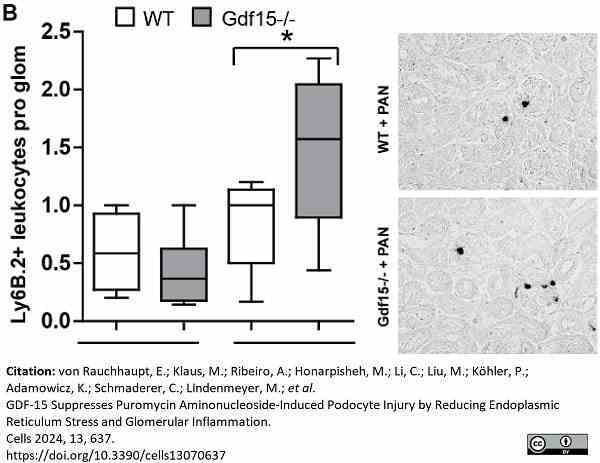
von Rauchhaupt, E. et al. (2024) GDF-15 Suppresses Puromycin Aminonucleoside-Induced Podocyte Injury by Reducing Endoplasmic Reticulum Stress and Glomerular Inflammation
Cells. 13 (7): 637.
- RRID
- AB_322950
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Mouse ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up