CD169 antibody | MOMA-1
Rat anti Mouse CD169
- Product Type
- Monoclonal Antibody
- Clone
- MOMA-1
- Isotype
- IgG2a
- Specificity
- CD169
Filter by Application:
IF F Reset| Rat anti Mouse CD169, clone MOMA-1 recognizes murine CD169, also known as sialoadhesin or Siglec-1. CD169 is a lectin-like receptor expressed by certain populations of macrophages including marginal zone metallophils of the spleen, subcapsular macrophages of lymph nodes and stromal macrophages in bone marrow (Morris et al. 1991). CD169 is a ~185 kDa sialic acid binding receptor containing 17 immunoglobulin-like domains (Crocker et al. 1992). Expression of CD169 can be induced on macrophages in culture by a serum factor and further modulated by cytokine exposure (McWilliam et al. 1992). Rat anti mouse CD169, clone MOMA-1 has been used for the in vivo depletion of specific macrophage populations (Kraal et al. 1988). |
- Target Species
- Mouse
- Species Cross-Reactivity
-
Target Species Cross Reactivity Human Rat - N.B. Antibody reactivity and working conditions may vary between species.
- Product Form
- Purified IgG - liquid
- Preparation
- MCA947G: Purified IgG prepared by affinity chromatography on Protein A from tissue culture supernatant
- MCA947GA: Purified IgG prepared by affinity chromatography on Protein G from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- <0.1% Sodium Azide (NaN3)
- Immunogen
- Stromal (reticular) elements from mouse lymph nodes.
- Approx. Protein Concentrations
- IgG concentration 1.0 mg/ml
- Fusion Partners
- Spleen cells from hyperimmunized mice were fused with cells from the murine SP2/0 myeloma.
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Immunofluorescence | |||
| Immunohistology - Frozen |
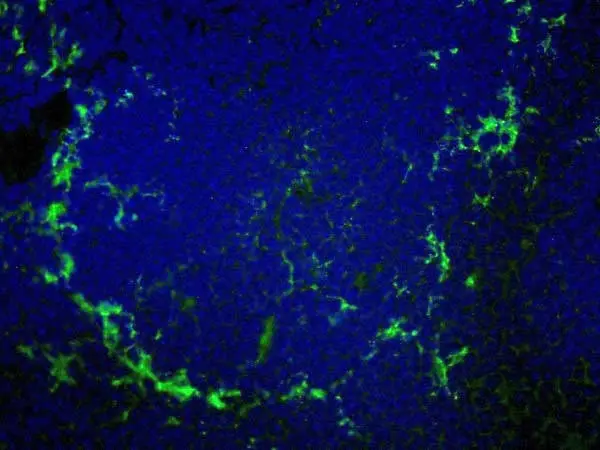
- Histology Positive Control Tissue
- Lymphoid tissue
Source Reference
-
Kraal, G. and Janse, M. (1986) Marginal metallophilic cells of the mouse spleen identified by a monoclonal antibody.
Immunology. 58: 665-9.
References for CD169 antibody
-
Kaisho, T. et al. (2001) IkappaB kinase alpha is essential for mature B cell development and function.
J Exp Med. 193: 417-26. -
Alcamo, E. et al. (2002) Requirement for the NF-κB family member RelA in the development of secondary lymphoid organs.
J Exp Med. 195: 233-44. -
Miosge, L.A. et al. (2002) Analysis of an ethylnitrosourea-generated mouse mutation defines a cell intrinsic role of nuclear factor kappaB2 in regulating circulating B cell numbers.
J Exp Med.196: 1113-9. -
Karlsson, M.C. et al. (2003) Macrophages control the retention and trafficking of B lymphocytes in the splenic marginal zone.
J Exp Med. 198: 333-40. -
Whipple, E.C. et al. (2004) Analyses of the in vivo trafficking of stoichiometric doses of an anti-complement receptor 1/2 monoclonal antibody infused intravenously in mice.
J Immunol. 173 (4): 2297-306. -
Benlagha, K. et al. (2004) Mechanisms governing B cell developmental defects in invariant chain-deficient mice.
J Immunol. 172: 2076-83. -
Girkontaite, I. et al. (2004) The sphingosine-1-phosphate (S1P) lysophospholipid receptor S1P3 regulates MAdCAM-1+ endothelial cells in splenic marginal sinus organization.
J Exp Med. 200 (11): 1491-501. -
Ferguson, A.R. et al. (2004) Marginal zone B cells transport and deposit IgM-containing immune complexes onto follicular dendritic cells.
Int Immunol. 16 (10): 1411-22.
View The Latest Product References
-
Höpken, U.E. et al. (2004) Distinct and overlapping roles of CXCR5 and CCR7 in B-1 cell homing and early immunity against bacterial pathogens.
J Leukoc Biol. 76 (3): 709-18. -
Cariappa, A. et al. (2005) The CD9 tetraspanin is not required for the development of peripheral B cells or for humoral immunity.
J Immunol. 175: 2925-30. -
Acevedo-Suárez, C.A. et al. (2005) Uncoupling of anergy from developmental arrest in anti-insulin B cells supports the development of autoimmune diabetes.
J Immunol. 174 (2): 827-33. -
Rolf, J. et al. (2005) The enlarged population of marginal zone/CD1d(high) B lymphocytes in nonobese diabetic mice maps to diabetes susceptibility region Idd11.
J Immunol. 174: 4821-7. -
Kanayama, N. et al. (2005) Analysis of marginal zone B cell development in the mouse with limited B cell diversity: role of the antigen receptor signals in the recruitment of B cells to the marginal zone.
J Immunol. 174 (3): 1438-45. -
Oetke, C. et al. (2006) The antigen recognized by MOMA-I is sialoadhesin.
Immunol Lett. 106: 96-98. -
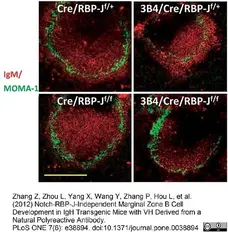
Caton, M.L. et al. (2007) Notch-RBP-J signaling controls the homeostasis of CD8- dendritic cells in the spleen.
J Exp Med. 204 (7): 1653-64. -
Cadman, E.T. et al. (2008) Alterations of splenic architecture in malaria are induced independently of Toll-like receptors 2, 4, and 9 or MyD88 and may affect antibody affinity.
Infect Immun. 76: 3924-31. -
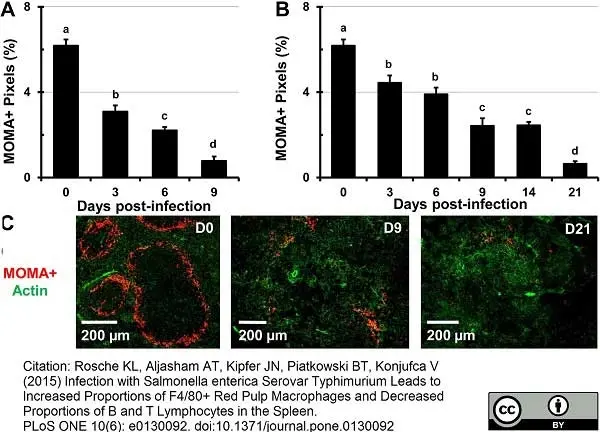
Gangadharan, B. et al. (2008) Murine gamma herpesvirus-induced fibrosis is associated with the development of alternatively activated macrophages.
J Leukoc Biol. 84: 50-8. -
Awasthi, A. et al. (2010) Rap1b facilitates NK cell functions via IQGAP1-mediated signalosomes.
J Exp Med. 207: 1923-38. -
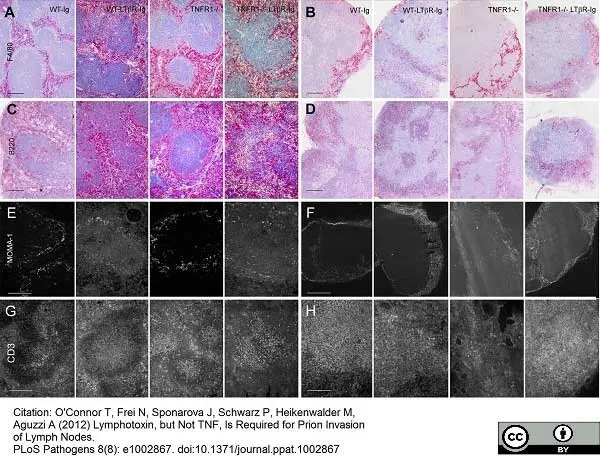
Tumanov, A.V. et al. (2010) Cellular source and molecular form of TNF specify its distinct functions in organization of secondary lymphoid organs.
Blood. 116 (18): 3456-64. -
Mattsson, J. et al. (2011) Complement activation and complement receptors on follicular dendritic cells are critical for the function of a targeted adjuvant.
J Immunol. 187: 3641-52. -
Carnrot, C. et al. (2011) Marginal zone B cells are naturally reactive to collagen type II and are involved in the initiation of the immune response in collagen-induced arthritis.
Cell Mol Immunol. 8 (4): 296-304. -
Rehm, A. et al. (2011) Cooperative function of CCR7 and lymphotoxin in the formation of a lymphoma-permissive niche within murine secondary lymphoid organs.
Blood. 118 (4): 1020-33. -
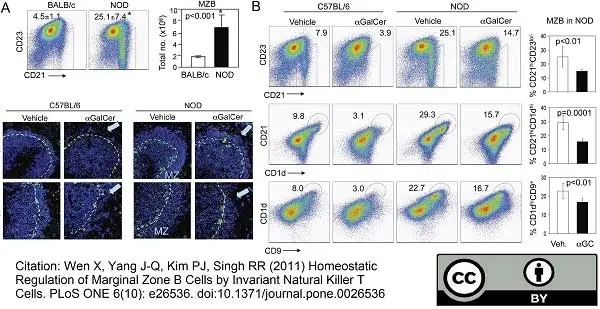
Birjandi, S.Z. et al. (2011) Alterations in marginal zone macrophages and marginal zone B cells in old mice.
J Immunol. 186: 3441-51. -
Bhattacharyya, S. et al. (2011) NFATc1 affects mouse splenic B cell function by controlling the calcineurin-NFAT signaling network.
J Exp Med. 208 (4): 823-39. -
Muppidi, J.R. et al. (2011) Cannabinoid receptor 2 positions and retains marginal zone B cells within the splenic marginal zone.
J Exp Med. 208 (10): 1941-8. -
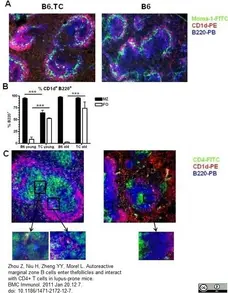
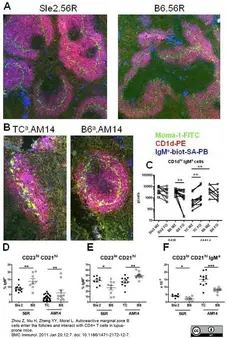
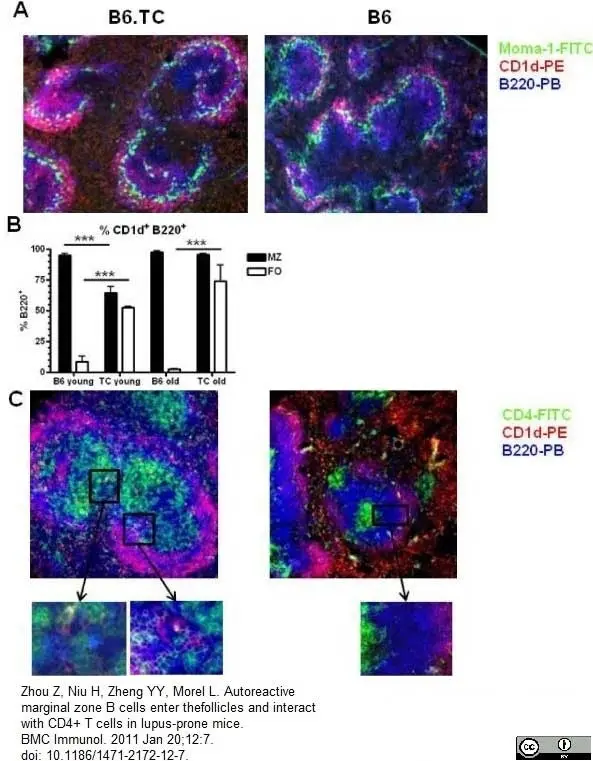
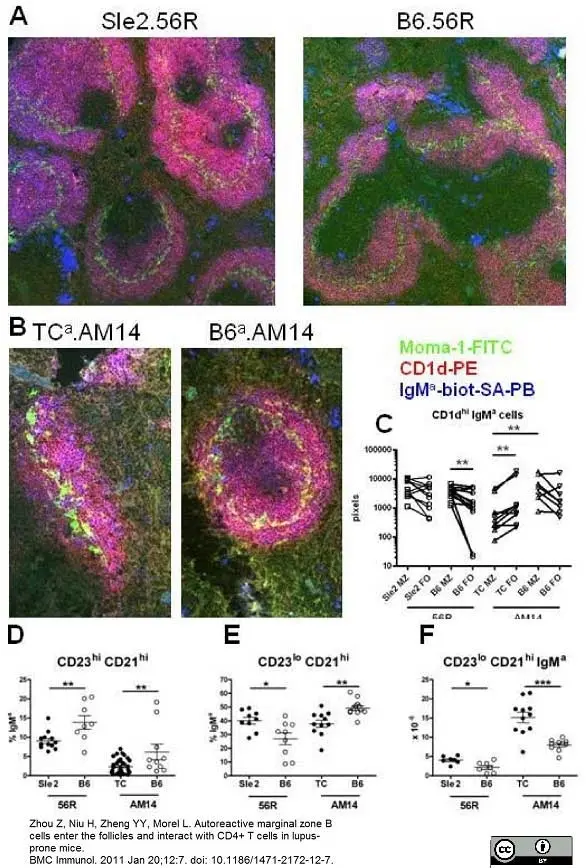
Zhou, Z. et al. (2011) Autoreactive marginal zone B cells enter the follicles and interact with CD4+ T cells in lupus-prone mice.
BMC Immunol. 12:7. -
Jang, I.K. et al. (2011) Growth-factor receptor-bound protein-2 (Grb2) signaling in B cells controls lymphoid follicle organization and germinal center reaction.
Proc Natl Acad Sci U S A. 108: 7926-31. -
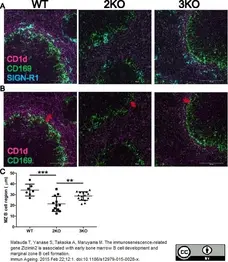
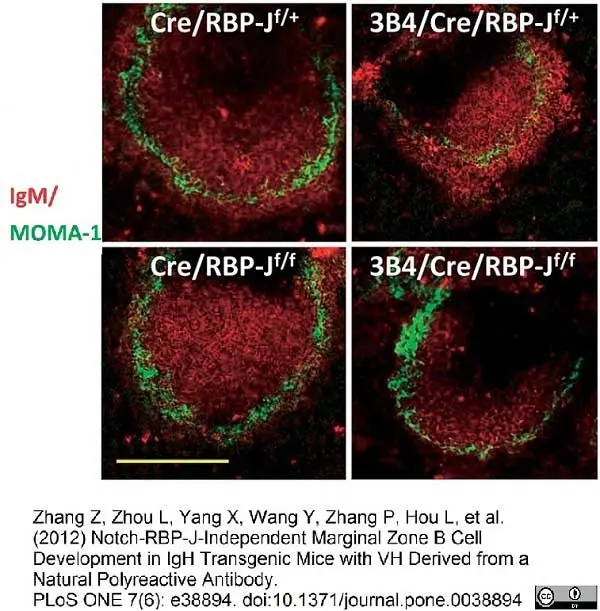
Zhang, Z. et al. (2012) Notch-RBP-J-Independent Marginal Zone B Cell Development in IgH Transgenic Mice with V(H) Derived from a Natural Polyreactive Antibody.
PLoS One. 7: e38894. -
Flores, M. et al. (2015) FcγRIIB prevents inflammatory type I IFN production from plasmacytoid dendritic cells during a viral memory response.
J Immunol. 194 (9): 4240-50. -
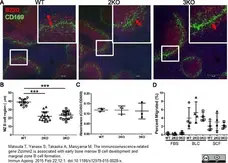
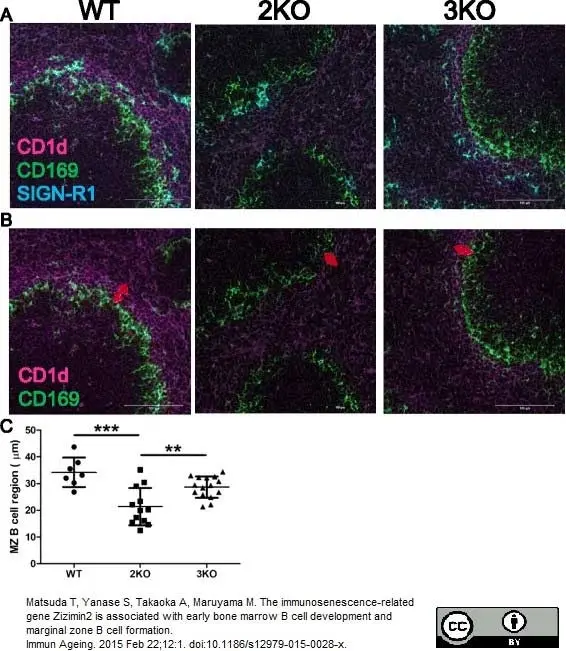
Matsuda T et al. (2015) The immunosenescence-related gene Zizimin2 is associated with early bone marrow B cell development and marginal zone B cell formation.
Immun Ageing. 12: 1. -
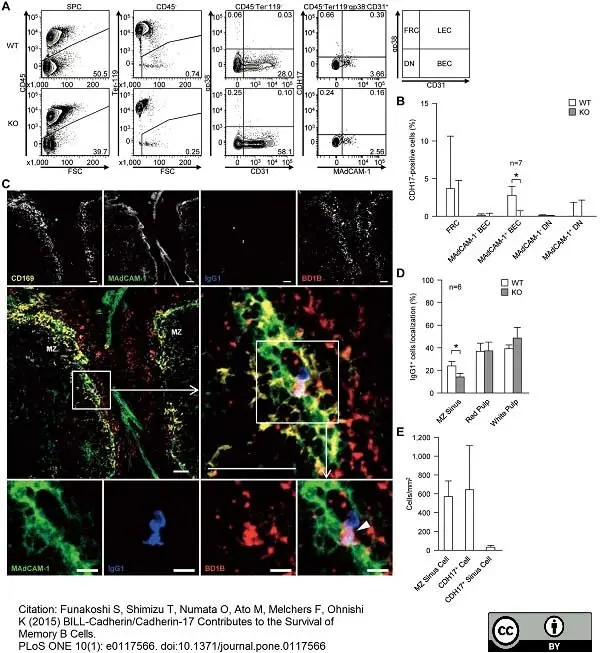
Funakoshi, S. et al. (2015) BILL-cadherin/cadherin-17 contributes to the survival of memory B cells.
PLoS One. 10 (1): e0117566. -
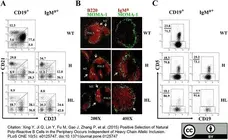
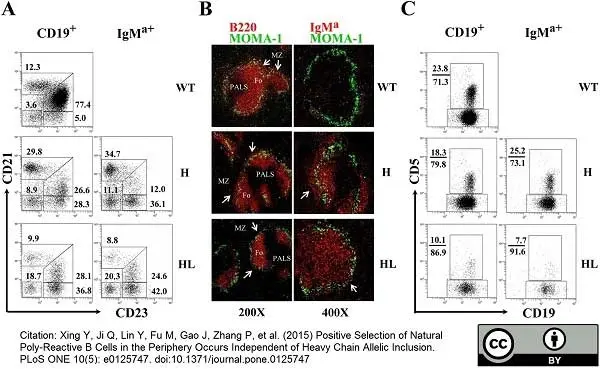
Xing Y et al. (2015) Positive Selection of Natural Poly-Reactive B Cells in the Periphery Occurs Independent of Heavy Chain Allelic Inclusion.
PLoS One. 10 (5): e0125747. -
Bradford, B.M. et al. (2016) Prion pathogenesis is unaltered following down-regulation of SIGN-R1.
Virology. 497: 337-345. -
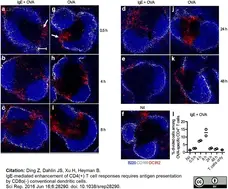
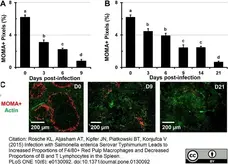
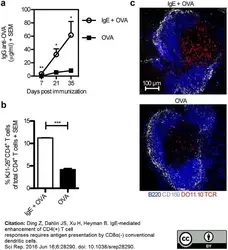
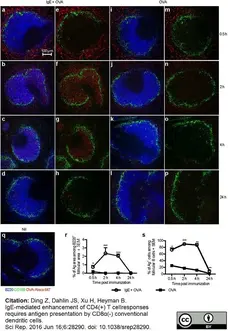
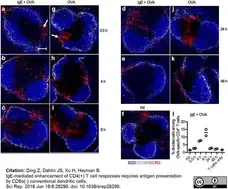
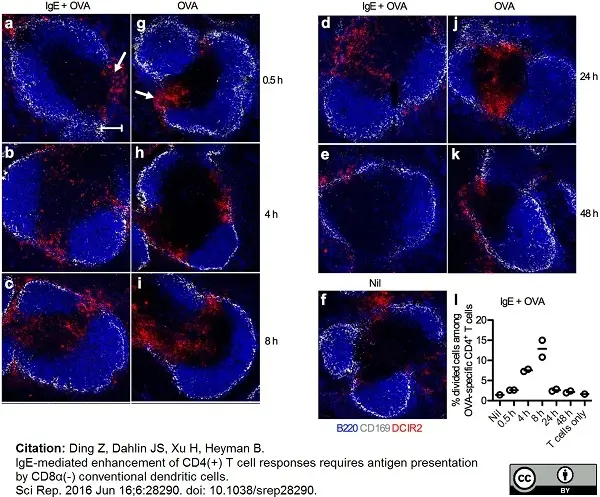
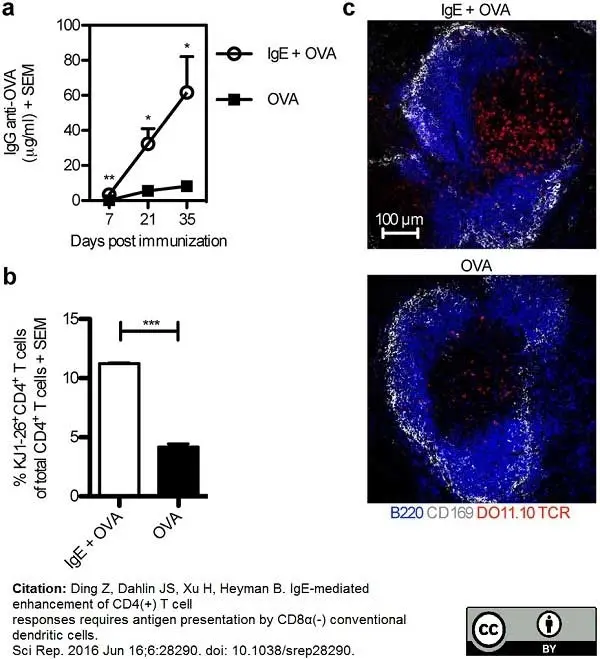
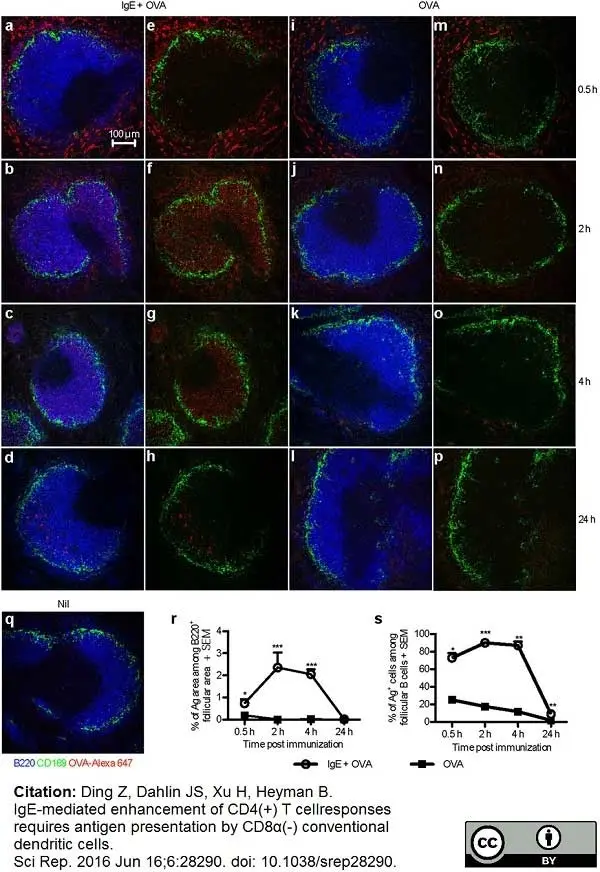
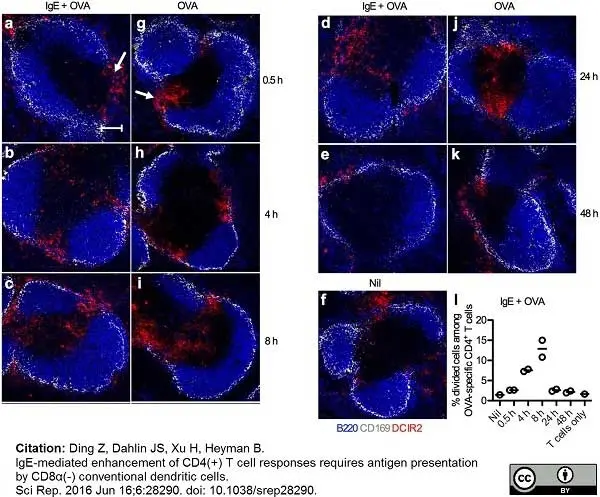
Ding, Z. et al. (2016) IgE-mediated enhancement of CD4(+) T cell responses requires antigen presentation by CD8α(-) conventional dendritic cells.
Sci Rep. 6: 28290. -
Oh, D.S. et al. (2017) Transient Depletion of CD169+ Cells Contributes to Impaired Early Protection and Effector CD8+ T Cell Recruitment against Mucosal Respiratory Syncytial Virus Infection.
Front Immunol. 8: 819. -
Bogie, J.F. et al. (2018) CD169 is a marker for highly pathogenic phagocytes in multiple sclerosis.
Mult Scler. 24 (3): 290-300. -
Tsai, C.Y. et al. (2018) Bystander inhibition of humoral immune responses by Epstein-Barr virus LMP1.
Int Immunol. 30 (12): 579-90. -
Dekker, J.D. et al. (2019) Loss of the FOXP1 Transcription Factor Leads to Deregulation of B Lymphocyte Development and Function at Multiple Stages.
Immunohorizons. 3 (10): 447-62. -
Vanderkerken, M. et al. (2020) TAO-kinase 3 governs the terminal differentiation of NOTCH2-dependent splenic conventional dendritic cells.
Proc Natl Acad Sci U S A. 117 (49): 31331-31342. -
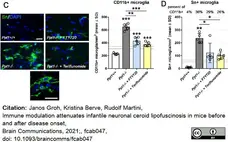
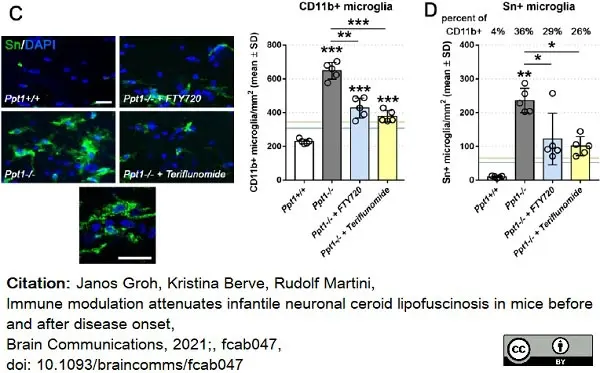
Groh, J. et al. (2021) Immune modulation attenuates infantile neuronal ceroid lipofuscinosis in mice before and after disease onset.
Brain Commun. 3 (2): fcab047. -
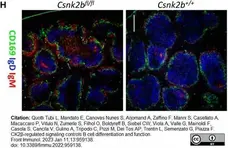
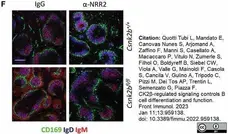
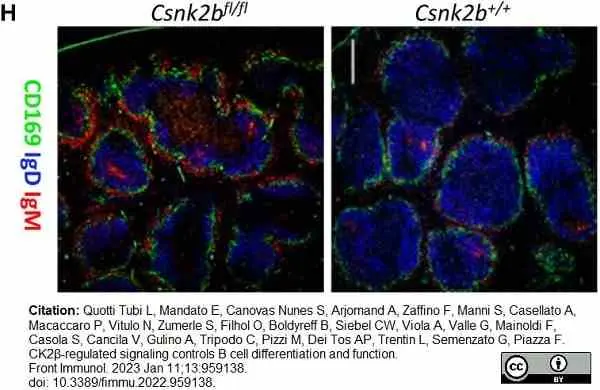
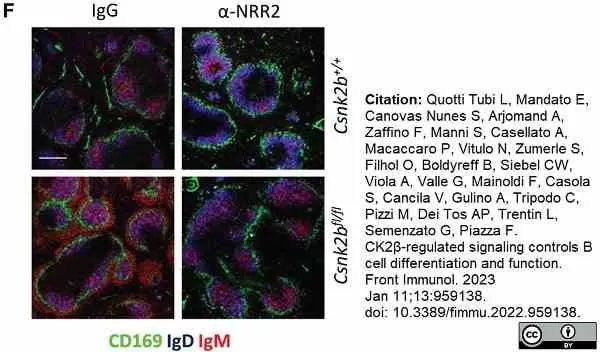
Quotti Tubi, L. et al. (2022) CK2β-regulated signaling controls B cell differentiation and function.
Front Immunol. 13: 959138. -
Ghilas, S. et al. (2021) Natural killer cells and dendritic epidermal γδ T cells orchestrate type 1 conventional DC spatiotemporal repositioning toward CD8+ T cells
iScience. 24 (9): 103059. -
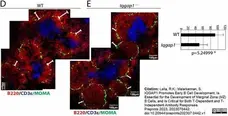
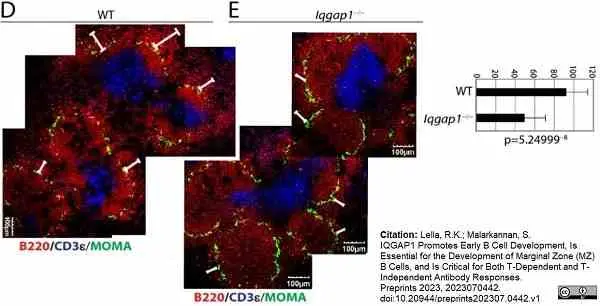
Lella, R.K. & Malarkannan, S. (2023) IQGAP1 Promotes Early B Cell Development, Is Essential for the Development of Marginal Zone (MZ) B Cells, and Is Critical for Both T-Dependent and T-Independent Antibody Responses.
Preprints 2023, 2023070442. -
Glaubitz, J. et al. (2020) Experimental pancreatitis is characterized by rapid T cell activation, Th2 differentiation that parallels disease severity, and improvement after CD4(+) T cell depletion.
Pancreatology. 20 (8): 1637-47. -
Van, K.J.D. et al. (2018) Dendritic Cell Targeting mRNA Lipopolyplexes Combine Strong Antitumor T-Cell Immunity with Improved Inflammatory Safety.
ACS Nano. 12 (10): 9815-29. -
Ozawa, M. et al. (2022) Micro- and Macro-Anatomical Frameworks of Lymph Nodes Indispensable for the Lymphatic System Filtering Function.
Front Cell Dev Biol. 10: 902601.
- Synonyms
- Sialoadhesin
- UniProt
- Q62230
- Entrez Gene
- Siglec1
- GO Terms
- GO:0005886 plasma membrane
- GO:0005515 protein binding
- GO:0007155 cell adhesion
- GO:0016021 integral to membrane
- GO:0005576 extracellular region
- GO:0005529 sugar binding
- GO:0006897 endocytosis
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Mouse ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up