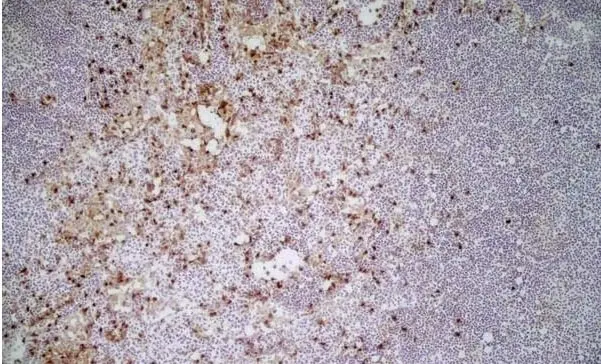
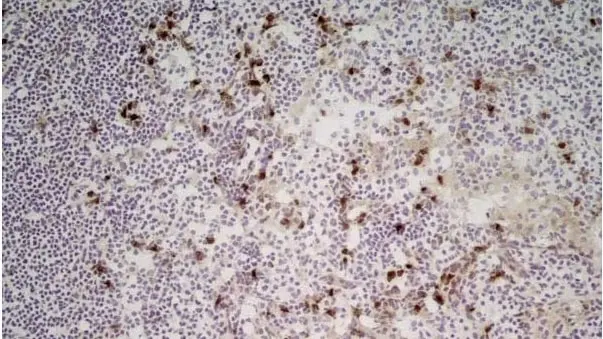
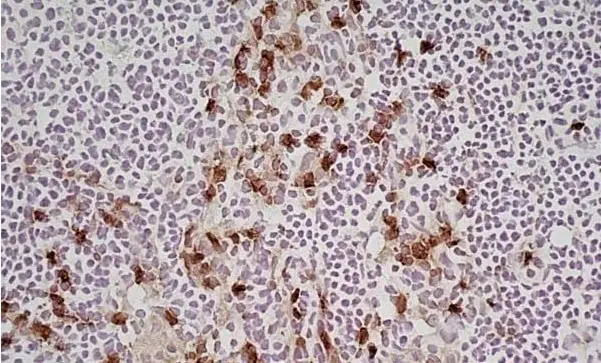
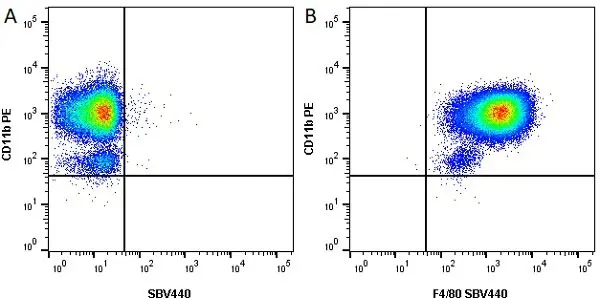
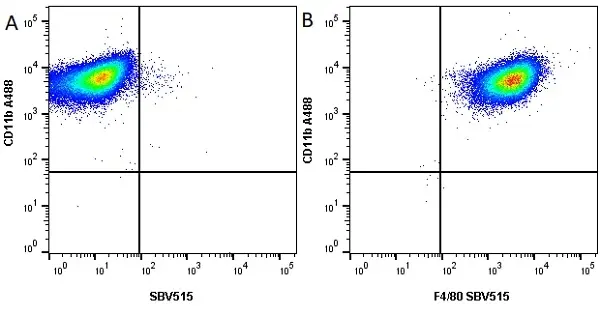
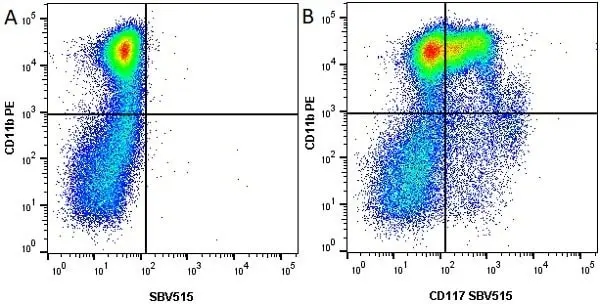
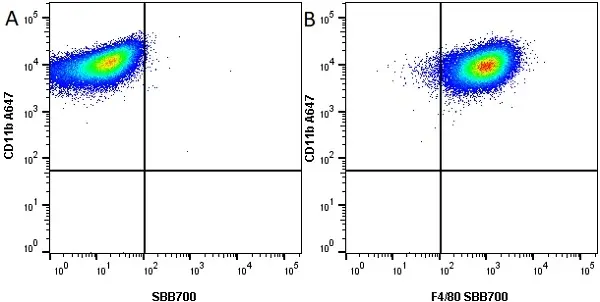
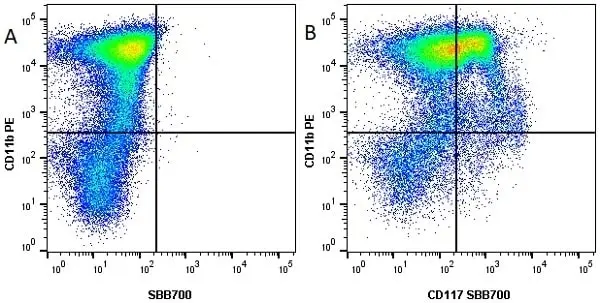
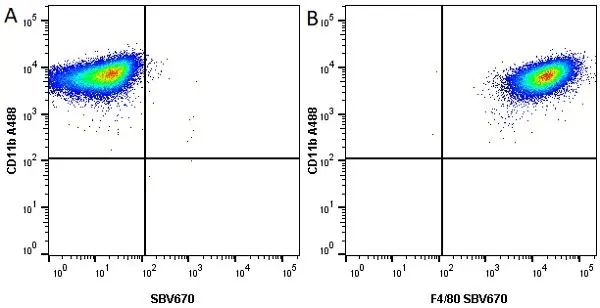
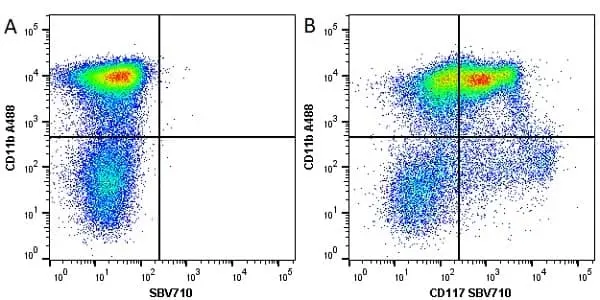
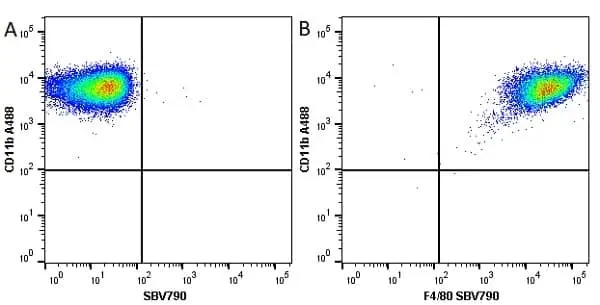
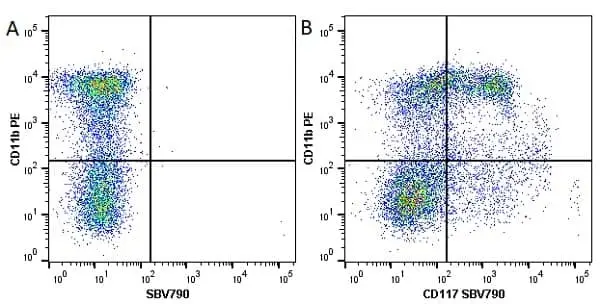
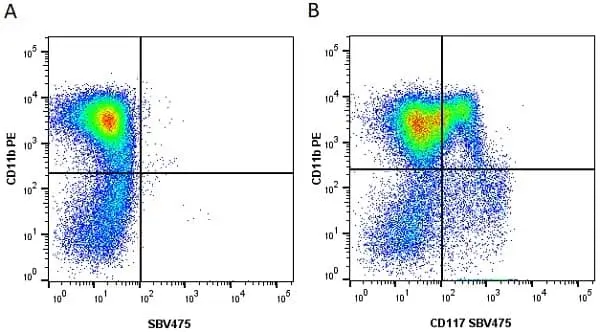
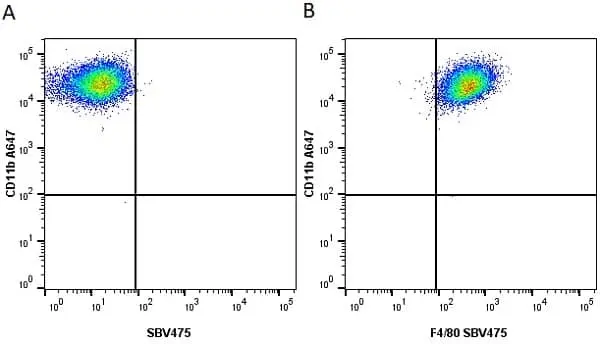
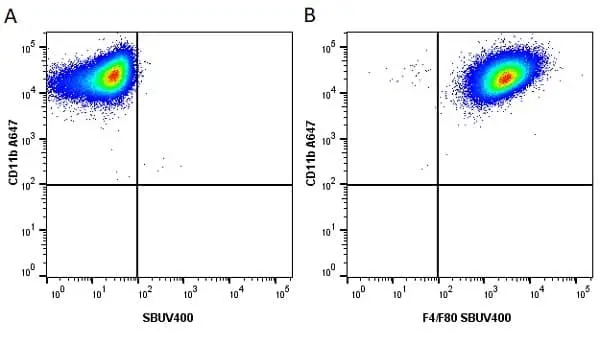
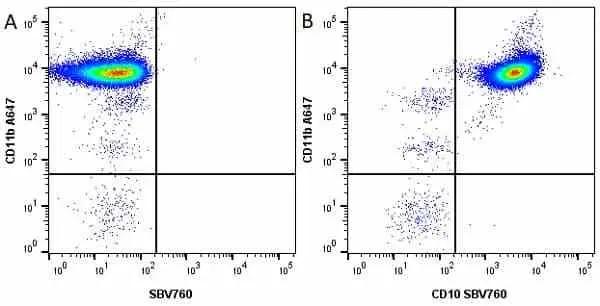
CD11b antibody | M1/70.15
Rat anti Mouse CD11b
- Product Type
- Monoclonal Antibody
- Clone
- M1/70.15
- Isotype
- IgG2b
- Specificity
- CD11b
Filter by Application:
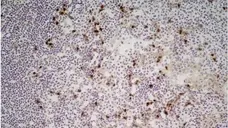
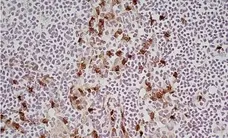
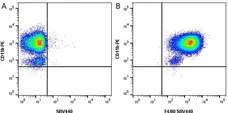
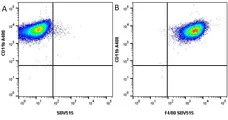
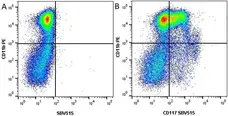
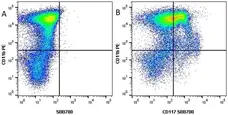
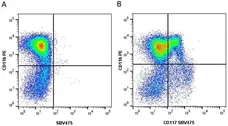
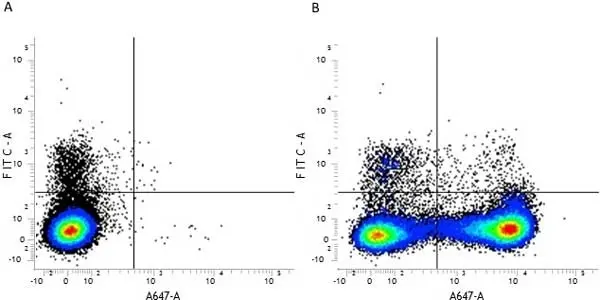
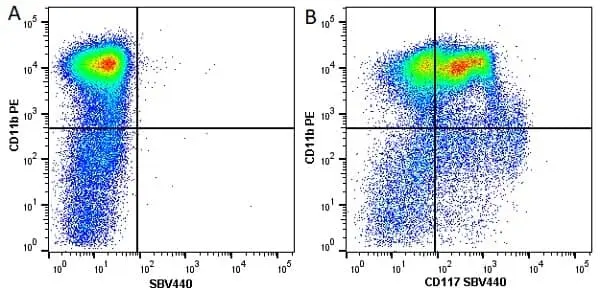
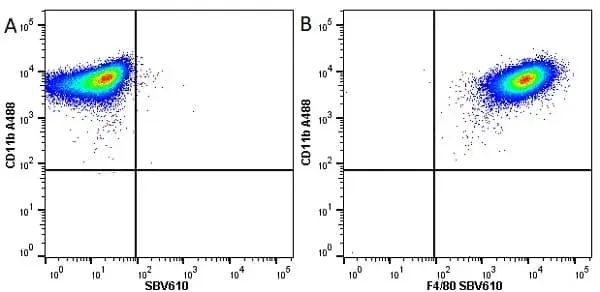
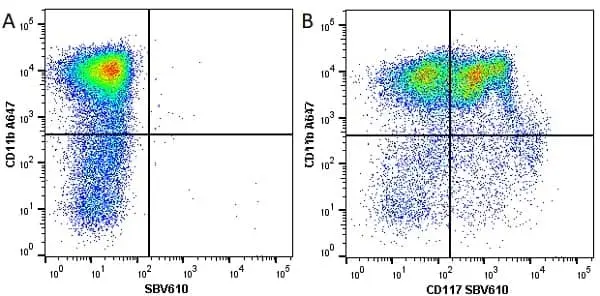
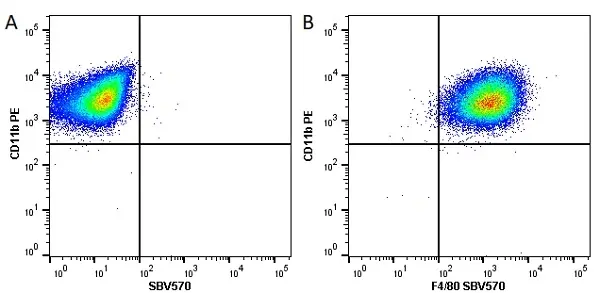
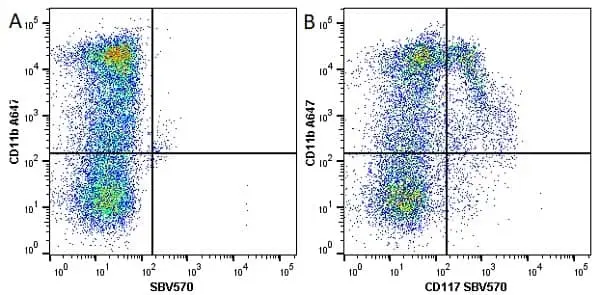
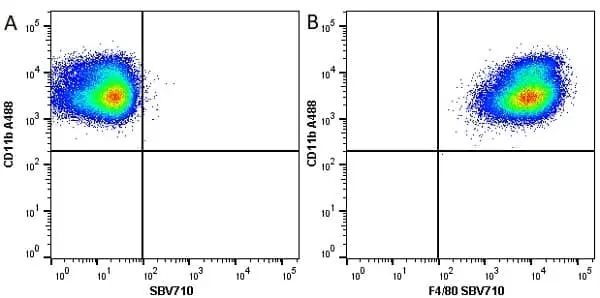
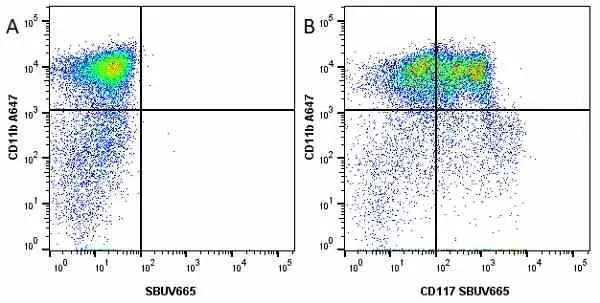
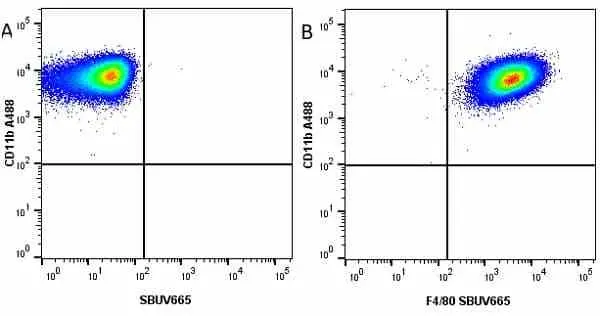
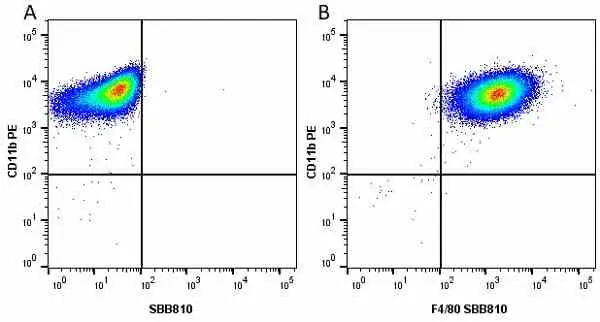
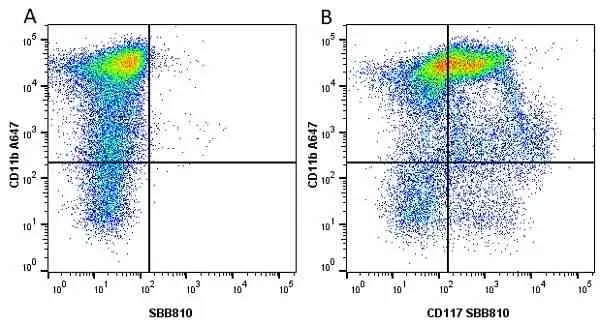
F IF C P Reset| Rat anti Mouse CD11b antibody, clone M1/70.15 recognizes the murine CD11b cell surface antigen also known as the alpha M integrin chain or MAC-1, a differentiation antigen expressed by granulocytes, monocytes, NK cells and tissue macrophages. The expression of CD11b increases during monocyte maturation and expression levels vary on tissue macrophages. Peritoneal macrophages are reported to express higher levels of CD11b than splenic macrophages. Rat anti Mouse CD11b antibody, clone M1/70.15 has been reported to block iC3b binding to its receptor (Beller et al. 1982). Rat anti Mouse CD11b antibody, clone M1/70.15 has been reported to as being suitable for use on PLP fixed paraffin embedded tissue but has not been tested for use on formalin fixed tissue (Whiteland et al. 1995). This product is routinely tested in flow cytometry on mouse peritoneal macrophages. |
- Target Species
- Mouse
- Species Cross-Reactivity
-
Target Species Cross Reactivity Human Rabbit - N.B. Antibody reactivity and working conditions may vary between species.
- Product Form
- Purified IgG - liquid
- Preparation
- Purified IgG prepared by affinity chromatography on Protein G from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
0.09% Sodium Azide - Carrier Free
- Yes
- Immunogen
- T cell enriched splenocytes from B10 mice.
- Approx. Protein Concentrations
- IgG concentration 1.0 mg/ml
- Fusion Partners
- Spleen cells from immunized DA rats were fused with cells of the NS1/1.Ag4.1 mouse myeloma cell line.
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Flow Cytometry | 1/50 | 1/50 Pack Size: 0.25 mg 1/200 Pack Size: 0.1 mg |
|
| Immunofluorescence | |||
| Immunohistology - Frozen | |||
| Immunohistology - Paraffin 1 | |||
| Immunoprecipitation |
- 1PLP fixation is recommended for optimal results, see Whiteland et al. for details
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Antigen Retrieval Buffer, pH8.0 | BUF025A | P | 500 ml | Log in | |||
| List Price | Your Price | ||||||
| Log in | |||||||
| Description | Antigen Retrieval Buffer, pH8.0 | ||||||
Source Reference
-
Springer, T. et al. (1978) Monoclonal xenogeneic antibodies to murine cell surface antigens: identification of novel leukocyte differentiation antigens.
Eur J Immunol. 8 (8): 539-51.
Antibody Characterization Reference
-
Springer, T. et al. (1979) Mac-1: a macrophage differentiation antigen identified by monoclonal antibody.
Eur J Immunol. 9 (4): 301-6.
References for CD11b antibody
-
Beller, D.I. et al. (1982) Anti-Mac-1 selectively inhibits the mouse and human type three complement receptor.
J Exp Med. 156 (4): 1000-9. -
Whiteland, J.L. et al. (1995) Immunohistochemical detection of T-cell subsets and other leukocytes in paraffin-embedded rat and mouse tissues with monoclonal antibodies.
J Histochem Cytochem. 43 (3): 313-20. -
Welt, F.G. et al. (2000) Neutrophil, not macrophage, infiltration precedes neointimal thickening in balloon-injured arteries.
Arterioscler Thromb Vasc Biol. 20 (12): 2553-8. -
Kapturczak, M.H. et al (2004) Heme oxygenase-1 modulates early inflammatory responses: evidence from the heme oxygenase-1-deficient mouse.
Am J Pathol. 165: 1045-53. -
Patel, P.C. and Harrison, R.E. (2008) Membrane ruffles capture C3bi-opsonized particles in activated macrophages.
Mol Biol Cell. 19: 4628-39. -
Huang, Q.Q. et al (2008) Role of H2-calponin in regulating macrophage motility and phagocytosis.
J Biol Chem. 283: 25887-99. -
Brochard, V. et al (2009) Infiltration of CD4+ lymphocytes into the brain contributes to neurodegeneration in a mouse model of Parkinson disease.
J Clin Invest. 119: 182-92. -
Hudcovic, T. et al (2009) Monocolonization with Bacteroides ovatus protects immunodeficient SCID mice from mortality in chronic intestinal inflammation caused by long-lasting dextran sodium sulfate treatment.
Physiol Res. 58: 101-10.
View The Latest Product References
-
Kroner, A. et al (2010) Ectopic T-cell specificity and absence of perforin and granzyme B alleviate neural damage in oligodendrocyte mutant mice.
Am J Pathol. 176: 549-55. -
Kanu, N. et al. (2010) The ATM cofactor ATMIN protects against oxidative stress and accumulation of DNA damage in the aging brain.
J Biol Chem. 285: 38534-42. -
Terrando, N. et al. (2010) The impact of IL-1 modulation on the development of lipopolysaccharide-induced cognitive dysfunction.
Crit Care. 14 (3): R88. -
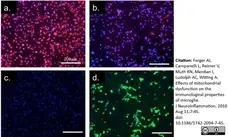
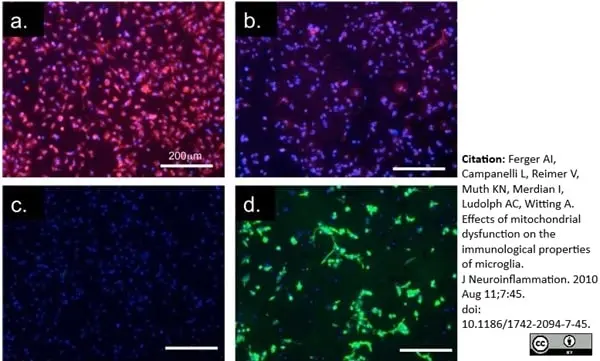
Ferger, A.I. et al (2010) Effects of mitochondrial dysfunction on the immunological properties of microglia.
J Neuroinflammation. 7: 45. -
Gales, A. et al (2010) PPARgamma controls dectin-1 expression required for host antifungal defense against Candida albicans.
PLoS Pathog. 6 : e1000714. -
Ghasemlou, N. et al. (2010) Mitogen-activated protein kinase-activated protein kinase 2 (MK2) contributes to secondary damage after spinal cord injury.
J Neurosci. 30: 13750-9. -
Lefevre, L. et al. (2010) PPARγ ligands switched high fat diet-induced macrophage M2b polarization toward M2a thereby improving intestinal Candida elimination.
PLoS One. 5: e12828. -
Samanta, J. et al. (2010) Noggin protects against ischemic brain injury in rodents.
Stroke. 41: 357-62. -
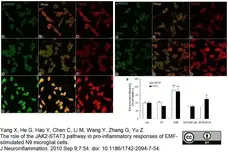
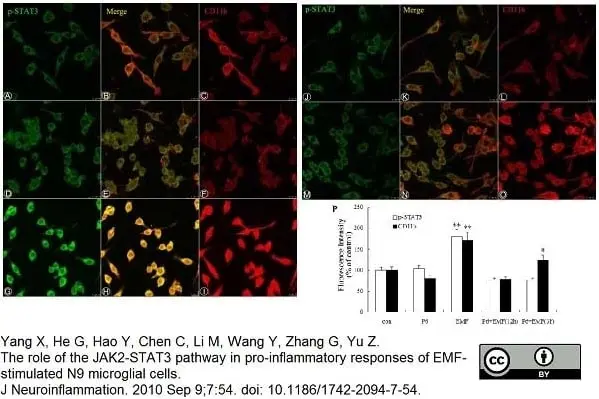
Yang, X. et al (2010) The role of the JAK2-STAT3 pathway in pro-inflammatory responses of EMF-stimulated N9 microglial cells.
J Neuroinflammation. 7: 54. -
Zhang, H. et al. (2010) A p38 mitogen-activated protein kinase-dependent mechanism of disinhibition in spinal synaptic transmission induced by tumor necrosis factor-alpha.
J Neurosci. 30: 12844-55. -
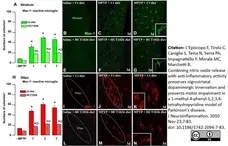
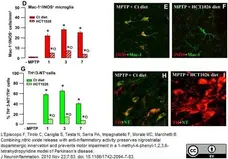
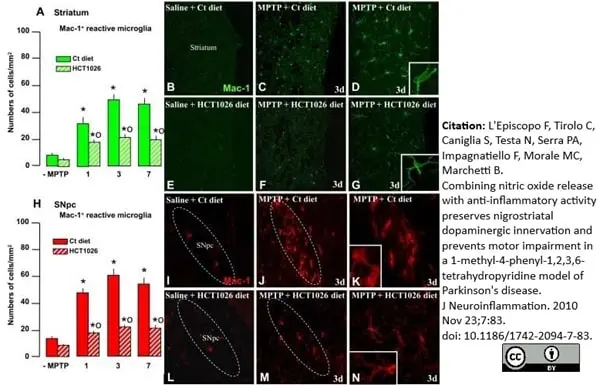
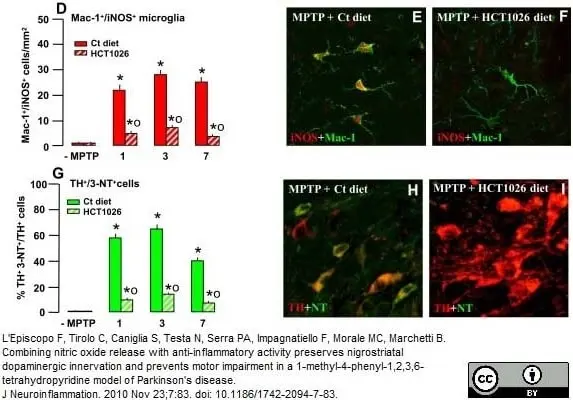
L'Episcopo, F. et al. (2010) Combining nitric oxide release with anti-inflammatory activity preserves nigrostriatal dopaminergic innervation and prevents motor impairment in a 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine model of Parkinson's disease.
J Neuroinflammation. 7: 83. -
Lebson, L. et al. (2010) Trafficking CD11b-positive blood cells deliver therapeutic genes to the brain of amyloid-depositing transgenic mice.
J Neurosci. 30: 9651-8. -
Kondo, Y. et al. (2011) Macrophages counteract demyelination in a mouse model of globoid cell leukodystrophy.
J Neurosci. 31: 3610-24. -
Geier, H. and Celli, J. (2011) Phagocytic Receptors Dictate Phagosomal Escape and Intracellular Proliferation of Francisella tularensis.
Infect Immun. 79: 2204-14. -
Redensek, A. et al. (2011) Expression and detrimental role of hematopoietic prostaglandin D synthase in spinal cord contusion injury.
Glia. 59: 603-14. -
Cunningham, K. et al. (2011) Conditional deletion of Ccm2 causes hemorrhage in the adult brain: a mouse model of human cerebral cavernous malformations.
Hum Mol Genet. 20: 3198-206. -
Fernández-Suárez,D. (2014) The monoacylglycerol lipase inhibitor JZL184 is neuroprotective and alters glial cell phenotype in the chronic MPTP mouse model
Neurobiol Aging. 35: 2603-16. -
Certo, M. et al. (2015) Activation of RXR/PPARγ underlies neuroprotection by bexarotene in ischemic stroke.
Pharmacol Res. 102: 298-307. -
Jones, R.S. et al. (2015) Inhibition of JAK2 attenuates the increase in inflammatory markers in microglia from APP/PS1 mice.
Neurobiol Aging. 36 (10): 2716-24. -
Nishikawa, K. et al. (2015) Resveratrol increases CD68⁺ Kupffer cells colocalized with adipose differentiation-related protein and ameliorates high-fat-diet-induced fatty liver in mice.
Mol Nutr Food Res. 59 (6): 1155-70. -
Khan, A.A. et al. (2015) Therapeutic Immunization with a Mixture of Herpes Simplex Virus Type 1 Glycoprotein D Derived "Asymptomatic" Human CD8+ T-cell Epitopes Decreases Spontaneous Ocular Shedding in Latently Infected HLA Transgenic Rabbits: Association with Low Frequency of Local PD-1+TIM-3+CD8+ Exhausted T Cells.
J Virol. pii: JVI.00788-15. -
Kim, B.W. et al. (2015) α-Asarone attenuates microglia-mediated neuroinflammation by inhibiting NF kappa B activation and mitigates MPTP-induced behavioral deficits in a mouse model of Parkinson's disease.
Neuropharmacology. 97: 46-57. -
Bains, M. & Roberts, J.L. (2016) Estrogen protects against dopamine neuron toxicity in primary mesencephalic cultures through an indirect P13K/Akt mediated astrocyte pathway.
Neurosci Lett. 610: 79-85. -
Amantea, D. et al. (2016) Azithromycin protects mice against ischemic stroke injury by promoting macrophage transition towards M2 phenotype.
Exp Neurol. 275 Pt 1: 116-25. -
Macrez, R. et al. (2016) Neuroendothelial NMDA receptors as therapeutic targets in experimental autoimmune encephalomyelitis.
Brain. 139 (Pt 9): 2406-19. -
Chen, Z.Z. et al. (2016) Memantine mediates neuroprotection via regulating neurovascular unit in a mouse model of focal cerebral ischemia.
Life Sci. 150: 8-14. -
Rich, M.C. et al. (2016) Site-targeted complement inhibition by a complement receptor 2-conjugated inhibitor (mTT30) ameliorates post-injury neuropathology in mouse brains.
Neurosci Lett. 617: 188-94. -
Werneburg, S. et al. (2016) Polysialylation and lipopolysaccharide-induced shedding of E-selectin ligand-1 and neuropilin-2 by microglia and THP-1 macrophages.
Glia. 64 (8): 1314-30. -
Amantea, D. et al. (2016) Neuroprotective Properties of a Macrolide Antibiotic in a Mouse Model of Middle Cerebral Artery Occlusion: Characterization of the Immunomodulatory Effects and Validation of the Efficacy of Intravenous Administration.
Assay Drug Dev Technol. 14 (5): 298-307. -
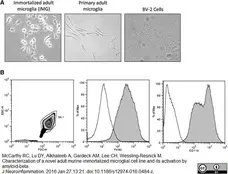
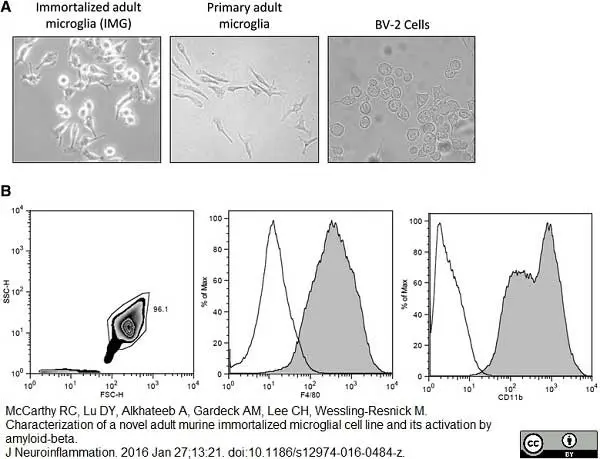
McCarthy, R.C. et al. (2016) Characterization of a novel adult murine immortalized microglial cell line and its activation by amyloid-beta.
J Neuroinflammation. 13: 21. -
Jiang, H. et al. (2017) Dense Intra-adipose Sympathetic Arborizations Are Essential for Cold-Induced Beiging of Mouse White Adipose Tissue.
Cell Metab. 26 (4): 686-692.e3. -
Zhang, J.C. et al. (2017) Prophylactic effects of sulforaphane on depression-like behavior and dendritic changes in mice after inflammation.
J Nutr Biochem. 39: 134-44. -
Petković, F. et al. (2017) Reduced cuprizone-induced cerebellar demyelination in mice with astrocyte-targeted production of IL-6 is associated with chronically activated, but less responsive microglia.
J Neuroimmunol. 310: 97-102. -
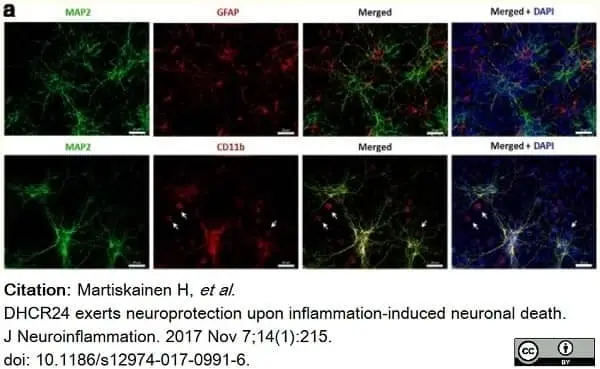
Martiskainen, H. et al. (2017) DHCR24 exerts neuroprotection upon inflammation-induced neuronal death.
J Neuroinflammation. 14 (1): 215. -
Olesen, M. N. et al. (2018) CD4 T cells react to local increase of α-synuclein in a pathology-associated variant-dependent manner and modify brain microglia in absence of brain pathology
Heliyon. 4 (1): e00513. -
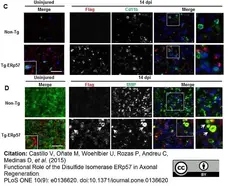
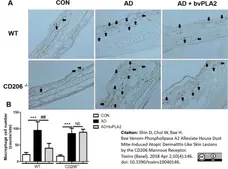
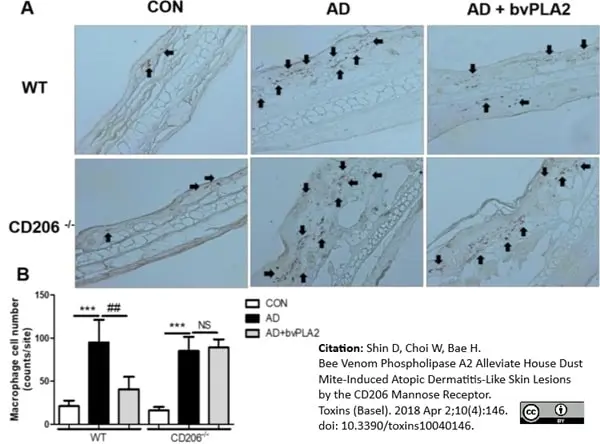
Shin, D. et al. (2018) Bee Venom Phospholipase A2 Alleviate House Dust Mite-Induced Atopic Dermatitis-Like Skin Lesions by the CD206 Mannose Receptor.
Toxins (Basel). 10 (4): 146. -
Dos-Santos-Pereira, M. et al. (2018) Microglial glutamate release evoked by α-synuclein aggregates is prevented by dopamine.
Glia. 66 (11): 2353-2365. -
Price, B.R. et al. (2020) Therapeutic Trem2 activation ameliorates amyloid-beta deposition and improves cognition in the 5XFAD model of amyloid deposition.
J Neuroinflammation. 17 (1): 238. -
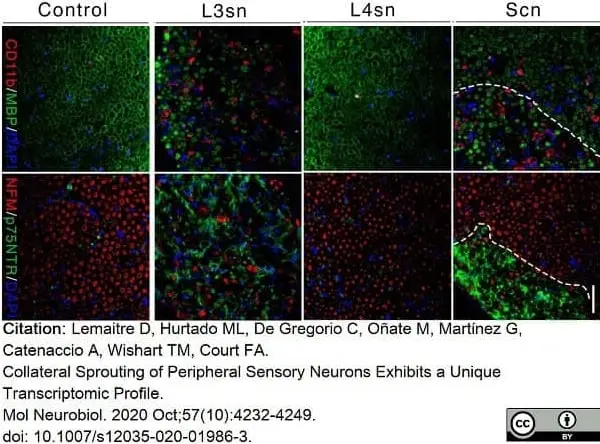
Lemaitre, D. et al. (2020) Collateral Sprouting of Peripheral Sensory Neurons Exhibits a Unique Transcriptomic Profile.
Mol Neurobiol. 57 (10): 4232-49. -
Lim, S.L. et al. (2020) Genetic Ablation of Hematopoietic Cell Kinase Accelerates Alzheimer's Disease-Like Neuropathology in Tg2576 Mice.
Mol Neurobiol. 57 (5): 2447-60. -
Paasila, P.J. et al. (2021) Ground state depletion microscopy as a tool for studying microglia-synapse interactions.
J Neurosci Res. 99 (6): 1515-32. -
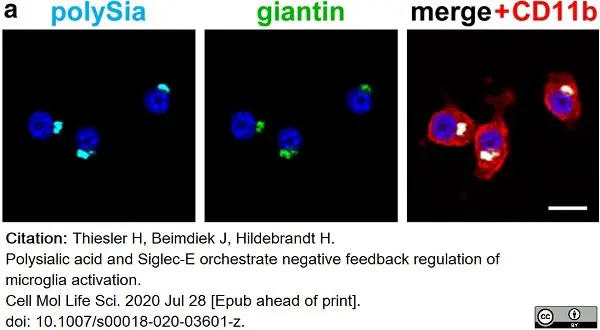
Thiesler, H. et al. (2021) Polysialic acid and Siglec-E orchestrate negative feedback regulation of microglia activation.
Cell Mol Life Sci. 78 (4): 1637-53. -
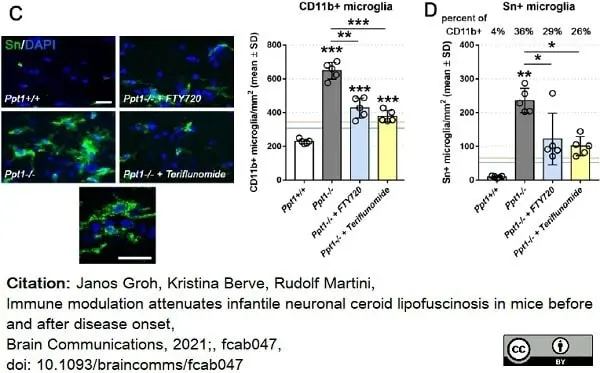
Groh, J. et al. (2021) Immune modulation attenuates infantile neuronal ceroid lipofuscinosis in mice before and after disease onset.
Brain Commun. 3 (2): fcab047. -
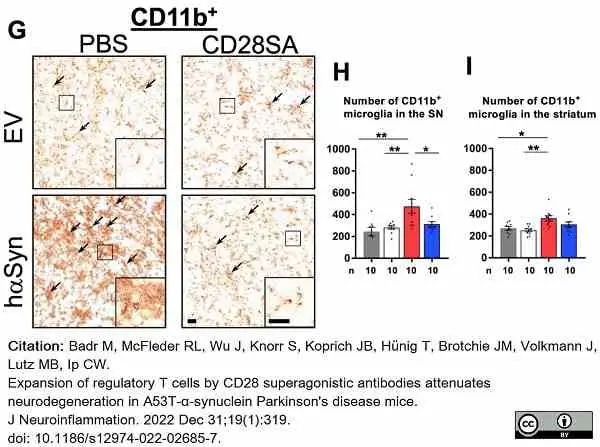
Badr, M. et al. (2022) Expansion of regulatory T cells by CD28 superagonistic antibodies attenuates neurodegeneration in A53T-α-synuclein Parkinson's disease mice.
J Neuroinflammation. 19 (1): 319. -
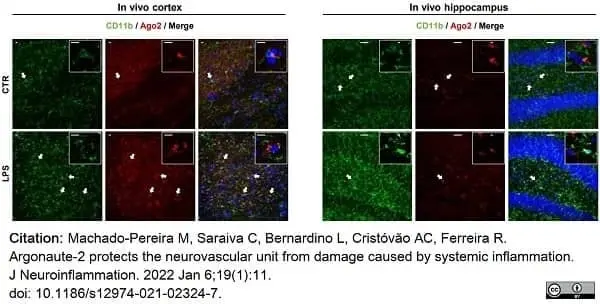
Machado-Pereira, M. et al. (2022) Argonaute-2 protects the neurovascular unit from damage caused by systemic inflammation.
J Neuroinflammation. 19 (1): 11. -
Karikari, A.A. et al. (2022) Neurodegeneration by α-synuclein-specific T cells in AAV-A53T-α-synuclein Parkinson's disease mice.
Brain Behav Immun. 101: 194-210. -
Zhao, Y. et al. (2022) Using Extracellular Vesicles Released by GDNF-Transfected Macrophages for Therapy of Parkinson Disease.
Cells. 11 (12): 1933. -
Ji, N. et al. (2022) VSIG4 Attenuates NLRP3 and Ameliorates Neuroinflammation via JAK2-STAT3-A20 Pathway after Intracerebral Hemorrhage in Mice.
Neurotox Res. 40 (1): 78-88. -
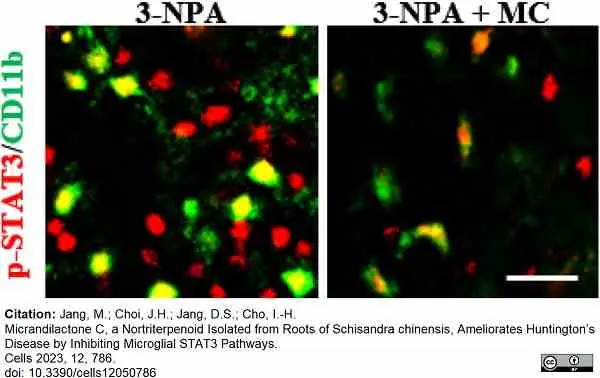
Jang, M. et al. (2023) Micrandilactone C, a Nortriterpenoid Isolated from Roots of Schisandra chinensis, Ameliorates Huntington’s Disease by Inhibiting Microglial STAT3 Pathways
Cells. 12 (5): 786. -
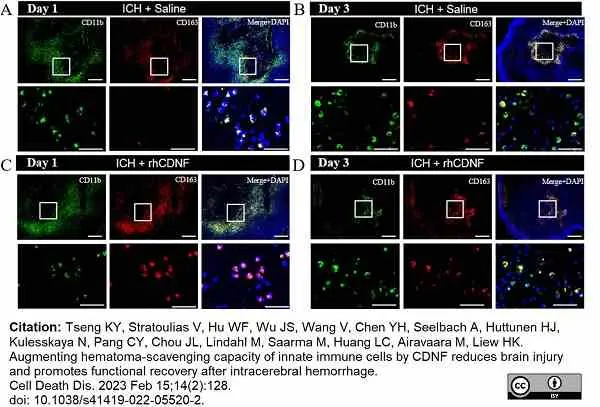
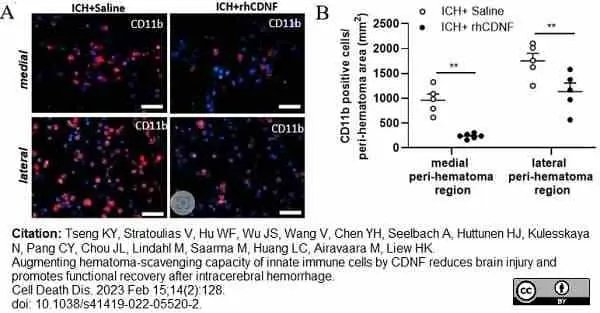
Tseng, K.Y. et al. (2023) Augmenting hematoma-scavenging capacity of innate immune cells by CDNF reduces brain injury and promotes functional recovery after intracerebral hemorrhage.
Cell Death Dis. 14 (2): 128. -
Wilton, D.K. et al. (2023) Microglia and complement mediate early corticostriatal synapse loss and cognitive dysfunction in Huntington's disease.
Nat Med. Oct 09 [Epub ahead of print]. -
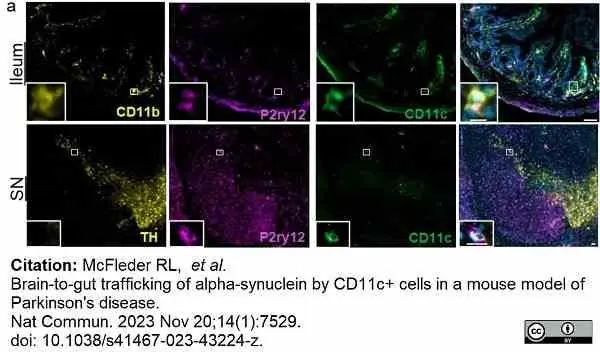
McFleder, R.L. et al. (2023) Brain-to-gut trafficking of alpha-synuclein by CD11c(+) cells in a mouse model of Parkinson's disease.
Nat Commun. 14 (1): 7529. -
Wang, Q. et al. (2018) Sarm1/Myd88-5 Regulates Neuronal Intrinsic Immune Response to Traumatic Axonal Injuries.
Cell Rep. 23 (3): 716-24. -
Türk Börü, Ü. et al. (2024) Alterations in the spinal cord, trigeminal nerve ganglion, and infraorbital nerve through inducing compression of the dorsal horn region at the upper cervical cord in trigeminal neuralgia.
Brain Res. 1832: 148842. -
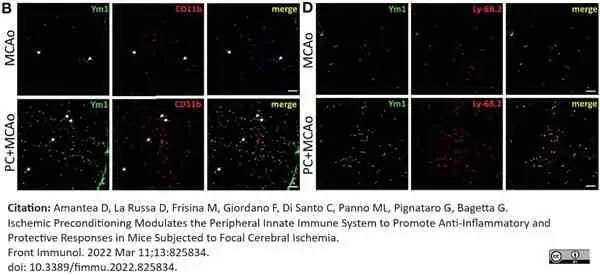
Amantea, D. et al. (2022) Ischemic Preconditioning Modulates the Peripheral Innate Immune System to Promote Anti-Inflammatory and Protective Responses in Mice Subjected to Focal Cerebral Ischemia.
Front Immunol. 13: 825834. -
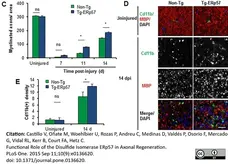
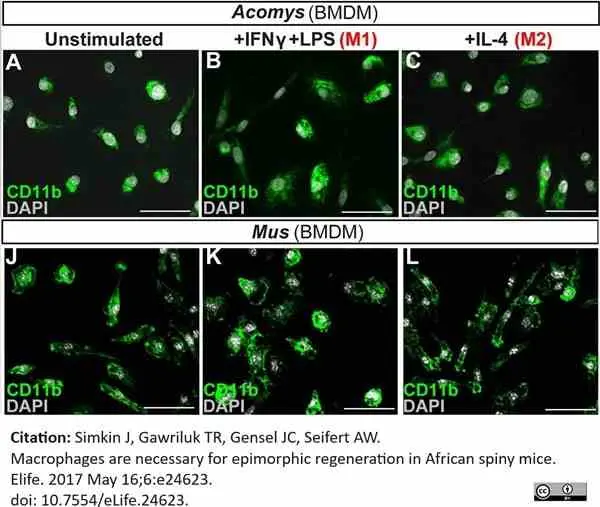
Simkin, J. et al. (2017) Macrophages are necessary for epimorphic regeneration in African spiny mice.
Elife. 6May 16 [Epub ahead of print]. -
Wang, X. et al. (2020) Acute effects of human protein S administration after traumatic brain injury in mice.
Neural Regen Res. 15 (11): 2073-81. -
Choi, Y. et al. (2025) Blood-derived APLP1(+) extracellular vesicles are potential biomarkers for the early diagnosis of brain diseases.
Sci Adv. 11 (1): eado6894. -
Burkhart, A. et al. (2024) Activation of glial cells induces proinflammatory properties in brain capillary endothelial cells in vitro.
Sci Rep. 14 (1): 26580. -
Wang, M.Q. et al. (2025) ClC-3-depedent polarization of microglia protects against cerebral ischemic injury in mice.
Int Immunopharmacol. 155: 114618.
- Synonyms
- CD11
- Integrin Alpha M Chain
- MAC-1
- RRID
- AB_321293
- UniProt
- P05555
- Entrez Gene
- Itgam
- GO Terms
- GO:0001846 opsonin binding
- GO:0004872 receptor activity
- GO:0008305 integrin complex
- GO:0005634 nucleus
- GO:0007159 leukocyte cell-cell adhesion
- GO:0007229 integrin-mediated signaling pathway
- GO:0008201 heparin binding
- GO:0009897 external side of plasma membrane
- GO:0030593 neutrophil chemotaxis
- View More GO Terms
- GO:0043395 heparan sulfate proteoglycan binding
- GO:0045123 cellular extravasation
- GO:0050798 activated T cell proliferation
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Mouse ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up