IL-6 antibody






Rabbit anti Sheep Interleukin-6
- Product Type
- Polyclonal Antibody
- Isotype
- Polyclonal IgG
- Specificity
- IL-6
| Rabbit anti Ovine interleukin-6 antibody recognizes ovine interleukin-6 (IL-6). No cross-reactivity is seen with ovine IL-1 beta, IL-8, MCP or TNF alpha. |
- Target Species
- Sheep
- Species Cross-Reactivity
-
Target Species Cross Reactivity Bovine - N.B. Antibody reactivity and working conditions may vary between species.
- Product Form
- Serum - liquid (diluted)
- Antiserum Preparation
- Antisera to ovine IL-6 were raised by repeated immunisation of rabbits with highly purified antigen.
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- <0.1% Sodium Azide (NaN3)
- Immunogen
- Recombinant IL-6.
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| ELISA | 1/500 | ||
| Western Blotting |
- ELISA
- The antibody may be used in a sandwich ELISA as a detection reagent for ovine IL-6, in combination with MCA1659 as a capture antibody.
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Goat anti Rabbit IgG (Fc):Biotin | STAR121B | E WB | 1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Goat anti Rabbit IgG (Fc):Biotin | ||||||
| Goat anti Rabbit IgG (Fc):FITC | STAR121F | F | 1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Goat anti Rabbit IgG (Fc):FITC | ||||||
| Goat anti Rabbit IgG (Fc):HRP | STAR121P | E WB | 1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Goat anti Rabbit IgG (Fc):HRP | ||||||
| Goat anti Rabbit IgG (H/L):HRP | STAR124P | C E WB | 1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Goat anti Rabbit IgG (H/L):HRP | ||||||
| Sheep anti Rabbit IgG:RPE | STAR35A | F | 1 ml |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Sheep anti Rabbit IgG:RPE | ||||||
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| TidyBlot Western Blot Detection Reagent:HRP | STAR209P | WB * | 0.5 ml | Log in | |||
| List Price | Your Price | ||||||
| Log in | |||||||
| Description | TidyBlot Western Blot Detection Reagent:HRP | ||||||
References for IL-6 antibody
-
Stephens, S. et al. (2003) Differences in cytokine synthesis by the subpopulations of dendritic cells from afferent lymph.
Immunology. 120: 48-57. -
Herath, S. et al. (2009) Expression of genes associated with immunity in the endometrium of cattle with disparate postpartum uterine disease and fertility.
Reprod Biol Endocrinol. 7: 55. -
Wang, Z. et al. (2008) Acute hypercapnia improves indices of tissue oxygenation more than dobutamine in septic shock.
Am J Respir Crit Care Med. 177: 178-83. -
Su, F. et al (2007) Beneficial effects of ethyl pyruvate in septic shock from peritonitis.
Arch Surg.142: 166-71. -
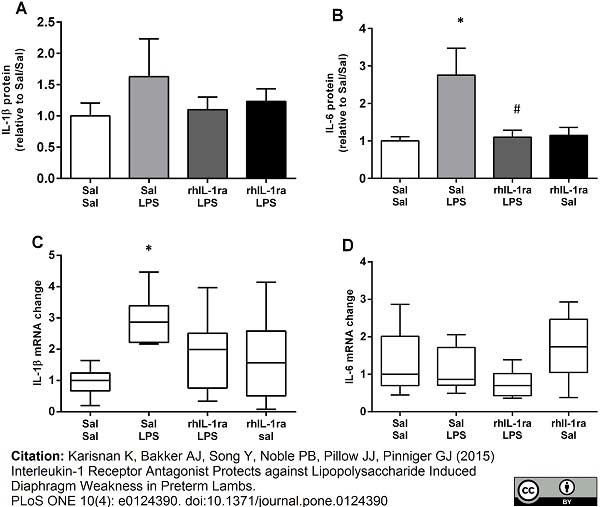
Karisnan, K. et al. (2015) Interleukin-1 Receptor Antagonist Protects against Lipopolysaccharide Induced Diaphragm Weakness in Preterm Lambs.
PLoS One. 10 (4): e0124390. -
Xu, A. et al. (2015) The Ovine Fetal and Placental Inflammatory Response to Umbilical Cord Occlusions With Worsening Acidosis.
Reprod Sci. 22 (11): 1409-20. -
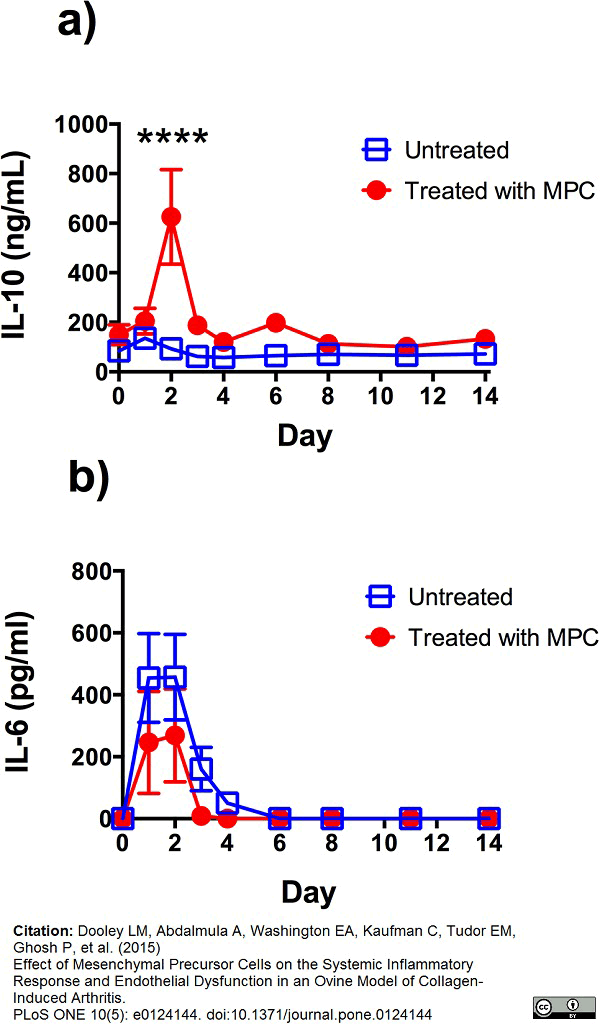
Dooley, L.M. et al. (2015) Effect of mesenchymal precursor cells on the systemic inflammatory response and endothelial dysfunction in an ovine model of collagen-induced arthritis.
PLoS One. 10 (5): e0124144. -
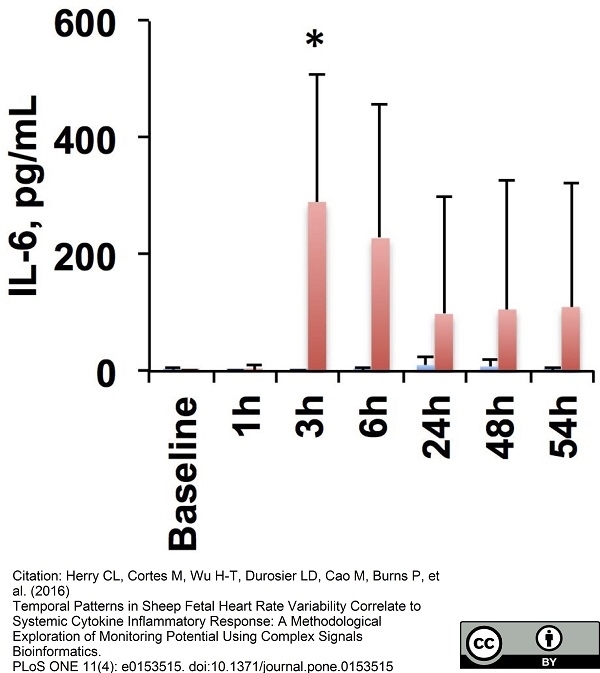
Herry, C.L. et al. (2016) Temporal Patterns in Sheep Fetal Heart Rate Variability Correlate to Systemic Cytokine Inflammatory Response: A Methodological Exploration of Monitoring Potential Using Complex Signals Bioinformatics.
PLoS One. 11 (4): e0153515.
View The Latest Product References
-
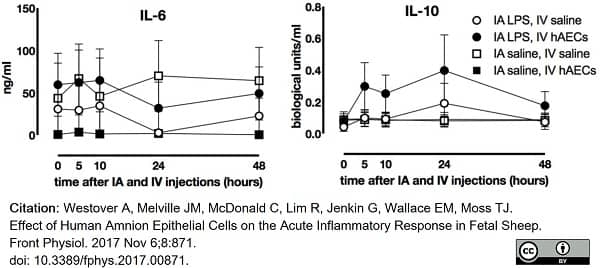
Westover, A. et al. (2017) Effect of Human Amnion Epithelial Cells on the Acute Inflammatory Response in Fetal Sheep.
Front Physiol. 8: 871. -
Castel, A. et al. (2021) Recording and manipulation of vagus nerve electrical activity in chronically instrumented unanesthetized near term fetal sheep.
J Neurosci Methods. 360: 109257. -
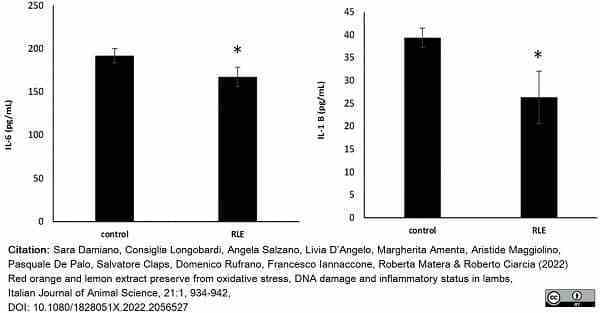
Damiano, S. et al. (2022) Red orange and lemon extract preserve from oxidative stress, DNA damage and inflammatory status in lambs
It J Anim Sci. 21 (1): 934-42. -
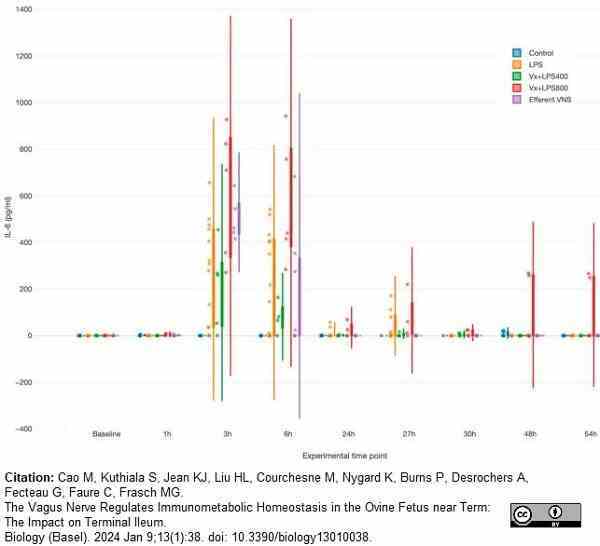
Cao, M. et al. (2024) The Vagus Nerve Regulates Immunometabolic Homeostasis in the Ovine Fetus near Term: The Impact on Terminal Ileum.
Biology (Basel).13 (1): 38. -
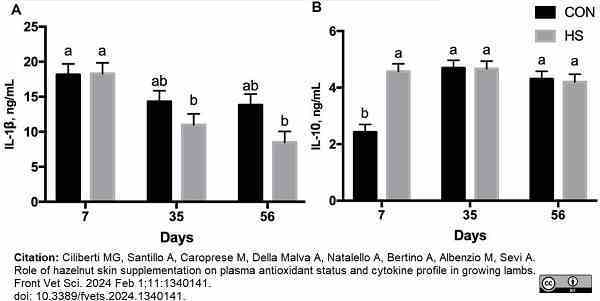
Ciliberti, M.G. et al. (2024) Role of hazelnut skin supplementation on plasma antioxidant status and cytokine profile in growing lambs.
Front Vet Sci. 11: 1340141.
- RRID
- AB_322150
- UniProt
- P29455
- Entrez Gene
- IL6
- GO Terms
- GO:0006953 acute-phase response
- GO:0005125 cytokine activity
- GO:0005138 interleukin-6 receptor binding
- GO:0005615 extracellular space
- GO:0006955 immune response
- GO:0008083 growth factor activity
AHP424
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Sheep ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up