Collagen IV antibody







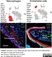
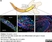
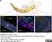
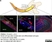
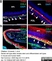




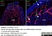
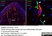

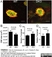
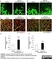

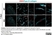











Rabbit anti Mouse Collagen IV
- Product Type
- Polyclonal Antibody
- Isotype
- Polyclonal IgG
- Specificity
- Collagen IV
| Rabbit anti Mouse Collagen IV antibody recognizes mouse collagen type IV. Collagen IV is a 1682 amino acid ~160 kDa (predicted) matrx protein and major component of glomerular basement membranes. Multiple isoforms exist each capable of forming triple helical structures with two other chains to form the type IV collagen network. The collagen IV alpha chain can be cleaved between residues 1444-1445 to yield the c-terminal 225 amino acid, ~28 kDa arresten fragment, collagen α2(IV) yields a c-terminal canstatin fragment while Collagen α3(IV) yeilds a tumstatin fragment. Collagen IV bears a single collagen IV NC1 (C-terminal non-collagenous) domain (UniProt: Q9QZR9). Mutations in collagen IV genes have been implicated in inherited nephropathies and potentially in cystic kidney disease and intracranial aneurysms (Plaisier et al. 2007). Rabbit amti Mouse Collagen IV antibody has been successfully employed for the detection of collagen IV by immunofluorescence and immunohistochemistry in mice (Tang et al. 2010), rats (Shen et al. 2014) and oragutan (Bredies et al. 2013). The following cross reactivities have been observed:
|
- Target Species
- Mouse
- Species Cross-Reactivity
-
Target Species Cross Reactivity Orangutan Rat Human - N.B. Antibody reactivity and working conditions may vary between species.
- Product Form
- Purified Ig - liquid
- Preparation
- Purified IgG prepared by antigen column chromatography
- Buffer Solution
- Phosphate buffered saline
0.1M citrate - Preservative Stabilisers
- <0.1% Sodium Azide (NaN3)
Antibiotic antimycotic mixture 1% - Immunogen
- Collagen IV purified from mouse EHS tumor.
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| ELISA | 1/2000 | ||
| Immunofluorescence | 1/40 | ||
| Immunohistology - Frozen | 1/500 | ||
| Immunohistology - Paraffin | 1/500 |
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Goat anti Rabbit IgG (Fc):Biotin | STAR121B | E WB | 1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Goat anti Rabbit IgG (Fc):Biotin | ||||||
| Goat anti Rabbit IgG (Fc):FITC | STAR121F | F | 1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Goat anti Rabbit IgG (Fc):FITC | ||||||
| Goat anti Rabbit IgG (Fc):HRP | STAR121P | E WB | 1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Goat anti Rabbit IgG (Fc):HRP | ||||||
| Goat anti Rabbit IgG (H/L):HRP | STAR124P | C E WB | 1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Goat anti Rabbit IgG (H/L):HRP | ||||||
| Sheep anti Rabbit IgG:RPE | STAR35A | F | 1 ml |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Sheep anti Rabbit IgG:RPE | ||||||
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Antigen Retrieval Buffer, pH8.0 | BUF025A | P | 500 ml | Log in | |||
| List Price | Your Price | ||||||
| Log in | |||||||
| Description | Antigen Retrieval Buffer, pH8.0 | ||||||
References for Collagen IV antibody
-
Xu Q. et al. (2004) Vascular development in the retina and inner ear: control by Norrin and Frizzled-4, a high-affinity ligand-receptor pair.
Cell. 116: 883-95. -
Kojima, T. et al. (2007) Proangiogenic role of ephrinB1/EphB1 in basic fibroblast growth factor-induced corneal angiogenesis.
Am J Pathol. 170: 764-73. -
Fantin, A. et al. (2010) Tissue macrophages act as cellular chaperones for vascular anastomosis downstream of VEGF-mediated endothelial tip cell induction.
Blood.116: 829-40. -
Chen, M. et al. (2010) Immune activation in retinal aging: a gene expression study.
Invest Ophthalmol Vis Sci. 51: 5888-96. -
Rubin, A.N. et al. (2010) The germinal zones of the basal ganglia but not the septum generate GABAergic interneurons for the cortex.
J Neurosci. 30: 12050-62. -
Scott, A. et al. (2010) Astrocyte-derived vascular endothelial growth factor stabilizes vessels in the developing retinal vasculature.
PLoS One. 5: e11863. -
Armulik, A. et al. (2010) Pericytes regulate the blood-brain barrier.
Nature. 468: 557-61. -
Tang, Z. et al. (2010) Survival effect of PDGF-CC rescues neurons from apoptosis in both brain and retina by regulating GSK3beta phosphorylation.
J Exp Med. 207: 867-80.
View The Latest Product References
-
Stenzel, D. et al. (2011) Integrin-dependent and -independent functions of astrocytic fibronectin in retinal angiogenesis.
Development. 138: 4451-63. -
Li, W. and Mukouyama, Y.S. (2011) Whole-mount immunohistochemical analysis for embryonic limb skin vasculature: a model system to study vascular branching morphogenesis in embryo.
J Vis Exp. 51: pii: 2620. -
Dulauroy, S. et al. (2012) Lineage tracing and genetic ablation of ADAM12(+) perivascular cells identify a major source of profibrotic cells during acute tissue injury.
Nat Med. 18: 1262-70. -
Powner, M.B. et al. (2012) Visualization of gene expression in whole mouse retina by in situ hybridization.
Nat Protoc. 7: 1086-96. -
Zuercher, J. et al. (2012) Norrin stimulates cell proliferation in the superficial retinal vascular plexus and is pivotal for the recruitment of mural cells.
Hum Mol Genet. 21: 2619-30. -
Arnold, T.D. et al. (2012) Defective retinal vascular endothelial cell development as a consequence of impaired integrin αVβ8-mediated activation of transforming growth factor-β.
J Neurosci. 32: 1197-206. -
Takagi, N. et al. (2012) Mineralocorticoid Receptor Blocker Protects against Podocyte-Dependent Glomerulosclerosis.
Nephron Extra. 2: 17-26. -
Ma, S. et al. (2012) Ric-8a, a Guanine Nucleotide Exchange Factor for Heterotrimeric G Proteins, Regulates Bergmann Glia-Basement Membrane Adhesion during Cerebellar Foliation.
J Neurosci. 32: 14979-93. -
McKenzie, J.A. et al. (2012) Apelin is required for non-neovascular remodeling in the retina.
Am J Pathol. 180: 399-409. -
Schulz, C. et al. (2012) A lineage of myeloid cells independent of Myb and hematopoietic stem cells.
Science. 336: 86-90. -
Edgar, K. et al. (2012) eNOS Overexpression Exacerbates Vascular Closure in the Obliterative Phase of OIR and Increases Angiogenic Drive in the Subsequent Proliferative Stage.
Invest Ophthalmol Vis Sci. 53: 6833-50. -
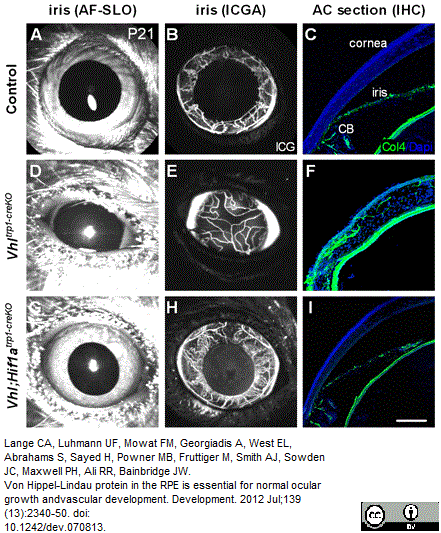
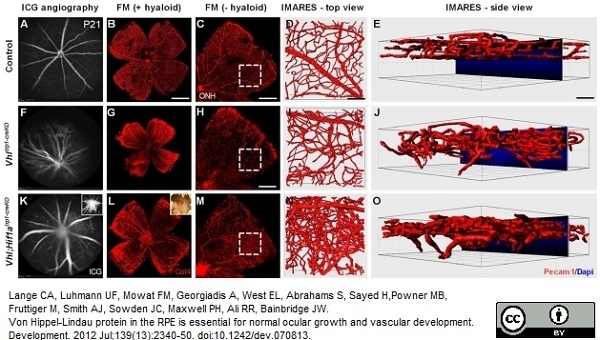
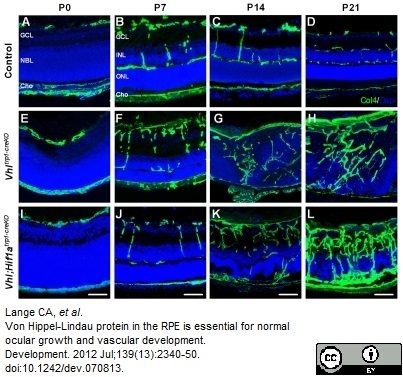
Lange, C.A. et al. (2012) Von Hippel-Lindau protein in the RPE is essential for normal ocular growth and vascular development.
Development. 139: 2340-50. -
Lutter, S. et al. (2012) Smooth muscle-endothelial cell communication activates Reelin signaling and regulates lymphatic vessel formation.
J Cell Biol. 197: 837-49. -
Bredies, K. et al. (2013) Computer-assisted counting of retinal cells by automatic segmentation after TV denoising.
BMC Ophthalmol. 13: 59. -
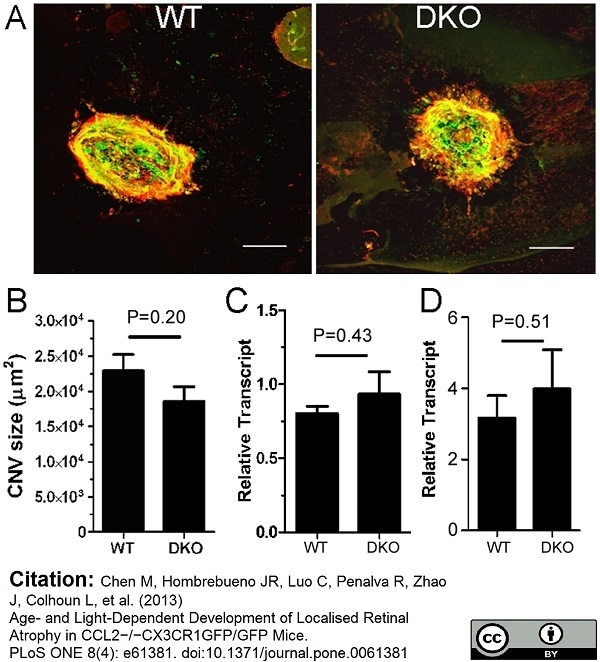
Chen, M. et al. (2013) Age- and light-dependent development of localised retinal atrophy in CCL2(-/-)CX3CR1(GFP/GFP) mice.
PLoS One. 8: e61381. -
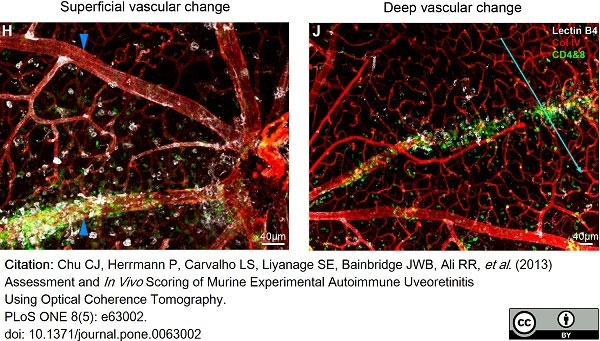
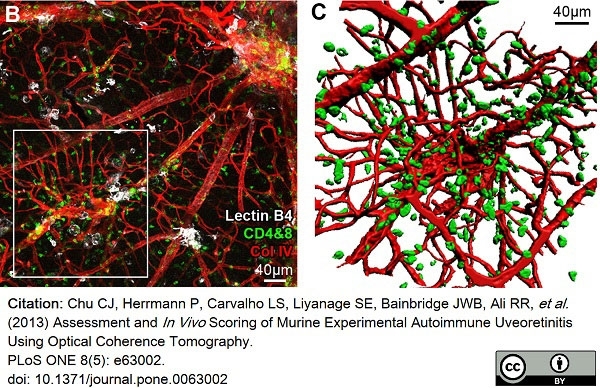
Chu, C.J. et al. (2013) Assessment and in vivo scoring of murine experimental autoimmune uveoretinitis using optical coherence tomography.
PLoS One. 8 (5): e63002. -
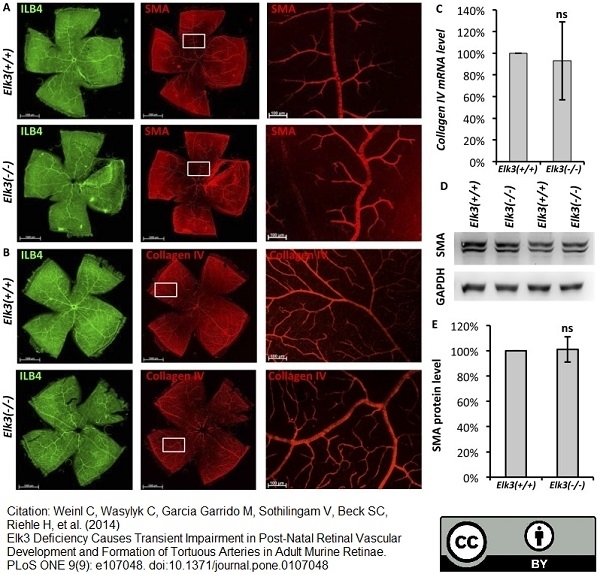
Weinl, C. et al. (2014) Elk3 deficiency causes transient impairment in post-natal retinal vascular development and formation of tortuous arteries in adult murine retinae.
PLoS One. 9: e107048. -
Scott, A. et al. (2014) Quantification of vascular tortuosity as an early outcome measure in oxygen induced retinopathy (OIR)
Exp Eye Res. 120: 55-60. -
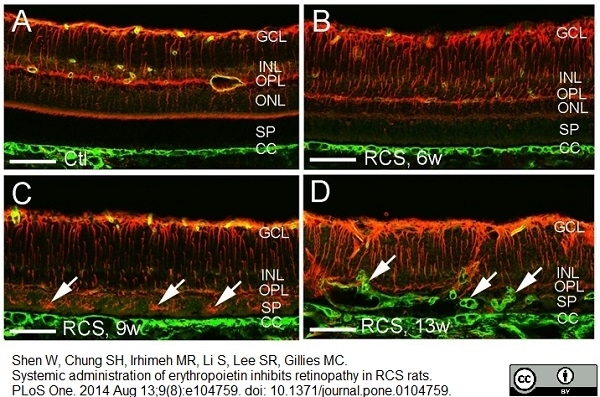
Shen, W. et al. (2014) Systemic Administration of Erythropoietin Inhibits Retinopathy in RCS Rats.
PLoS One. 9: e104759. -
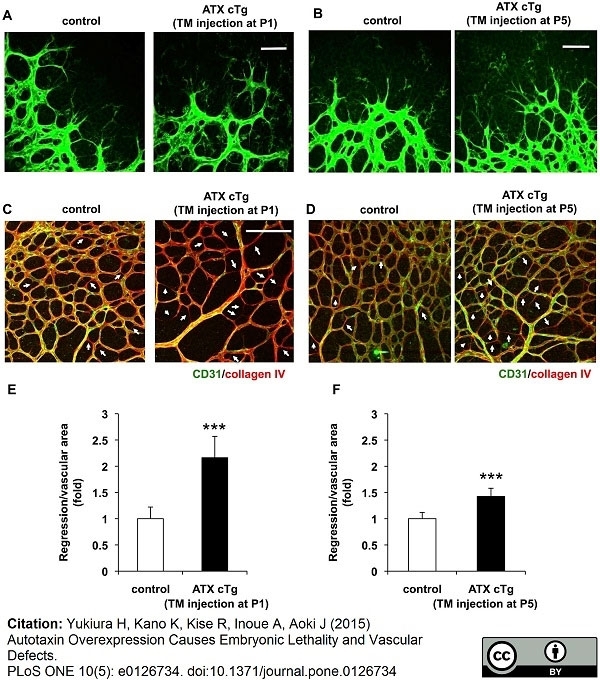
Yukiura H et al. (2015) Autotaxin overexpression causes embryonic lethality and vascular defects.
PLoS One. 10 (5): e0126734. -
Wu, W.K. et al. (2015) IL-4 regulates specific Arg-1(+) macrophage sFlt-1-mediated inhibition of angiogenesis.
Am J Pathol. 185 (8): 2324-35. -
Williams, J.A. et al. (2016) Regulation of C3 Activation by the Alternative Complement Pathway in the Mouse Retina.
PLoS One. 11 (8): e0161898. -
Piñero, G. et al. (2017) Lithium Reversibly Inhibits Schwann Cell Proliferation and Differentiation Without Inducing Myelin Loss.
Mol Neurobiol. 54 (10): 8287-307. -
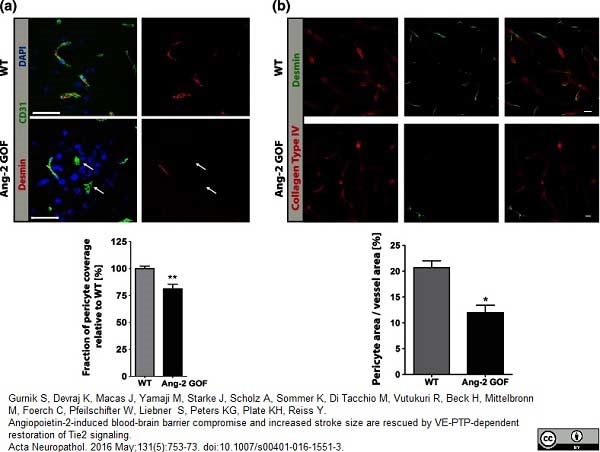
Gurnik, S. et al. (2016) Angiopoietin-2-induced blood-brain barrier compromise and increased stroke size are rescued by VE-PTP-dependent restoration of Tie2 signaling.
Acta Neuropathol. 131 (5): 753-73. -
Misra, A. et al. (2016) Integrin β3 inhibition is a therapeutic strategy for supravalvular aortic stenosis.
J Exp Med. 213 (3): 451-63. -
Yanagida, K. et al. (2017) Size-selective opening of the blood-brain barrier by targeting endothelial sphingosine 1-phosphate receptor 1.
Proc Natl Acad Sci U S A. 114 (17): 4531-6. -
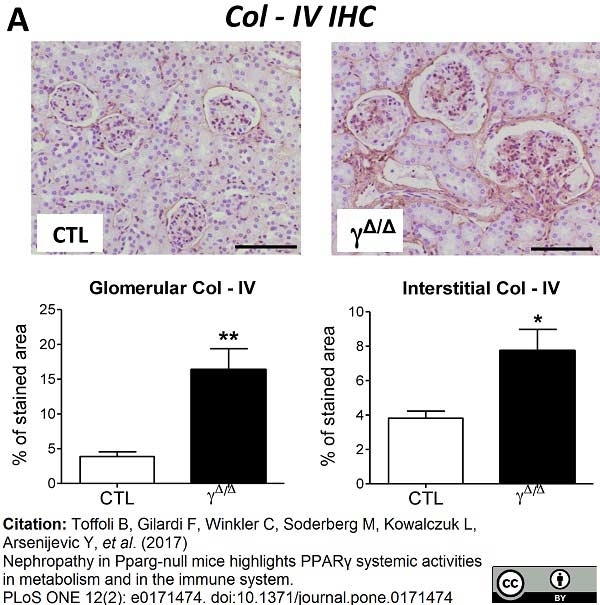
Toffoli, B. et al. (2017) Nephropathy in Pparg-null mice highlights PPARγ systemic activities in metabolism and in the immune system.
PLoS One. 12 (2): e0171474. -
Fernández-robredo, P. et al. (2017) Neuropilin 1 Involvement in Choroidal and Retinal Neovascularisation.
PLoS One. 12 (1): e0169865. -
Kim, B. et al. (2018) Endothelial pyruvate kinase M2 maintains vascular integrity.
J Clin Invest. 128 (10): 4543-56. -
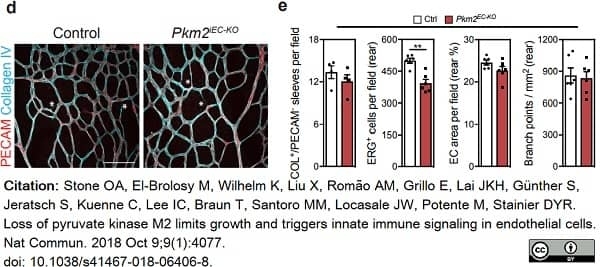
Stone, O.A. et al. (2018) Loss of pyruvate kinase M2 limits growth and triggers innate immune signaling in endothelial cells.
Nat Commun. 9 (1): 4077. -
Niaudet, C. et al. (2019) Adgrf5 contributes to patterning of the endothelial deep layer in retina.
Angiogenesis. 22 (4): 491-505. -
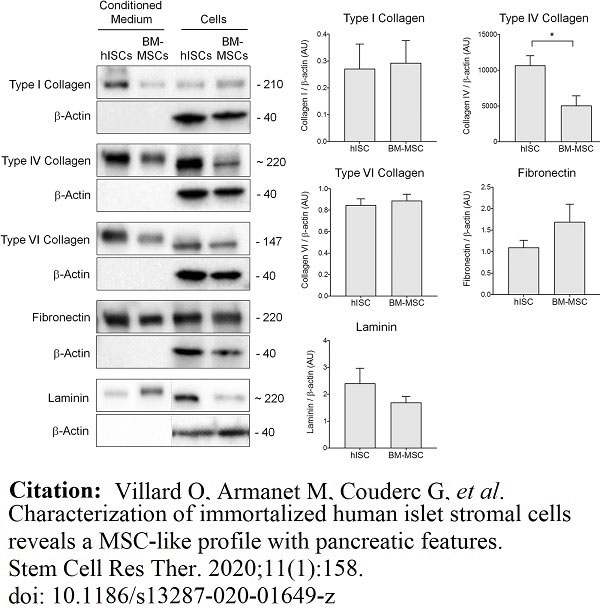
Villard, O. et al. (2020) Characterization of immortalized human islet stromal cells reveals a MSC-like profile with pancreatic features.
Stem Cell Res Ther. 11 (1): 158. -
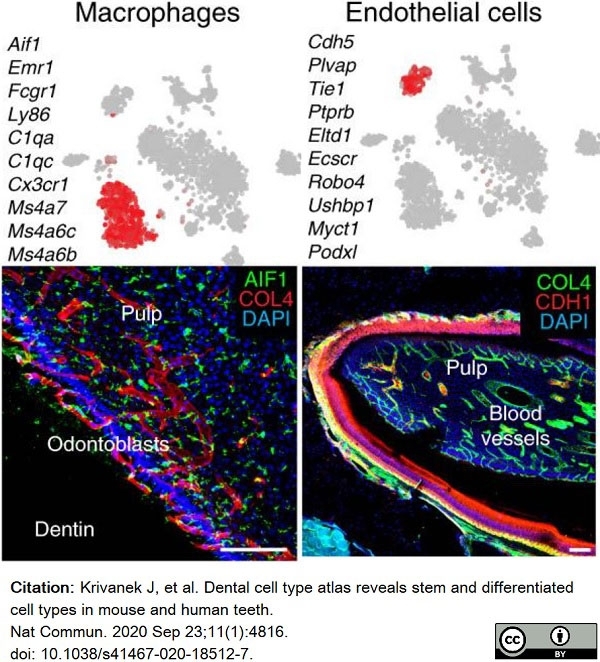
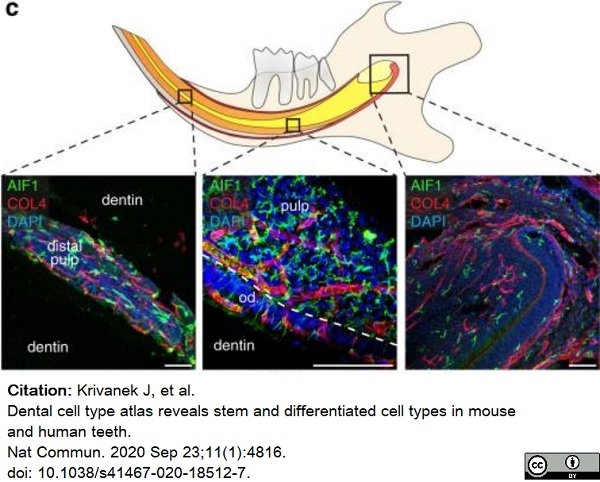
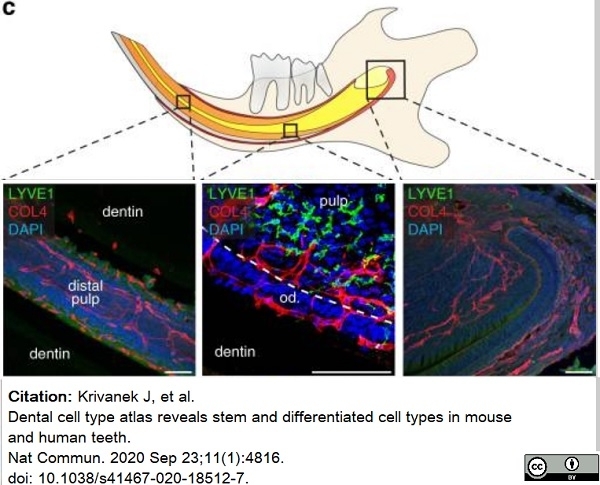
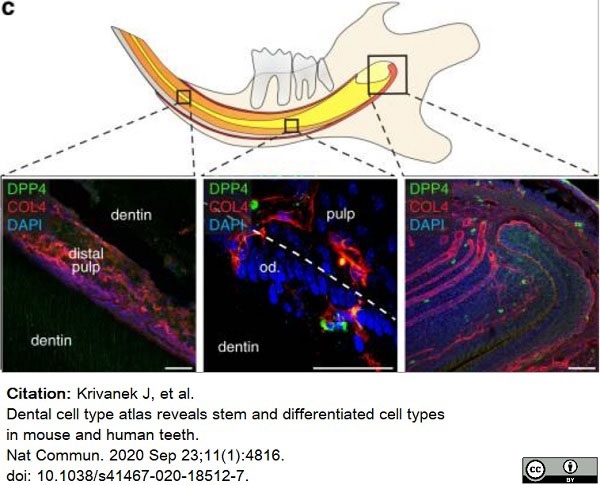
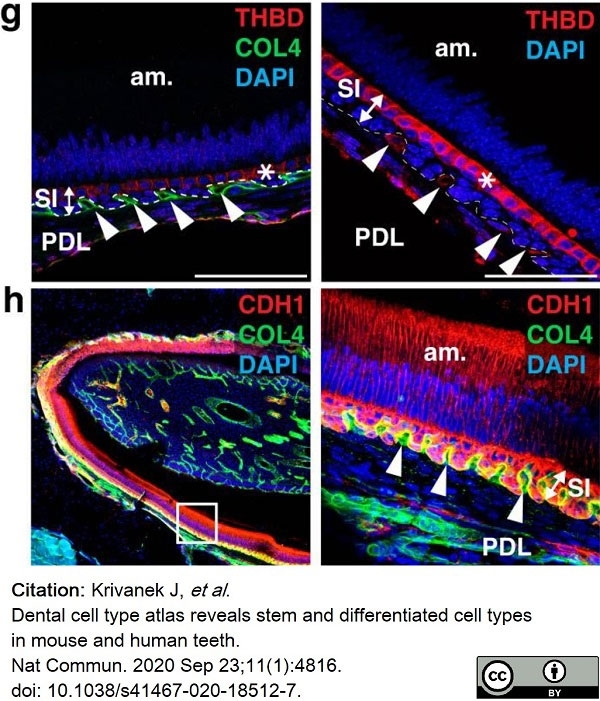
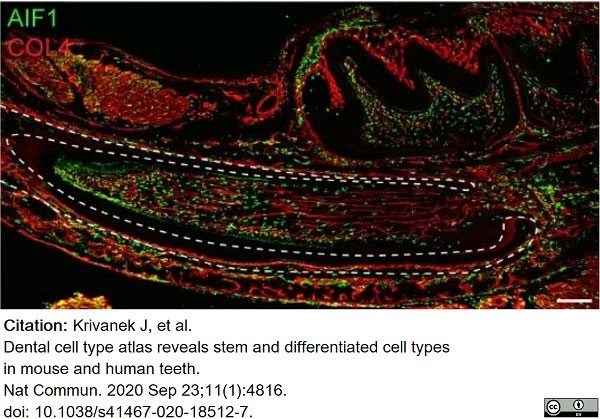
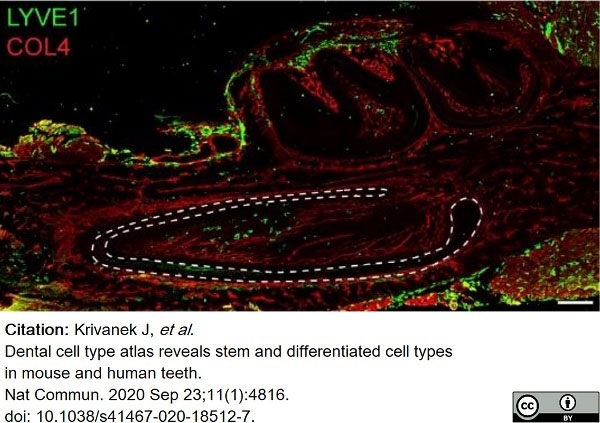
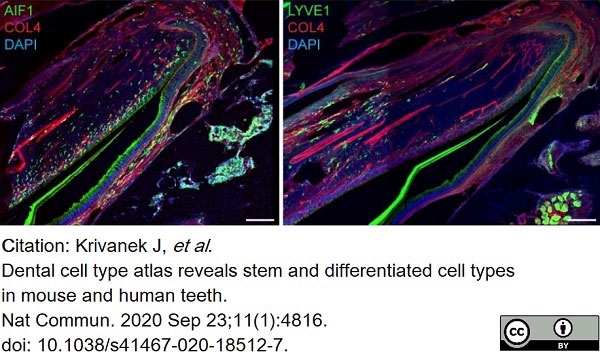
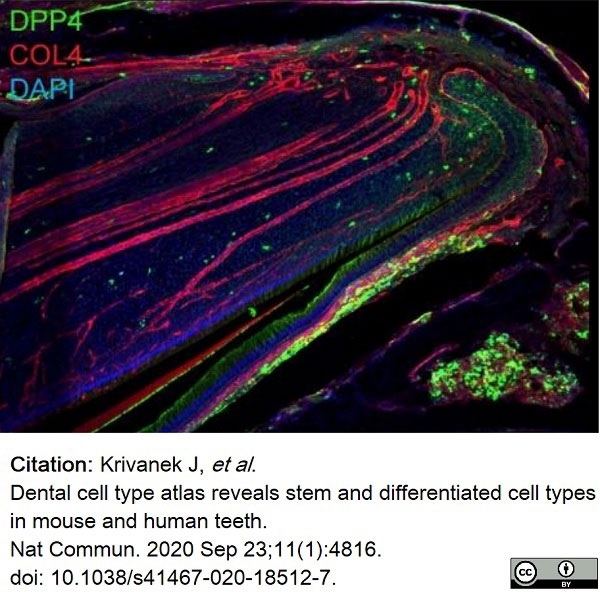
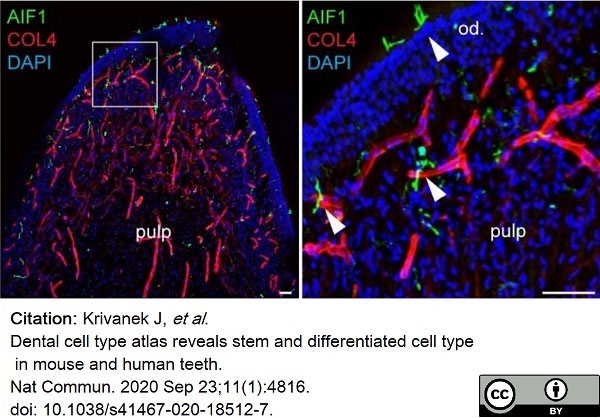
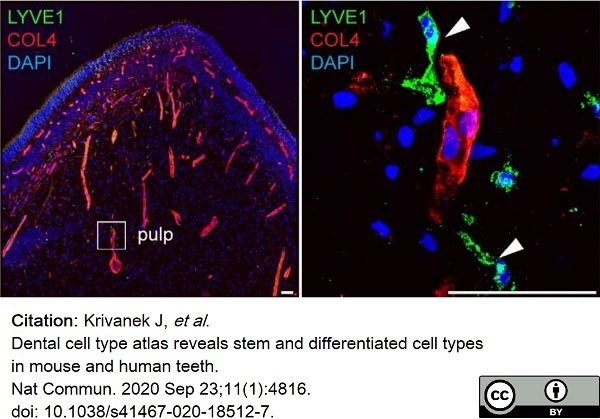
Krivanek, J. et al. (2020) Dental cell type atlas reveals stem and differentiated cell types in mouse and human teeth.
Nat Commun. 11 (1): 4816. -
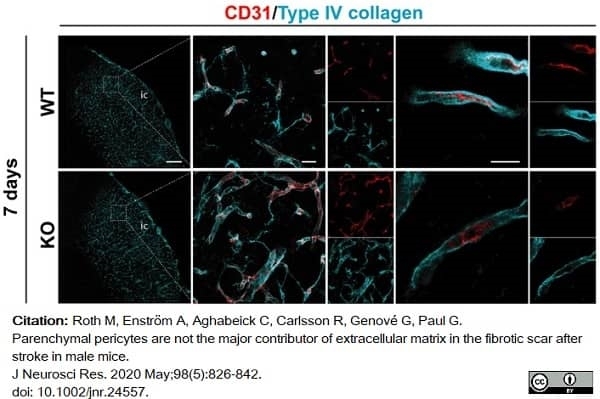
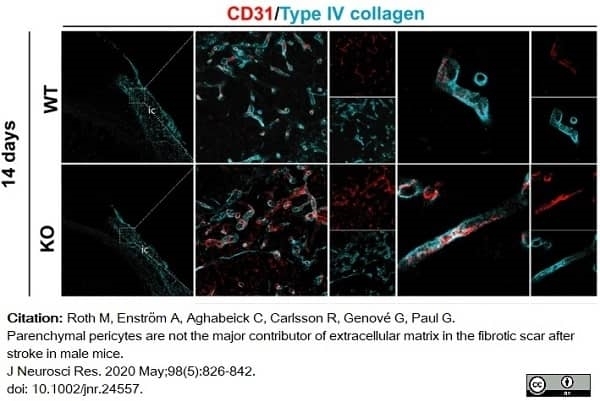
Roth, M. et al. (2020) Parenchymal pericytes are not the major contributor of extracellular matrix in the fibrotic scar after stroke in male mice.
J Neurosci Res. 98 (5): 826-842. -
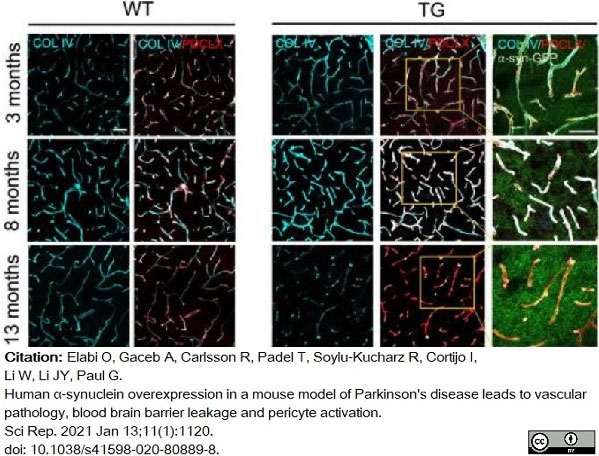
Elabi, O. et al. (2021) Human α-synuclein overexpression in a mouse model of Parkinson's disease leads to vascular pathology, blood brain barrier leakage and pericyte activation.
Sci Rep. 11 (1): 1120. -
Månberg, A. et al. (2021) Altered perivascular fibroblast activity precedes ALS disease onset.
Nat Med. 27 (4): 640-6. -
Mohanta, S.K. et al. (2022) Neuroimmune cardiovascular interfaces control atherosclerosis.
Nature. 605 (7908): 152-9. -
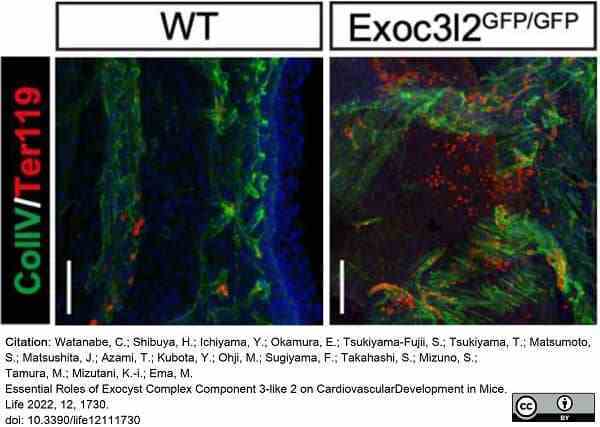
Watanabe, C. et al. (2022) Essential Roles of Exocyst Complex Component 3-like 2 on Cardiovascular Development in Mice.
Life (Basel). 12 (11): 1730. -
Tsuji-Tamura, K. & Ogawa, M. (2023) FOXO1 promotes endothelial cell elongation and angiogenesis by up-regulating the phosphorylation of myosin light chain 2.
Angiogenesis. 26 (4): 523-45. -
Yin, C. et al. (2019) ApoE attenuates unresolvable inflammation by complex formation with activated C1q.
Nat Med. 25 (3): 496-506. -
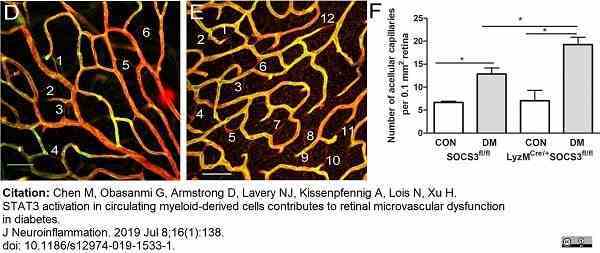
Chen, M. et al. (2018) Cytokine Signaling Protein 3 Deficiency in Myeloid Cells Promotes Retinal Degeneration and Angiogenesis through Arginase-1 Up-Regulation in Experimental Autoimmune Uveoretinitis.
Am J Pathol. 188 (4): 1007-20. -
Schley, G. et al. (2018) Multiparametric magnetic resonance imaging of experimental chronic kidney disease: A quantitative correlation study with histology.
PLoS One. 13 (7): e0200259. -
Zhao, W. et al. (2019) Endothelial CDS2 deficiency causes VEGFA-mediated vascular regression and tumor inhibition.
Cell Res. 29 (11): 895-910. -
Kool, H.M. et al. (2019) Inhibition of retinoic acid signaling induces aberrant pericyte coverage and differentiation resulting in vascular defects in congenital diaphragmatic hernia.
Am J Physiol Lung Cell Mol Physiol. 317 (3): L317-L331. -
Cottarelli, A. et al. (2020) Fgfbp1 promotes blood-brain barrier development by regulating collagen IV deposition and maintaining Wnt/β-catenin signaling.
Development. 147 (16): dev185140. -
Santamaría, R. et al. (2023) Capillary pruning couples tissue perfusion and oxygenation with cardiomyocyte maturation in the postnatal mouse heart.
Front Cell Dev Biol. 11: 1256127. -
Chen, M. et al. (2019) STAT3 activation in circulating myeloid-derived cells contributes to retinal microvascular dysfunction in diabetes.
J Neuroinflammation. 16 (1): 138. -
Kautzman, A.G. et al. (2018) Sox2 regulates astrocytic and vascular development in the retina.
Glia. 66 (3): 623-36. -
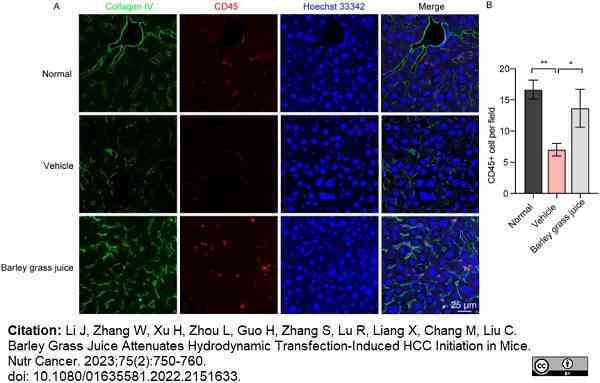
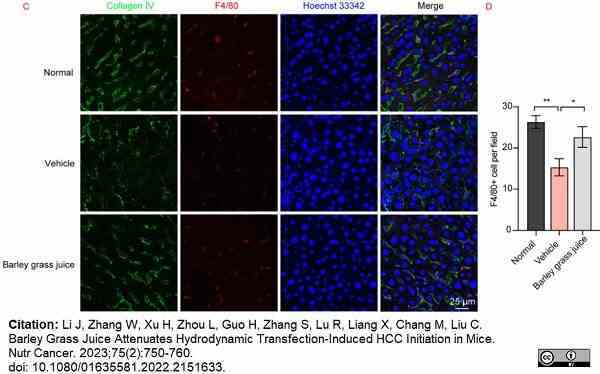
Li, J. et al. (2023) Barley Grass Juice Attenuates Hydrodynamic Transfection-Induced HCC Initiation in Mice.
Nutr Cancer. 75 (2): 750-60. -
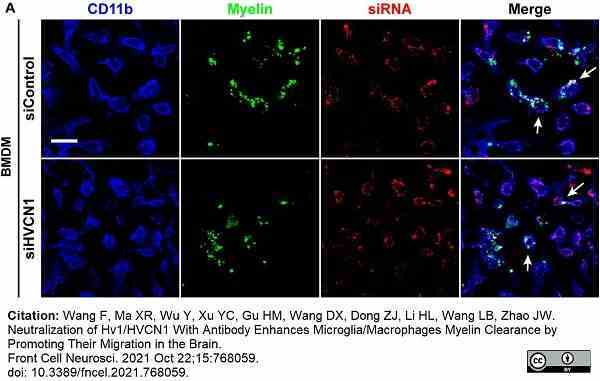
Wang, F. et al. (2021) Neutralization of Hv1/HVCN1 With Antibody Enhances Microglia/Macrophages Myelin Clearance by Promoting Their Migration in the Brain.
Front Cell Neurosci. 15: 768059. -
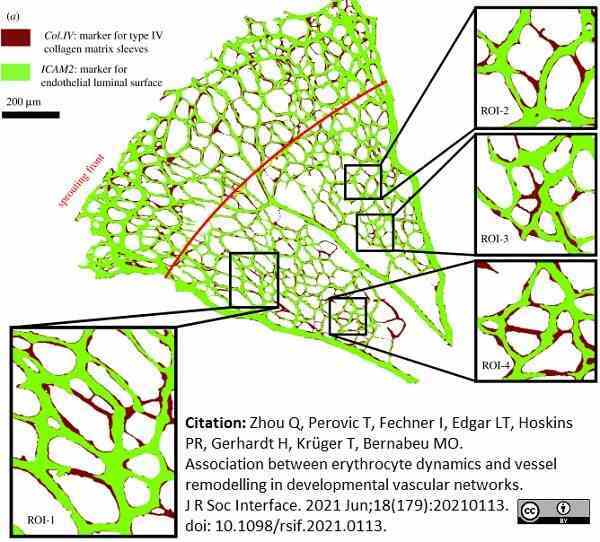
Zhou, Q. et al. (2021) Association between erythrocyte dynamics and vessel remodelling in developmental vascular networks.
J R Soc Interface. 18 (179): 20210113.
- RRID
- AB_2082660
- UniProt
- P02463
- P08122
- Q9QZS0
- Q9QZR9
- Q80V57
- Q6PFB1
- Entrez Gene
- Col4a1
- Col4a2
- Col4a4
- Col4a3
- GO Terms
- GO:0001525 angiogenesis
- GO:0005201 extracellular matrix structural constituent
- GO:0005587 collagen type IV
- GO:0007528 neuromuscular junction development
- GO:0005488 binding
- GO:0032836 glomerular basement membrane development
- GO:0007155 cell adhesion
- GO:0005178 integrin binding
- GO:0006917 induction of apoptosis
- View More GO Terms
- GO:0006919 activation of caspase activity
- GO:0007166 cell surface receptor linked signaling pathway
- GO:0008283 cell proliferation
- GO:0016525 negative regulation of angiogenesis
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Mouse ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up