Tocilizumab antibody | AbD21345_hIgG1





Human anti Tocilizumab
- Product Type
- Monoclonal Antibody
- Clone
- AbD21345_hIgG1
- Isotype
- IgG1
- Specificity
- Tocilizumab
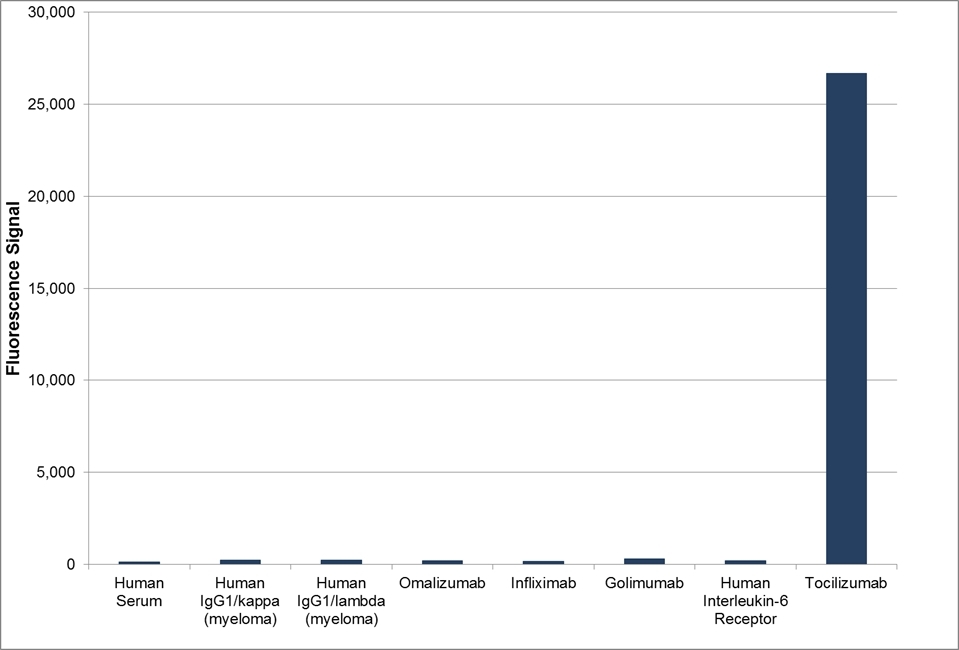
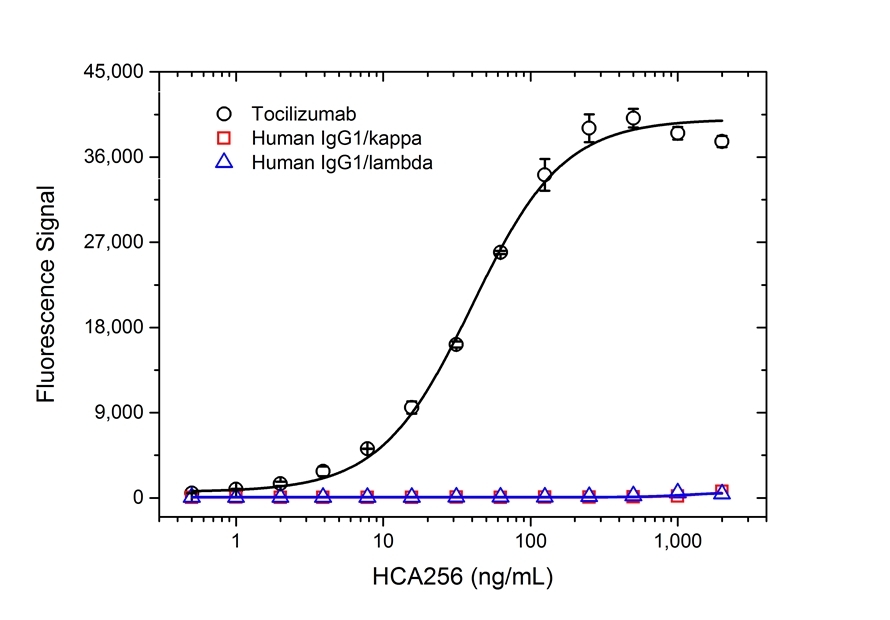
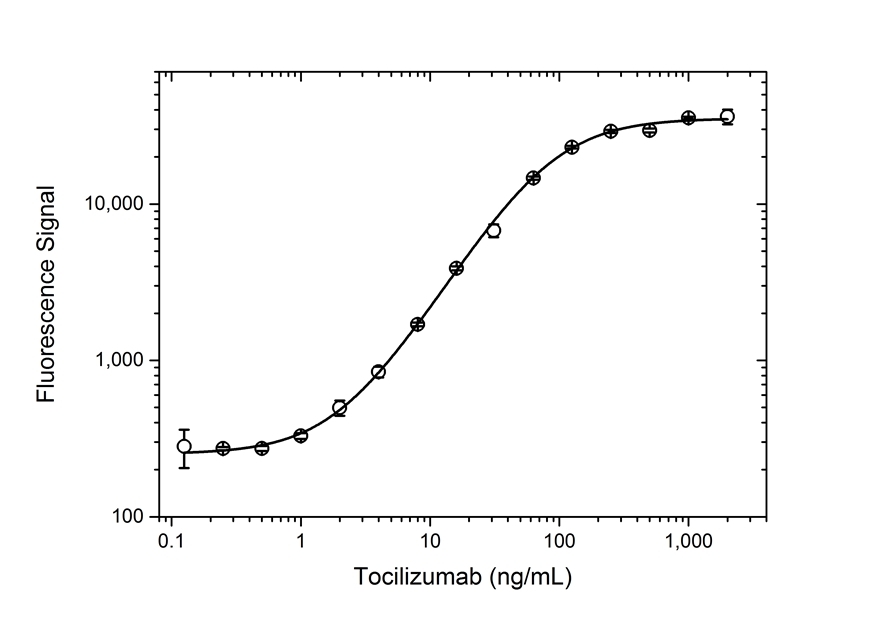
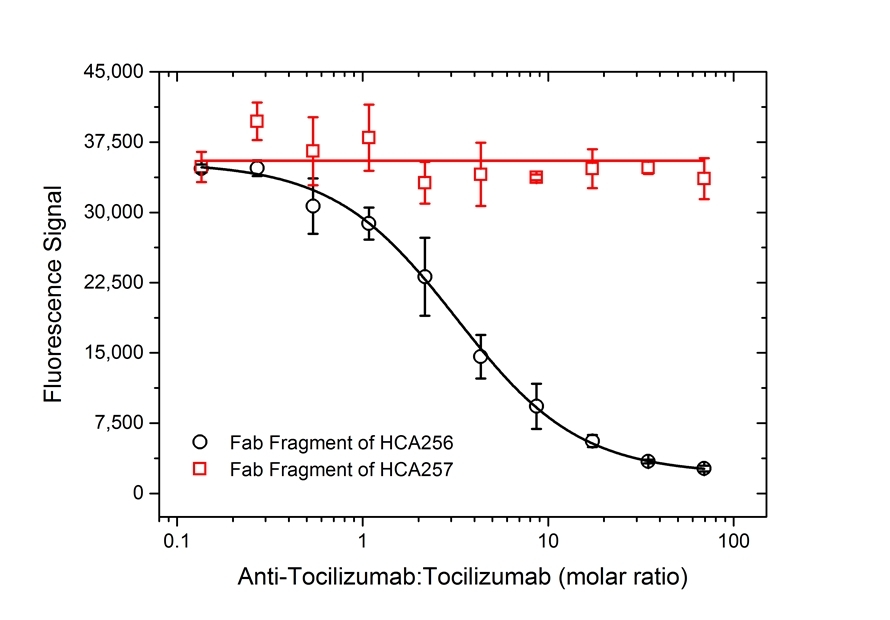
| Human Anti-Tocilizumab Antibody, clone AbD21345_hIgG1is a paratope specific, anti-idiotypic antibody that binds to free tocilizumab but not to human Interleukin-6 Receptor (IL-6R) or the drug/IL-6R complex. Clone AbD21345_hIgG1 is a fully human recombinant monoclonal antibody with IgG1 isotype and is suitable as a surrogate positive control or calibrator (low affinity) in an anti-drug antibody (ADA) assay. Tocilizumab (RoActemra/Actemra) is a humanized IgG1/kappa monoclonal antibody approved for the treatment of moderate to severe rheumatoid arthritis and for the treatment of systemic juvenile idiopathic arthritis. The therapeutic antibody is directed against IL-6R and acts by blocking the binding of IL-6R (both the soluble form, sIL-6R, and the membrane bound form, mIL-6R) to IL-6, thus impeding the pro-inflammatory properties of this cytokine. View a summary of all anti-tocilizumab antibodies |
|
- Product Form
- Human IgG1 antibody (lambda light chain) selected from the HuCAL phage display library and expressed in a human cell line. This antibody is supplied as a liquid.
- Preparation
- Purified IgG prepared by affinity chromatography on Protein A
- Source
- HKB-11
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- 0.09% Sodium Azide (NaN3)
- Immunogen
- Tocilizumab
- Affinity
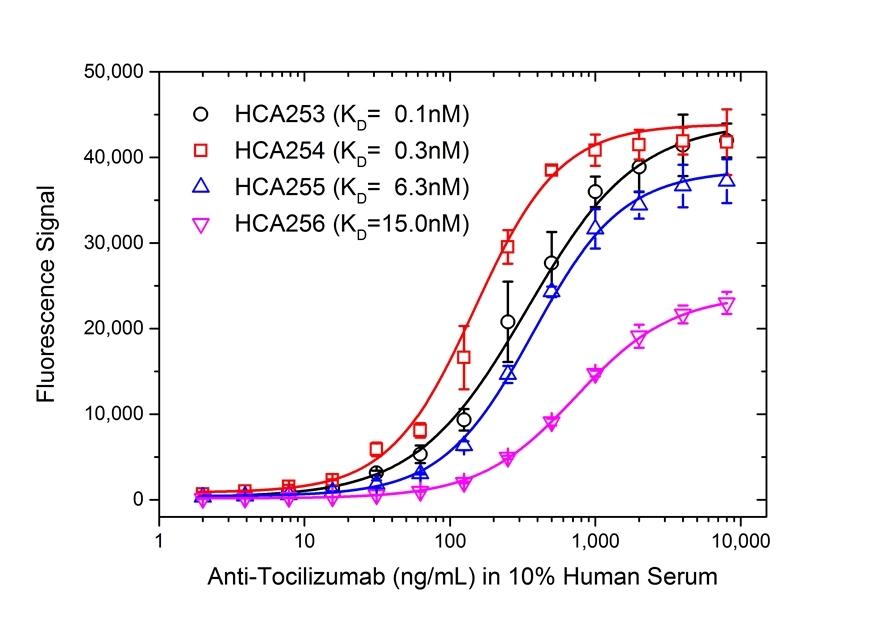
- The intrinsic affinity of the monovalent form of this antibody is KD = 15 nM as measured by real time, label free molecular interaction analysis on immobilized tocilizumab.
- Approx. Protein Concentrations
- Antibody concentration 0.5 mg/ml
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
- Acknowledgements
- This product and/or its use is covered by claims of U.S. patents, and/or pending U.S. and non-U.S. patent applications owned by or under license to Bio-Rad Laboratories, Inc. See bio-rad.com/en-us/trademarks for details.
Actemra and RoActemra are registered trademarks of Hoffman-La Roche and Chugai Pharmaceuticals co., Ltd. - Licensed Use
- For in vitro research purposes and for commercial applications for the provision of in vitro testing services to support preclinical and clinical drug development. Any re-sale in any form or any other commercial application needs a written agreement with Bio-Rad.
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| ELISA |
- Technical Advice
- Recommended protocols and further information about HuCAL recombinant antibody technology can be found in the HuCAL Antibodies Technical Manual.
- ELISA
- This product may be used in a direct ELISA or as a detection reagent in a bridging ELISA together with HCA252 as the capture reagent.
Protocol: PK bridging ELISA to measure free drug.
This antibody is fully human and can be used as a reference standard in an ADA assay.
Protocol: ADA bridging ELISA.
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Hispec Assay Diluent | BUF049A | E IY | 50 ml | Log in | |||
| List Price | Your Price | ||||||
| Log in | |||||||
| Description | Hispec Assay Diluent | ||||||
| Human anti Tocilizumab | HCA252 | E * | 0.1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Human anti Tocilizumab | ||||||
| Human anti Tocilizumab | HCA253 | E | 0.1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Human anti Tocilizumab | ||||||
| Human anti Tocilizumab | HCA254 | E | 0.1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Human anti Tocilizumab | ||||||
| Human anti Tocilizumab | HCA255 | E | 0.1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Human anti Tocilizumab | ||||||
| Human anti Tocilizumab | HCA257 | E | 0.1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Human anti Tocilizumab | ||||||
| LYNX Rapid HRP Antibody Conjugation Kit | LNK001P | CJ | 1 Conjugation For 400µg Antibody | Log in | |||
| List Price | Your Price | ||||||
| Log in | |||||||
| Description | LYNX Rapid HRP Antibody Conjugation Kit | ||||||
| LYNX Rapid HRP Antibody Conjugation Kit | LNK002P | CJ | 3 Conjugations For 400µg Antibody | Log in | |||
| List Price | Your Price | ||||||
| Log in | |||||||
| Description | LYNX Rapid HRP Antibody Conjugation Kit | ||||||
| LYNX Rapid HRP Antibody Conjugation Kit | LNK003P | CJ | 1 Conjugation For 4mg Antibody | Log in | |||
| List Price | Your Price | ||||||
| Log in | |||||||
| Description | LYNX Rapid HRP Antibody Conjugation Kit | ||||||
| LYNX Rapid HRP Antibody Conjugation Kit | LNK004P | CJ | 5 Conjugations For 4mg Antibody | Log in | |||
| List Price | Your Price | ||||||
| Log in | |||||||
| Description | LYNX Rapid HRP Antibody Conjugation Kit | ||||||
| LYNX Rapid HRP Antibody Conjugation Kit | LNK005P | CJ | 1 Conjugation For 20mg Antibody | Log in | |||
| List Price | Your Price | ||||||
| Log in | |||||||
| Description | LYNX Rapid HRP Antibody Conjugation Kit | ||||||
| LYNX Rapid HRP Antibody Conjugation Kit | LNK006P | CJ | 3 Conjugations For 40µg Antibody | Log in | |||
| List Price | Your Price | ||||||
| Log in | |||||||
| Description | LYNX Rapid HRP Antibody Conjugation Kit | ||||||
- Synonyms
- ACTEMRA®
- ROACTEMRA®
HCA256
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
Always be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up