Golimumab antibody | AbD25455




Human anti Golimumab
- Product Type
- Monoclonal Antibody
- Clone
- AbD25455
- Isotype
- HuCAL Fab monovalent
- Specificity
- Golimumab
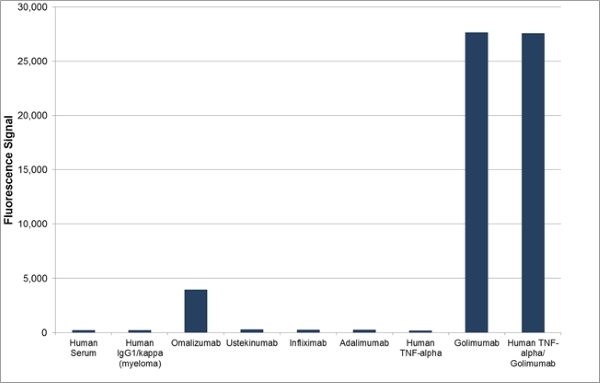
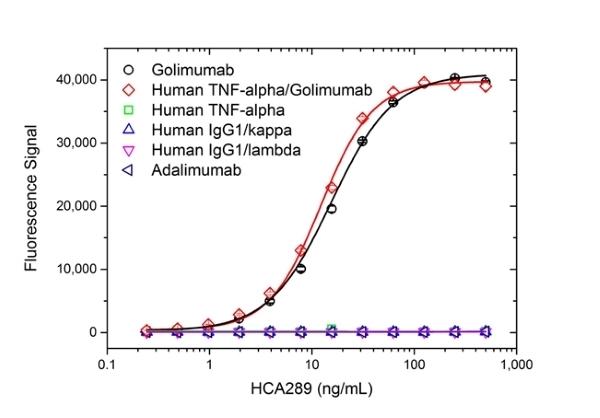
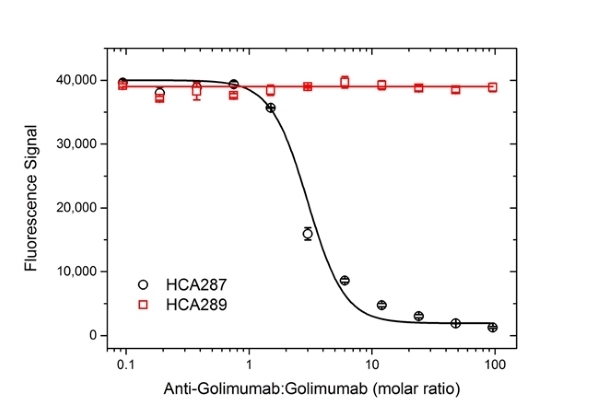
| Human Anti-Golimumab Antibody, clone AbD25455 is an anti-idiotypic antibody that specifically recognizes the monoclonal antibody drug golimumab. This antibody binds to both free golimumab and to golimumab bound to its target tumor necrosis factor alpha (TNFα). Clone AbD25455 does not inhibit the binding of golimumab to its target TNFα and can be used to measure total golimumab and biosimilar products in bioanalytical assays. Clone AbD25455 is an affinity-matured variant of clone AbD20897 (HCA242), with about 7-fold affinity improvement. A pair of anti golimumab antibodies can be used to develop a pharmacokinetic (PK) bridging assay to measure free drug. This antibody, in monovalent Fab format, is recommended as the capture antibody, paired with an antibody in full immunoglobulin format, clone AbD25451_hIgG1 (HCA290), as the detection antibody. Golimumab, marketed under the brand name Simponi is a recombinant human IgG1/kappa monoclonal antibody approved for the treatment of rheumatoid arthritis, psoriatic arthritis and ankylosing spondylitis. This therapeutic antibody, directed against TNFα, acts by blocking the binding of TNFα to its receptors, resulting in a down-regulation of the inflammatory response associated with autoimmune diseases. View a summary of all anti-golimumab antibodies |
- Product Form
- A monovalent human recombinant Fab (lambda light chain) selected from the HuCAL phage display library, expressed in E. coli. The antibody is tagged with a DYKDDDDK tag and a HIS-tag (HHHHHH) at the C-terminus of the antibody heavy chain. This antibody is supplied as a liquid.
- Preparation
- Metal chelate affinity chromatography
- Source
- E.coli
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- 0.09% Sodium Azide (NaN3)
- Immunogen
- Golimumab
- Affinity
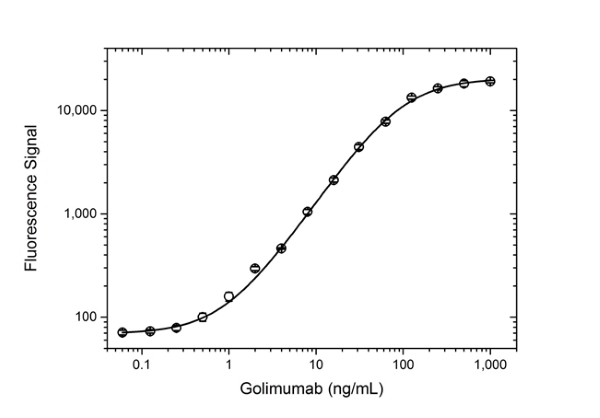
- The intrinsic affinity of the monovalent form of Human anti Golimumab antibody, clone AbD25455 is KD = 0.9 nM as measured by real time, label free molecular interaction analysis on immobilized Golimumab.
- Approx. Protein Concentrations
- Antibody concentration 0.5 mg/ml
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
- Acknowledgements
- This product and/or its use is covered by claims of U.S. patents, and/or pending U.S. and non-U.S. patent applications owned by or under license to Bio-Rad Laboratories, Inc. See bio-rad.com/en-us/trademarks for details.
Simponi is a trademark of Janssen Biotech, Inc. in the USA and Merck & Co, Inc. in Europe.
His-tag is a trademark of EMD Biosciences.
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| ELISA |
- Technical Advice
- Recommended protocols and further information about HuCAL recombinant antibody technology can be found in the HuCAL Antibodies Technical Manual.
- ELISA
- Human anti Golimumab antibody, clone AbD25455 can be used in a direct or indirect ELISA system or as capture antibody for golimumab in a bridging ELISA together with HCA290 (AbD25455_hIgG1) as the detection reagent.
Protocol: PK bridging ELISA to measure total drug.
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Human anti Golimumab | HCA290 | E | 0.1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Human anti Golimumab | ||||||
| Human anti Golimumab:HRP | HCA290P | E | 0.1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Human anti Golimumab:HRP | ||||||
| LYNX Rapid HRP Antibody Conjugation Kit | LNK001P | CJ | 1 Conjugation For 400µg Antibody | Log in | |||
| List Price | Your Price | ||||||
| Log in | |||||||
| Description | LYNX Rapid HRP Antibody Conjugation Kit | ||||||
| Mouse anti Penta Histidine Tag:HRP | MCA5995P | E WB | 125 µl |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse anti Penta Histidine Tag:HRP | ||||||
| Mouse anti Human IgG (Fc) CH2 Domain:HRP | MCA647P | C * E | 0.2 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse anti Human IgG (Fc) CH2 Domain:HRP | ||||||
References for Golimumab antibody
-
Harth, S. et al. (2019) Generation by phage display and characterization of drug-target complex-specific antibodies for pharmacokinetic analysis of biotherapeutics.
MAbs. 11 (1): 178-90.
- Synonyms
- Simponi
- Licensed Use
- For in vitro research purposes and for commercial applications for the provision of in vitro testing services to support preclinical and clinical drug development. Any re-sale in any form or any other commercial application needs a written agreement with Bio-Rad.
HCA289
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
Always be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up