MHC Class II Monomorphic antibody | YKIX334.2
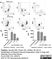
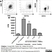
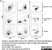



Rat anti Dog MHC Class II Monomorphic
- Product Type
- Monoclonal Antibody
- Clone
- YKIX334.2
- Isotype
- IgG2a
- Specificity
- MHC Class II Monomorphic
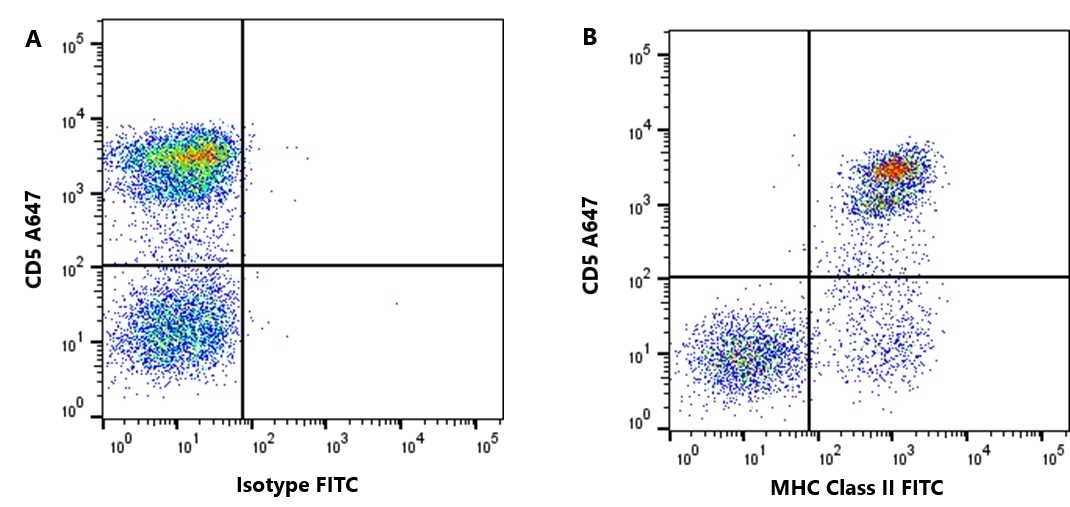
| Rat anti Dog MHC Class II Monomorphic antibody, clone YKIX334.2 recognizes a monomorphic epitope on canine MHC Class II and was classified at the First Canine Leucocyte Antigen Workshop (Cobbold et al. 1994).The major histocompatibility complex (MHC) is a cluster of genes that are important in the immune response to infections. In dogs, this is referred to as the dog leukocyte antigen (DLA) region. Rat anti Dog MHC Class II immunoprecipitates an antigen of ~32/34 kDa and blocks the proliferation of MHC Class II dependent responses in vitro. In dogs, MHC Class II is expressed by all peripheral blood mononuclear cells. |
- Target Species
- Dog
- Species Cross-Reactivity
-
Target Species Cross Reactivity Hooded Seal - N.B. Antibody reactivity and working conditions may vary between species.
- Product Form
- Purified IgG - liquid
- Preparation
- Purified IgG prepared by affinity chromatography on Protein G from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- 0.09% sodium azide (NaN3)
- Carrier Free
- Yes
- Immunogen
- Con A activated canine peripheral blood cells.
- Approx. Protein Concentrations
- IgG concentration 1.0 mg/ml
- Fusion Partners
- Spleen cells from immunized DA rats were fused with cells of the rat Y3/Ag1.2.3 myeloma cell line.
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Flow Cytometry | 1/50 | 1/100 | |
| Immunohistology - Frozen | |||
| Immunoprecipitation |
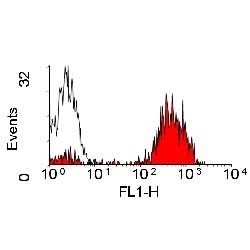
- Flow Cytometry
- Use 10μl of the suggested working dilution to label 106 cells in 100μl
References for MHC Class II Monomorphic antibody
-
Cobbold, S. & Metcalfe, S. (1994) Monoclonal antibodies that define canine homologues of human CD antigens: summary of the First International Canine Leukocyte Antigen Workshop (CLAW).
Tissue Antigens. 43 (3): 137-54. -
Watson, C.J. et al. (1994) Immunosuppression of canine renal allograft recipients by CD4 and CD8 monoclonal antibodies.
Tissue Antigens. 43 (3): 155-62. -
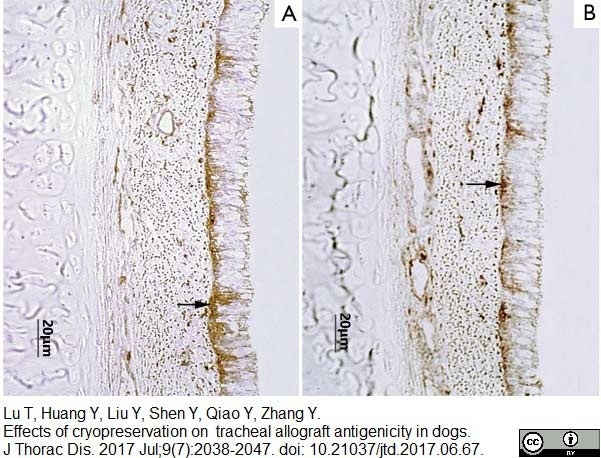
Liu, Y. et al. (2000) Immunosuppressant-free allotransplantation of the tracheaThe antigenicity of tracheal grafts can be reduced by removing the epithelium and mixed glands from the graft by detergent treatment.
J Thorac Cardiovasc Surg. 120: 108-14. -
Sanchez, M.A. et al. (2004) Organ-specific immunity in canine visceral leishmaniasis: analysis of symptomatic and asymptomatic dogs naturally infected with Leishmania chagasi.
Am J Trop Med Hyg. 70: 618-24. -
Reis, A.B. et al. (2006) Phenotypic features of circulating leucocytes as immunological markers for clinical status and bone marrow parasite density in dogs naturally infected by Leishmania chagasi.
Clin Exp Immunol.146: 303-11. -
Bonnefont-Rebeix, C. et al. (2007) Toll-like receptor 3 (TLR3): a new marker of canine monocytes-derived dendritic cells (cMo-DC).
Vet Immunol Immunopathol. 2007 Jul 15;118(1-2):134-9. -
Schütze, N. et al. (2009) Inactivated parapoxvirus ovis activates canine blood phagocytes and T lymphocytes.
Vet Microbiol. 137: 260-7. -
Bund, D. et al. (2010) Canine-DCs using different serum-free methods as an approach to provide an animal-model for immunotherapeutic strategies.
Cell Immunol. 263: 88-98.
View The Latest Product References
-
Mito, K. et al. (2010) IFNγ markedly cooperates with intratumoral dendritic cell vaccine in dog tumor models.
Cancer Res. 70: 7093-101. -
Araújo, M.S. et al. (2011) Immunological changes in canine peripheral blood leukocytes triggered by immunization with first or second generation vaccines against canine visceral leishmaniasis.
Vet Immunol Immunopathol. 141: 64-75. -
Larsen, A.K. et al. (2013) Entry and elimination of marine mammal Brucella spp. by hooded seal (Cystophora cristata) alveolar macrophages in vitro.
PLoS One. 8: e70186. -
Lin, S-C. et al. (2014) Immune Characterization of Peripheral Blood Mononuclear cells of the Dogs Restored from Innoculation of Canine Transmissible Venereal Tumor Cells.
Tai Vet J. 40 (04): 181-90. -
Constantinoiu, C.C. et al. (2015) Mucosal tolerance of the hookworm Ancylostoma caninum in the gut of naturally infected wild dogs.
Parasite Immunol. 37 (10): 510-20. -
Bonnefont-Rebeix, C. et al. (2016) Characterization of a novel canine T-cell line established from a spontaneously occurring aggressive T-cell lymphoma with large granular cell morphology.
Immunobiology. 221 (1): 12-22. -
Lu, T. et al. (2017) Effects of cryopreservation on tracheal allograft antigenicity in dogs.
J Thorac Dis. 9 (7): 2038-2047. -
Reineking, W. et al. (2018) Canine primary jejunal and colonic epithelial cells predominantly express TLR5 and TLR9 but do not change TLR expression pattern after stimulation with certain Toll-like receptor ligands.
Vet Immunol Immunopathol. 206: 16-24. -
Martini, V. et al. (2019) Prognostic role of non-neoplastic lymphocytes in lymph node aspirates from dogs with diffuse large B-cell lymphoma treated with chemo-immunotherapy.
Res Vet Sci. 125: 130-5. -
Grudzien, M. et al. (2021) A newly established canine NK-type cell line and its cytotoxic properties.
Vet Comp Oncol. 19 (3): 567-77. -
Bragato, J.P. et al. (2022) miRNA-21 regulates CD69 and IL-10 expression in canine leishmaniasis.
PLoS One. 17 (3): e0265192. -
Riccardo, F. et al. (2022) Antigen mimicry as an effective strategy to induce CSPG4-targeted immunity in dogs with oral melanoma: a veterinary trial.
J Immunother Cancer. 10 (5): e004007. -
Jaensch, S.M. et al. (2022) Clinicopathologic and immunophenotypic features in dogs with presumptive large granular lymphocyte leukaemia.
Aust Vet J. 100 (11): 527-32. -
Yang, V.K. et al. (2021) Intravenous administration of allogeneic Wharton jelly-derived mesenchymal stem cells for treatment of dogs with congestive heart failure secondary to myxomatous mitral valve disease.
Am J Vet Res. 82 (6): 487-93. -
Crain, S.K. et al. (2019) Extracellular Vesicles from Wharton's Jelly Mesenchymal Stem Cells Suppress CD4 Expressing T Cells Through Transforming Growth Factor Beta and Adenosine Signaling in a Canine Model.
Stem Cells Dev. 28 (3): 212-26. -
Martini, V. et al. (2018) A retrospective study of flow cytometric characterization of suspected extranodal lymphomas in dogs.
J Vet Diagn Invest. 30 (6): 830-6. -
Sainz, Á. et al. (2021) Effect of chemically modified tetracycline-8 (CMT-8) on hematology, blood chemistry, cytokines and peripheral blood lymphocyte subsets of healthy dogs.
Res Vet Sci. 136: 200-8. -
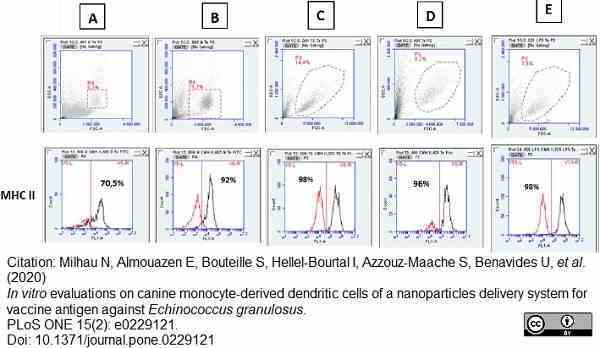
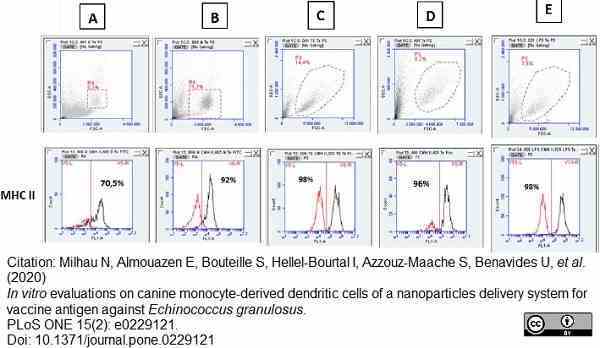
Milhau, N. et al. (2020) In vitro evaluations on canine monocyte-derived dendritic cells of a nanoparticles delivery system for vaccine antigen against Echinococcus granulosus.
PLoS One. 15 (2): e0229121. -
Sheng, R. et al. (2023) Prognostic significance of CD25 expression in dogs with a noninvasive diagnosis of B-cell lymphoma treated with CHOP chemotherapy.
Vet Comp Oncol. 21 (1): 28-35.
- RRID
- AB_11203404
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Dog ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up