Infliximab antibody | AbD17841_hIgG1






Human anti Infliximab:HRP
- Product Type
- Monoclonal Antibody
- Clone
- AbD17841_hIgG1
- Isotype
- IgG1
- Specificity
- Infliximab
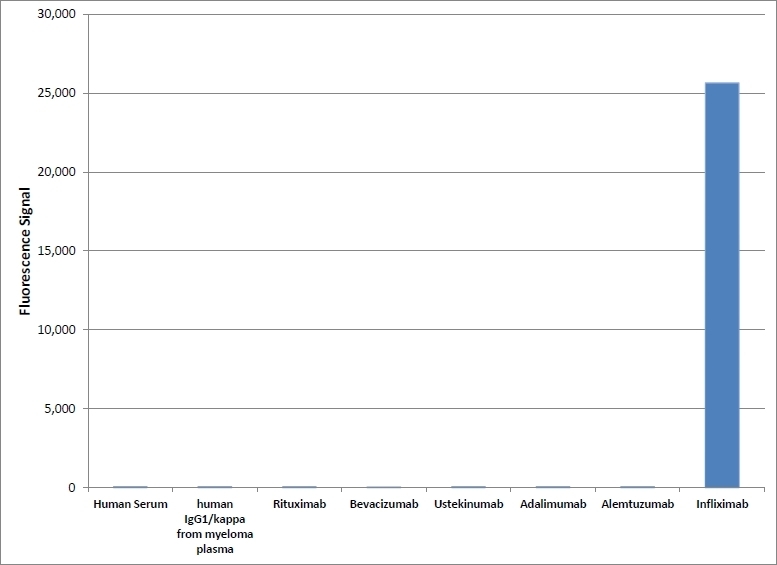
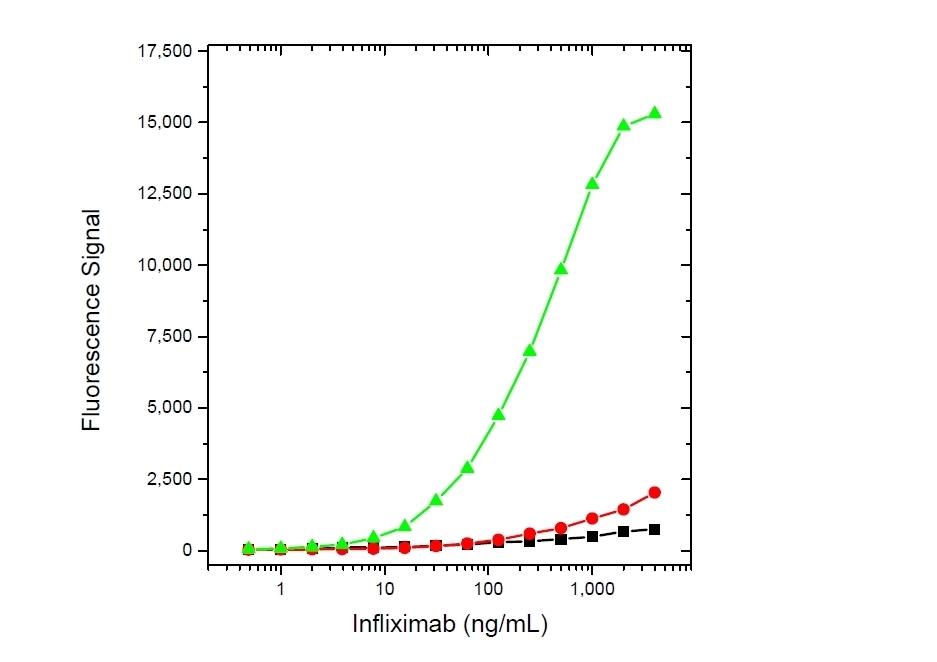
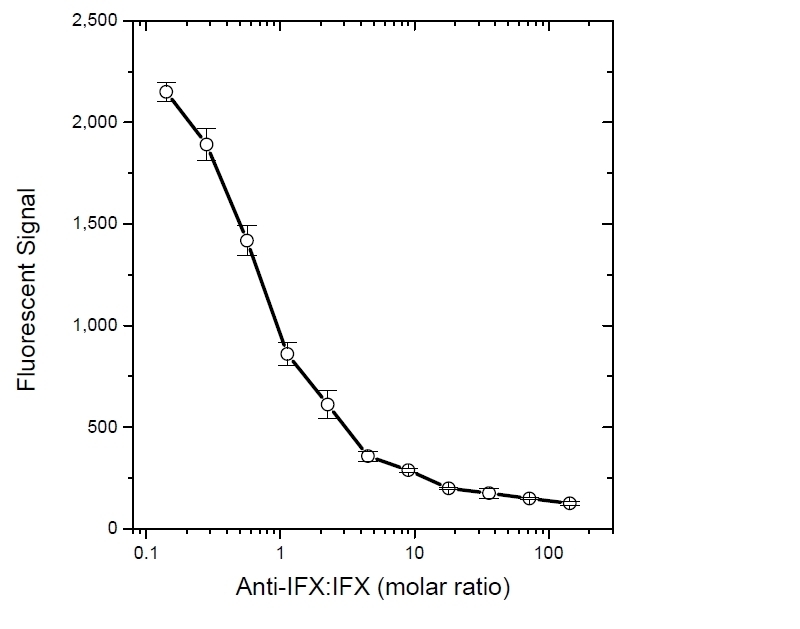
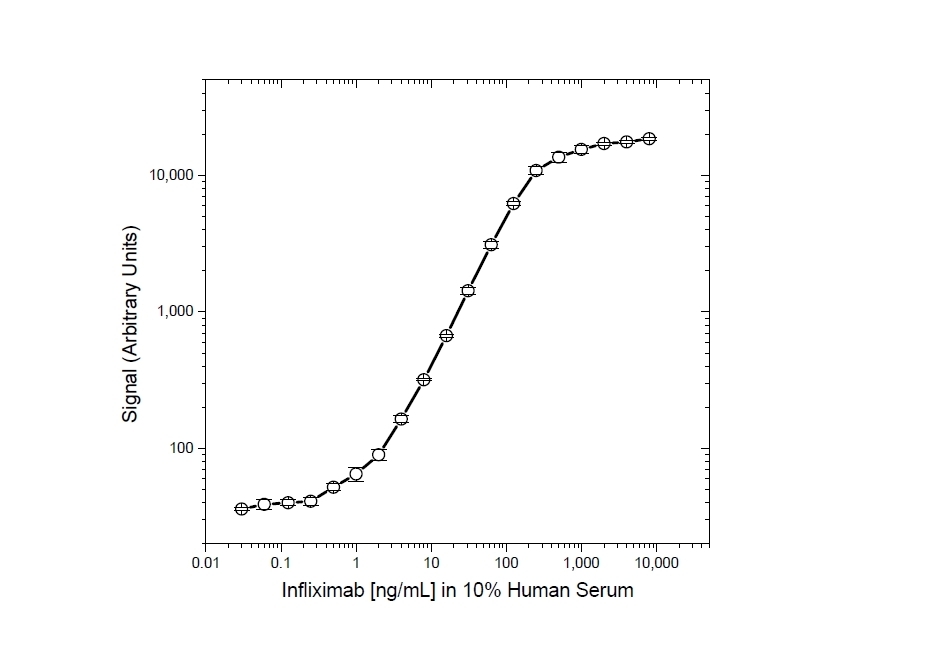
| Human Anti-Infliximab, clone AbD17841_hIgG1 is an anti-idiotypic antibody that specifically recognizes the infliximab monoclonal antibody and inhibits the binding of infliximab to its target. It can be used in bioanalytical assays to measure the levels of infliximab and biosimilar products, such as Inflectra and Renflexis, in patient samples. Human anti Infliximab, clone AbD17841_hIgG1 is a fully human antibody and may be used to develop and calibrate immune response assays to measure the anti-drug antibody (ADA) response in patient sera. In the HRP-conjugated format, clone AbD17841_IgG1 is suitable as a detection antibody in pharmacokinetic (PK) assays. Infliximab (branded as Remicade) is a chimeric monoclonal antibody drug (IgG1/kappa) that has been approved for treatment of psoriasis, Crohn's disease, ankylosing spondylitis, psoriatic arthritis, rheumatoid arthritis, and ulcerative colitis. Infliximab is directed against Tumor Necrosis Factor Alpha (TNFα) and acts by blocking the binding of this cytokine to its receptors. Infliximab also induces apoptosis of TNFα expressing T-lymphocytes. View a summary of all Anti-Infliximab Antibodies. |
|
- Product Form
- Human IgG1 antibody (kappa light chain) selected from the HuCAL phage display library and expressed in a human cell line. Conjugated to horseradish peroxidase (HRP) - liquid.
- Preparation
- Purified IgG prepared by affinity chromatography on Protein A
- Source
- HKB-11
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- 0.0095% MIT
- Immunogen
- Infliximab
- Affinity
- The monovalent intrinsic affinity of this antibody in an unconjugated format was measured as KD=1.8 nM by real time, label-free molecular interaction analysis on immobilized infliximab.
- Approx. Protein Concentrations
- Total protein concentration 1.0 mg/ml
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
- Acknowledgements
- This product and/or its use is covered by claims of U.S. patents, and/or pending U.S. and non-U.S. patent applications owned by or under license to Bio-Rad Laboratories, Inc. See bio-rad.com/en-us/trademarks for details.
Remicade is a registered trademark of Janssen Biotech Inc. Inflectra is a registered trademark of Hospira, a Pfizer Company. RENFLEXIS is a trademark of Merck Sharp & Dohme Corp.
When ready to use, thaw and aliquot the sample as needed. Store aliquots at -70°C, if available, otherwise store at -20°C. It is not recommended to keep aliquots at 4°C for more than one week.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| ELISA | 1/100 | 1/1000 |
- Technical Advice
- Recommended protocols and further information about HuCAL recombinant antibody technology can be found in the HuCAL Antibodies Technical Manual
- ELISA
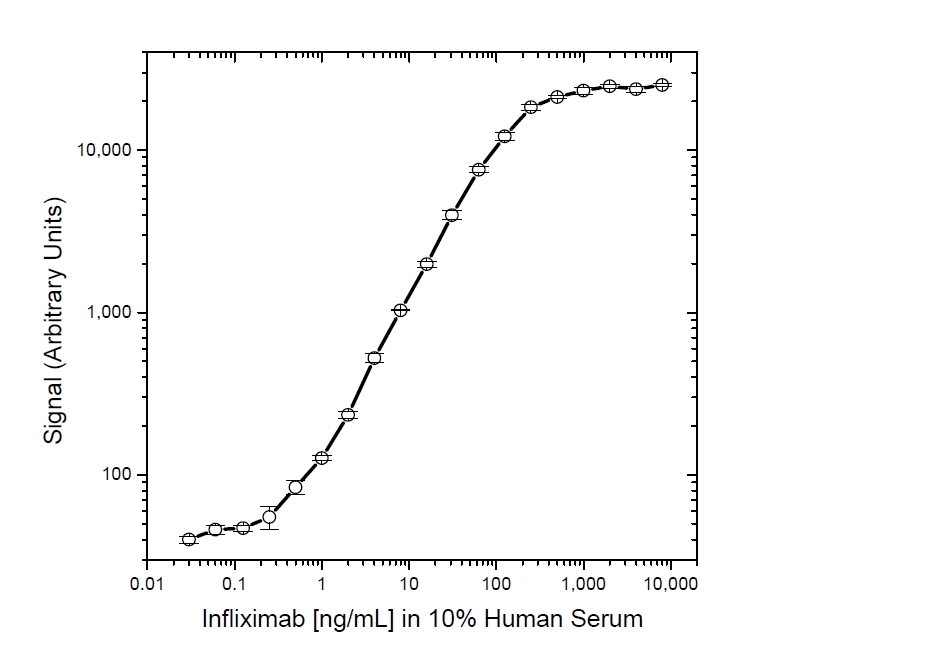
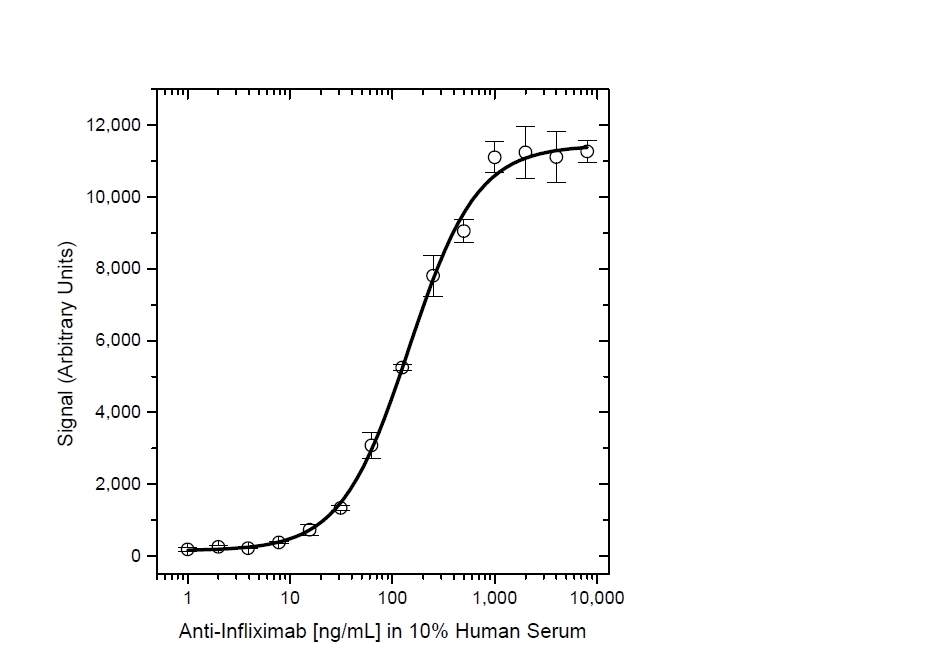
- This product may be used in as a detection antibody in an Infliximab bridging ELISA together with HCA212 as the capture reagent.
Protocol: PK bridging ELISA to measure free drug.
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Human anti Infliximab | HCA212 | E | 0.1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Human anti Infliximab | ||||||
| Human anti Infliximab | HCA214 | E | 0.1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Human anti Infliximab | ||||||
| Human anti Infliximab | HCA215 | E | 0.1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Human anti Infliximab | ||||||
| Human anti Infliximab | HCA216 | E | 0.1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Human anti Infliximab | ||||||
| Recombinant Human TNF Alpha | PHP051 | E FN WB | 50 µg | Log in | |||
| List Price | Your Price | ||||||
| Log in | |||||||
| Description | Recombinant Human TNF Alpha | ||||||
References for Infliximab antibody
-
Hernández-Breijo, B. et al. (2016) Standardization of the homogeneous mobility shift assay protocol for evaluation of anti-infliximab antibodies. Application of the method to Crohn's disease patients treated with infliximab.
Biochem Pharmacol. Sep 21. pii: S0006-2952(16)30302-1. [Epub ahead of print]
- Synonyms
- Remicade
- Licensed Use
- For in vitro. research purposes and for commercial applications for the provision of in vitro. testing services to support preclinical and clinical drug development. Any re-sale in any form or any other commercial application needs a written agreement with Bio-Rad.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
Always be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up