Collagen I antibody

Rabbit anti Rat Collagen I
- Product Type
- Polyclonal Antibody
- Isotype
- Polyclonal IgG
- Specificity
- Collagen I
| Rabbit anti Rat Collagen I antibody recognizes both native and heat denatured rat collagen type I. Collagen is located in the extracellular matrix of connective tissues. It is part of the interacting network of proteoglycans and proteins that provides a structural framework for both soft and calcified connective tissues. Type I collagen (~95 kDa) is found in bone, cornea, skin and tendon. Mutations in the encoding gene are associated with osteogenesis imperfecta (Sykes et al. 1990), Ehlers Danlos syndrome (Burrows et al. 1996), and idiopathic osteoporosis (Pernow et al. 2006). Bio-Rad currently do not support the use Rabbit anti Rat Collagen I antibody in western Blotting. However, it has been reported that this antibody has been used successfully for the quantitation of collagen I in rabbit aortic tissue by western blotting (Valero-Muñoz et al. 2014). |
- Target Species
- Rat
- Product Form
- Purified IgG - lyophilized
- Reconstitution
- Reconstitute with 500 µl of sterile DI water. Vial contains 0.05 mg of lyophilized antibody.
- Preparation
- Purified IgG prepared by affinity chromatography on immobilized rat collagen I
- Buffer Solution
- 0.1M Sodium Chloride
0.01M Sodium Phosphate
0.01M Sodium Borate - Preservative Stabilisers
- 1% Mannitol
1% Dextran - Immunogen
- Type I Collagen from rat tail tendon.
- Regulatory
- For research purposes only
- Guarantee
- Guaranteed until date of expiry. Please see product label.
After reconstitution store at -20oC.
Storage in frost-free freezers is not recommended. Avoid repeated freezing and thawing as this may denature the antibody.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| ELISA | 1/1500 | 1/6000 | |
| Immunofluorescence | 1/10 | 1/60 | |
| Immunohistology - Frozen | 1/10 | 1/60 | |
| Immunohistology - Paraffin |
- Immunohistology
- It has been reported that this antibody is suitable for use on paraffin embedded formalin fixed sections following treatment with 1 mg/ml pepsin in 0.5 M acetic acid for 2 hours at 37ºC and multiple buffer washes.
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Goat anti Rabbit IgG (Fc):Biotin | STAR121B | E WB | 1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Goat anti Rabbit IgG (Fc):Biotin | ||||||
| Goat anti Rabbit IgG (Fc):FITC | STAR121F | F | 1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Goat anti Rabbit IgG (Fc):FITC | ||||||
| Goat anti Rabbit IgG (Fc):HRP | STAR121P | E WB | 1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Goat anti Rabbit IgG (Fc):HRP | ||||||
| Goat anti Rabbit IgG (H/L):HRP | STAR124P | C E WB | 1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Goat anti Rabbit IgG (H/L):HRP | ||||||
| Sheep anti Rabbit IgG:RPE | STAR35A | F | 1 ml |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Sheep anti Rabbit IgG:RPE | ||||||
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Antigen Retrieval Buffer, pH8.0 | BUF025A | P | 500 ml | Log in | |||
| List Price | Your Price | ||||||
| Log in | |||||||
| Description | Antigen Retrieval Buffer, pH8.0 | ||||||
| TidyBlot Western Blot Detection Reagent:HRP | STAR209P | WB * | 0.5 ml | Log in | |||
| List Price | Your Price | ||||||
| Log in | |||||||
| Description | TidyBlot Western Blot Detection Reagent:HRP | ||||||
References for Collagen I antibody
-
Novotná, J. et al. (2001) Hyperoxia and recovery from hypoxia alter collagen in peripheral pulmonary arteries similarly.
Physiol Res. 50 (2): 153-63. -
Hartner A, et al. (2006) Angiotensin II formation in the kidney and nephrosclerosis in Ren-2 hypertensive rats.
Nephrol Dial Transplant. 21:1778-85. -
Klanke B, et al. (2008) Blood pressure versus direct mineralocorticoid effects on kidney inflammation and fibrosis in DOCA-salt hypertension.
Nephrol Dial Transplant. 23: 3456-63. -
Daniel, C. et al. (2009) Thrombospondin-2 therapy ameliorates experimental glomerulonephritis via inhibition of cell proliferation, inflammation, and TGF-beta activation.
Am J Physiol Renal Physiol. 297: F1299-309. -
Jones, H.B. et al. (2012) Quantitative Histopathological Assessment of Retardation of Islets of Langerhans Degeneration in Rosiglitazone-dosed Obese ZDF Rats Using Combined Insulin and Collagens (I and III) Immunohistochemistry with Automated Image Analysis and Statistical Modeling.
Toxicol Pathol. 41: 425-44. -
Aktaş, A. et al. (2013) Ankaferd-Induced Early Soft Tissue Wound Healing in an Experimental Rat Model.
Turkiye Klinikleri J Med Sci 2013;33(6): 1344-53. -
Jones, H.B. et al. (2013) Quantitative histopathological assessment of retardation of islets of langerhans degeneration in rosiglitazone-dosed obese ZDF rats using combined insulin and collagens (I and III) immunohistochemistry with automated image analysis and statistical modeling.
Toxicol Pathol. 41 (3): 425-44. -
Valero-Muñoz, M. et al. (2014) Carob pod insoluble fiber exerts anti-atherosclerotic effects in rabbits through sirtuin-1 and peroxisome proliferator-activated receptor-γ coactivator-1α.
J Nutr. 144: 1378-84.
View The Latest Product References
-
Oshita, T. et al. (2016) Adipose-Derived Stem Cells Improve Collagenase-Induced Tendinopathy in a Rat Model.
Am J Sports Med. 44 (8): 1983-9. -
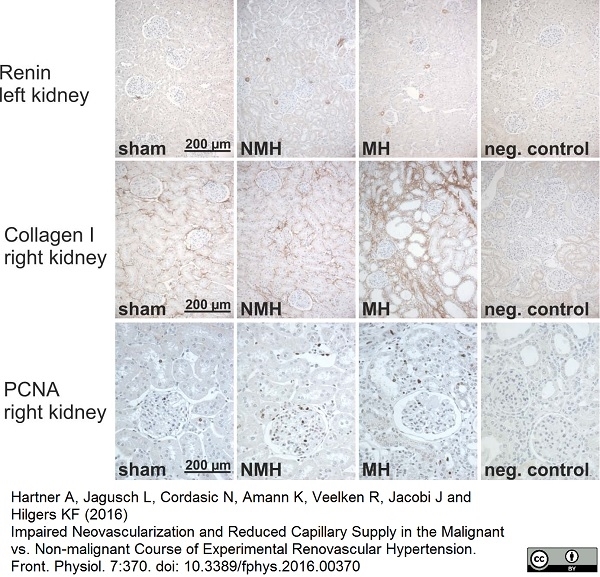
Hartner, A. et al. (2016) Impaired Neovascularization and Reduced Capillary Supply in the Malignant vs. Non-malignant Course of Experimental Renovascular Hypertension.
Front Physiol. 7: 370. -
Martínez-Martínez E et al. (2016) The lysyl oxidase inhibitor (β-aminopropionitrile) reduces leptin profibrotic effects and ameliorates cardiovascular remodeling in diet-induced obesity in rats.
J Mol Cell Cardiol. 92: 96-104. -
Dergilev, K.V. et al. (2017) Comparison of cardiac stem cell sheets detached by Versene solution and from thermoresponsive dishes reveals similar properties of constructs.
Tissue Cell. 49 (1): 64-71. -
Segreto, F. et al. (2018) The role of angiogenesis, inflammation and estrogen receptors in breast implant capsules development and remodeling.
J Plast Reconstr Aesthet Surg. 71 (5): 637-43. -
Menendez-Castro, C. et al. (2020) Correlations Between Interleukin-11 Expression and Hypertensive Kidney Injury in a Rat Model of Renovascular Hypertension.
Am J Hypertens. 33 (4): 331-40.
- RRID
- AB_620498
- UniProt
- P02454
- P02466
- Entrez Gene
- Col1a1
- Col1a2
- GO Terms
- GO:0005615 extracellular space
- GO:0009612 response to mechanical stimulus
- GO:0031960 response to corticosteroid stimulus
- GO:0042542 response to hydrogen peroxide
- GO:0043434 response to peptide hormone stimulus
- GO:0051591 response to cAMP
- GO:0071300 cellular response to retinoic acid
- GO:0071363 cellular response to growth factor stimulus
- GO:0005201 extracellular matrix structural constituent
- View More GO Terms
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Rat ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up