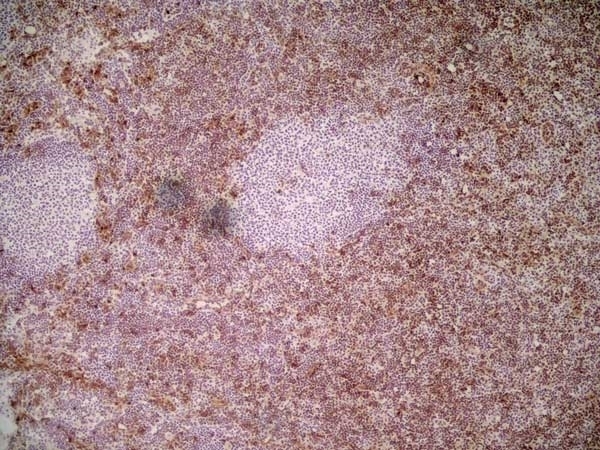
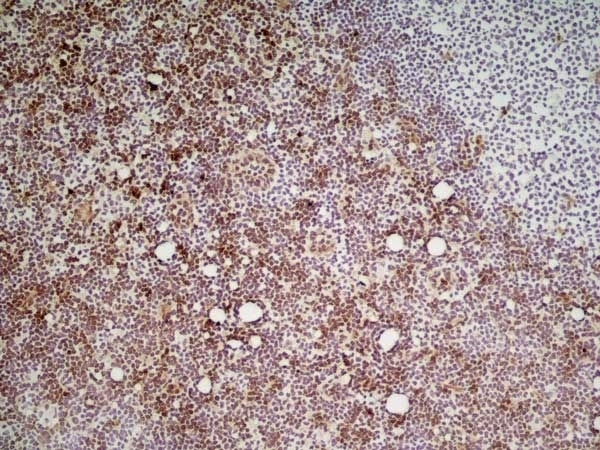
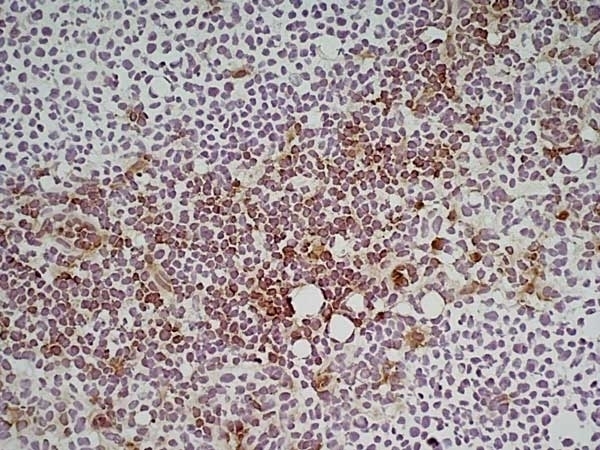
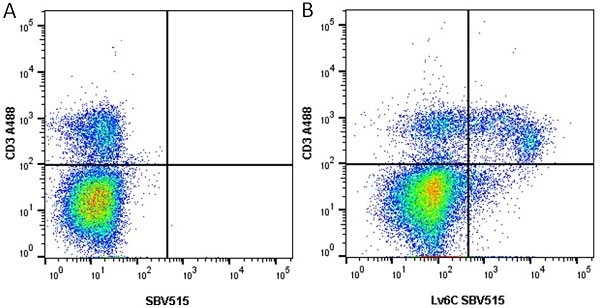
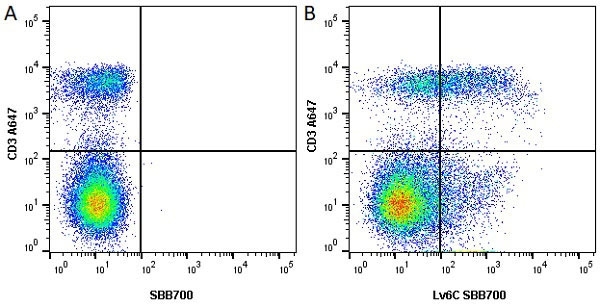
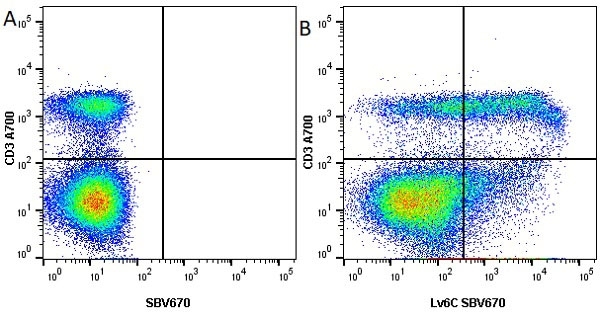
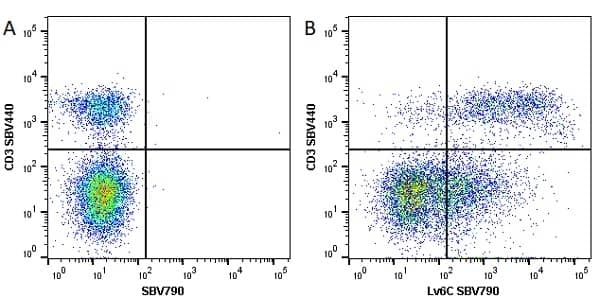
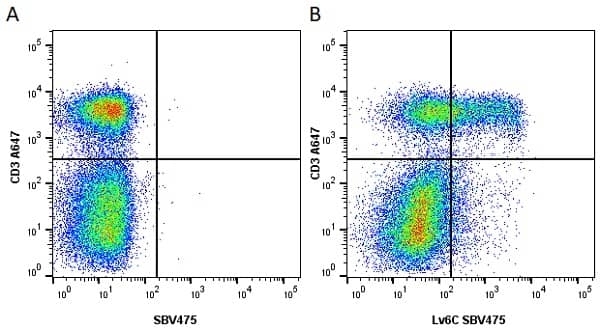
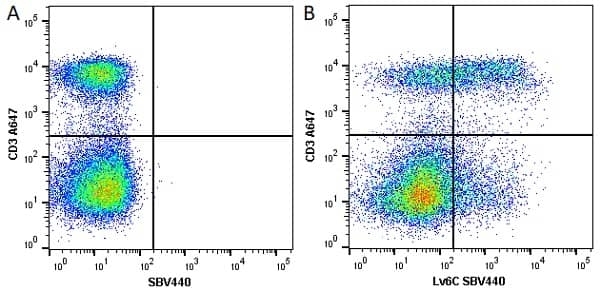
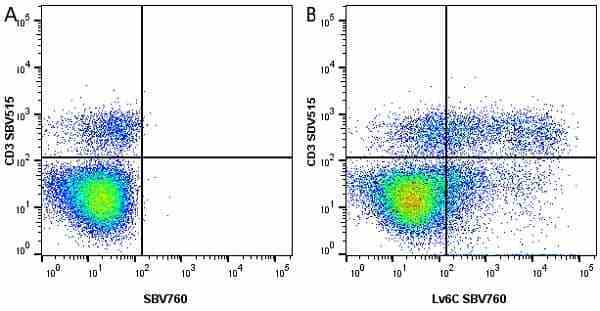
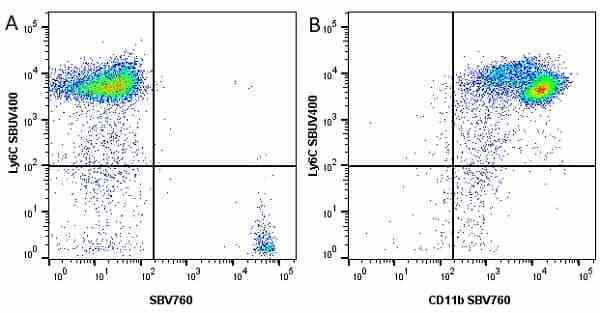
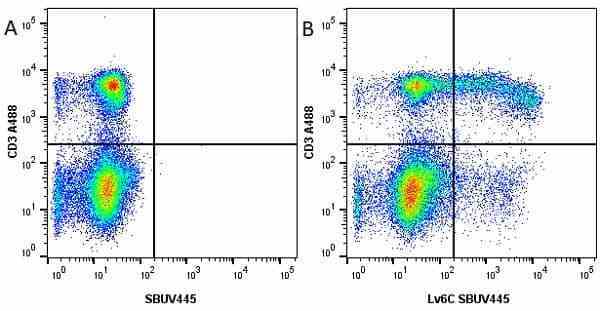
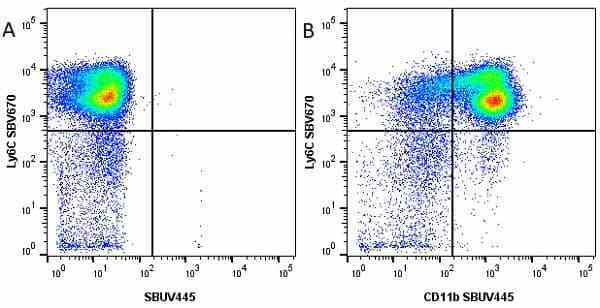
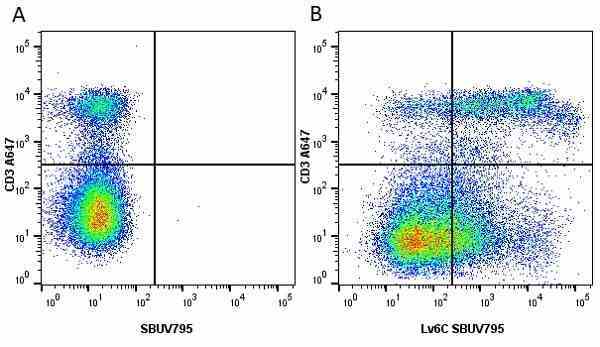
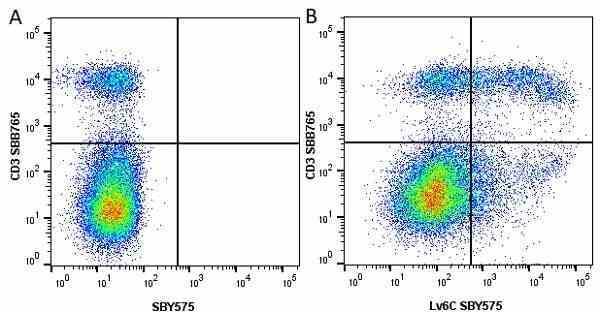
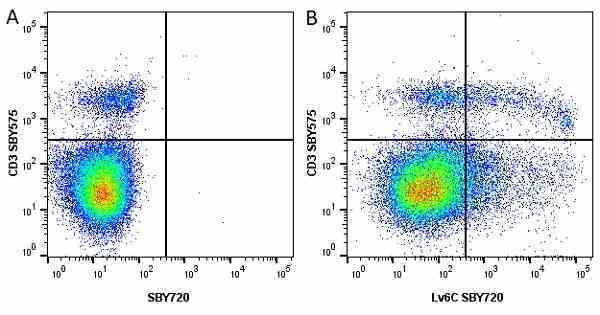
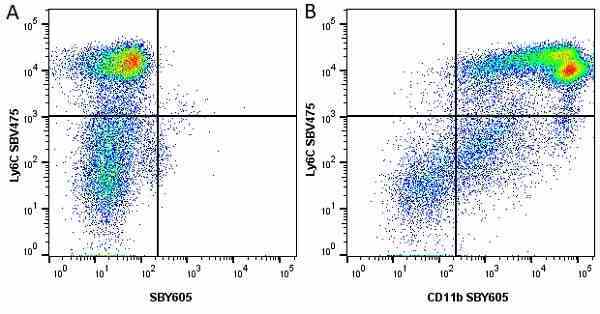
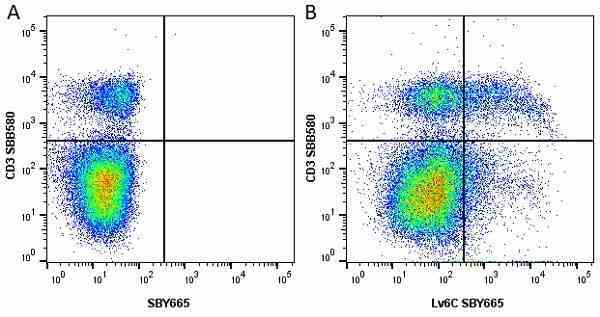
Ly-6C antibody | ER-MP20








































Rat anti Mouse Ly-6C:RPE
- Product Type
- Monoclonal Antibody
- Clone
- ER-MP20
- Isotype
- IgG2a
- Specificity
- Ly-6C
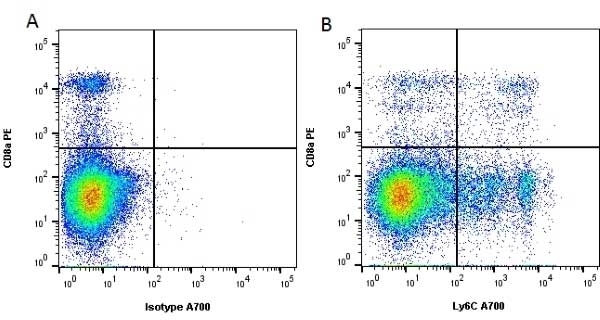
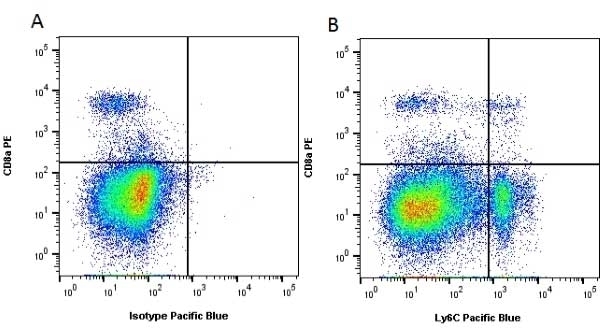
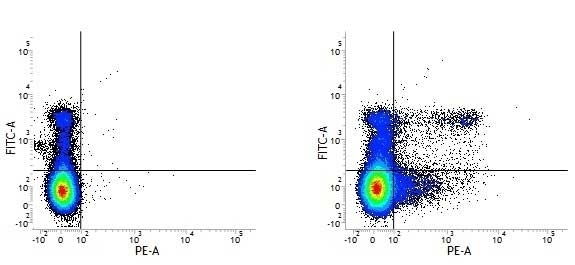
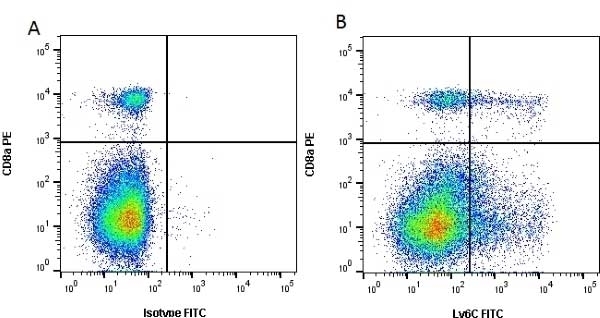
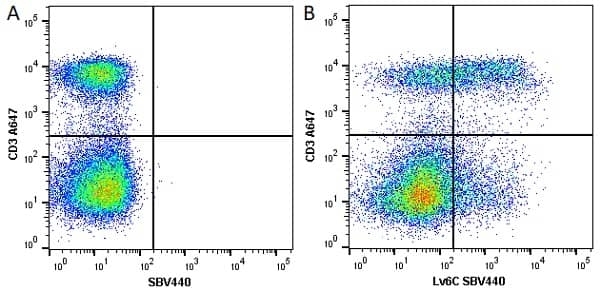
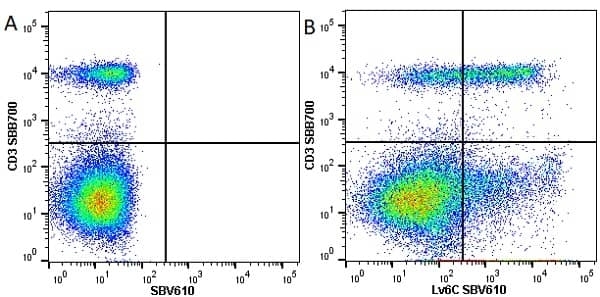
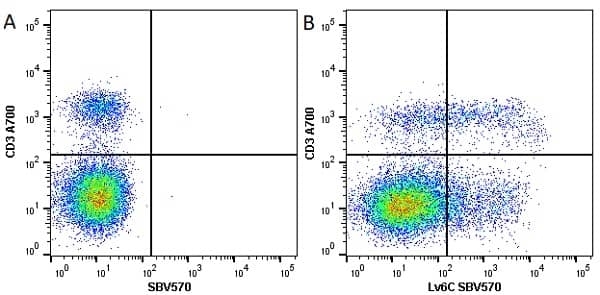
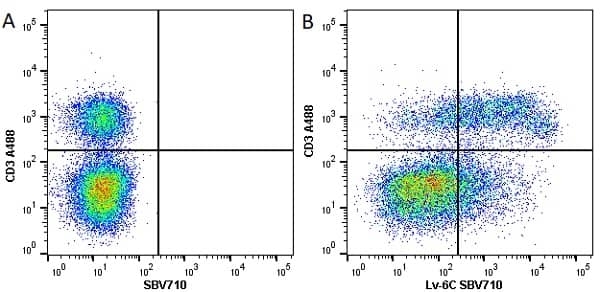
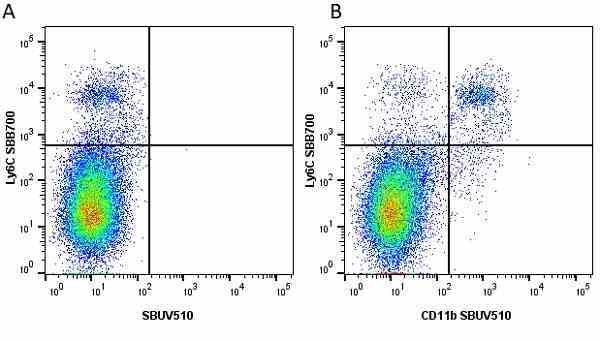
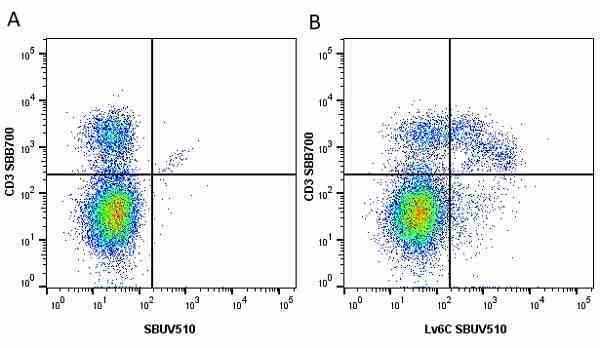
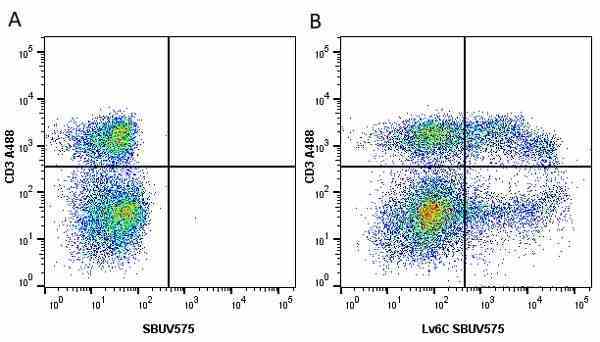
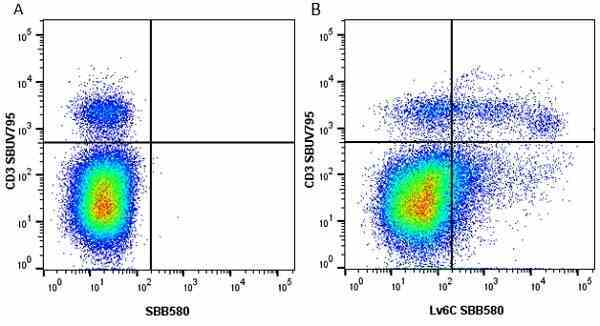
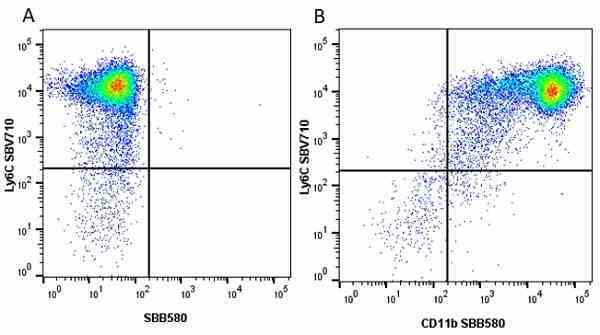
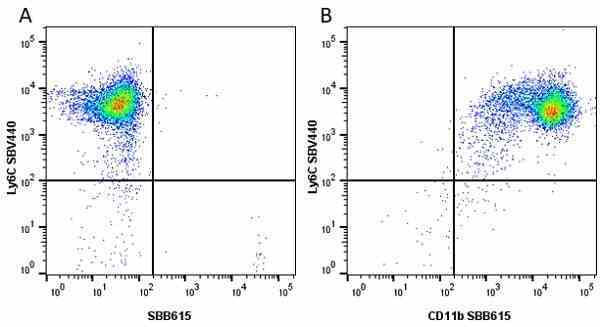
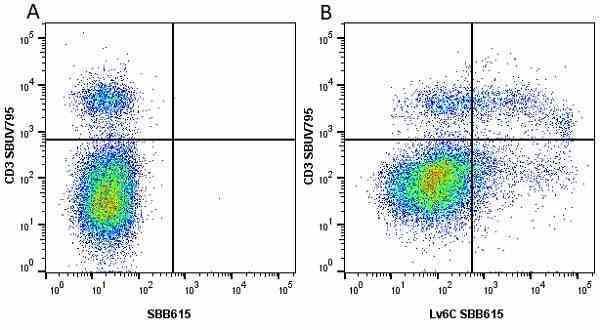
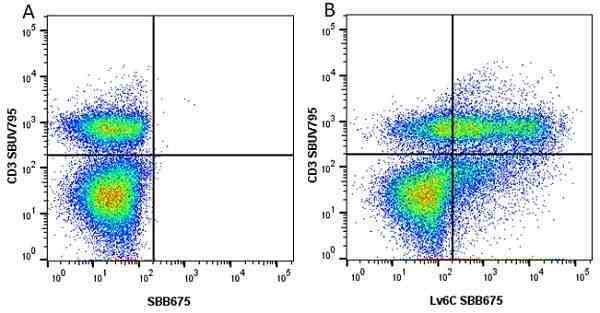
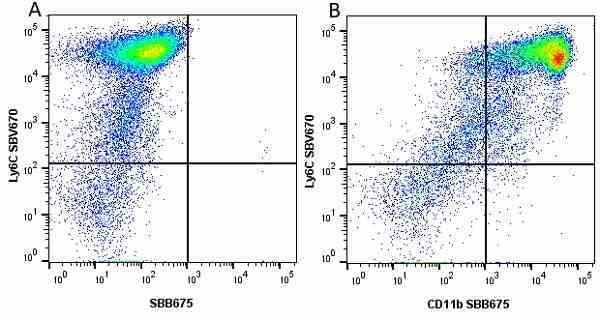
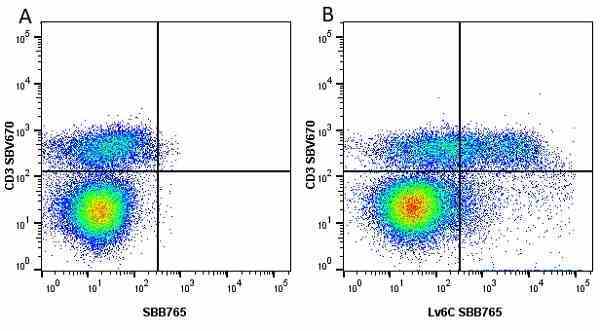
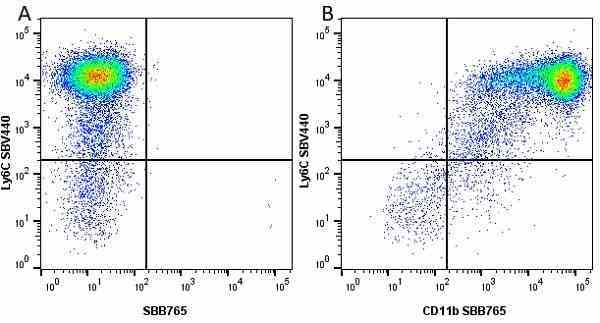
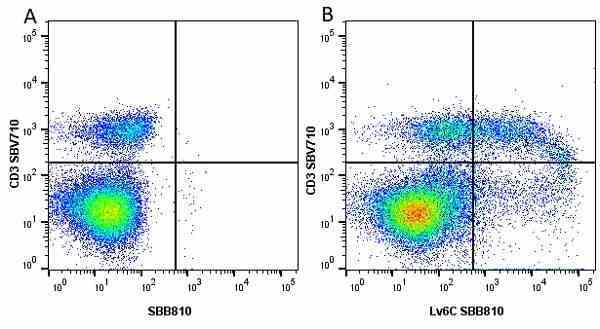
| Rat anti Mouse Ly-6C antibody, clone ER-MP20 recognizes murine Ly-6C, a 131 amino acid ~14 kDa differentiation antigen, expressed on macrophage/dendritic cell precursors in mid-stage development (late CFU-M, monoblasts and immature monocytes), granulocytes, and on a wide range of endothelial cells and subpopulations of B- and T-lymphocytes. Rat anti Mouse Ly-6C antibody, clone ER-MP20 is able to distinguish multiple mouse blood monocyte subsets: immature Ly-6Chi monocytes are recruited to acute peripheral inflammation and develop into Ly-6C+ exudate macrophages, whereas more mature Ly-6C-/lo monocytes are precursors for tissue macrophages and dendritic cells in steady state. Rat anti Mouse Ly-6C, clone ER-MP20 can be used in conjunction with clone ER-MP12 in two colour flow cytometric analysis, to identify different stages of myeloid progenitor cells in mouse bone marrow (Leenen et al. 1990). Rat anti Mouse Ly-6C was originally described as recognizing a protein encoded by the LY6C gene. It has subsequently become apparent that the LY6C locus demonstrates polymorphism and the LY6C gene has been re-designated LY6C2. The LY6C1 gene encodes a similar protein with ~95% sequence homology to LY6C2. |
- Target Species
- Mouse
- Product Form
- Purified IgG conjugated to R. Phycoerythrin (RPE) - lyophilized
- Reconstitution
- Reconstitute with 1 ml distilled water
- Preparation
- Purified IgG prepared by affinity chromatography on Protein G from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- 0.09% sodium azide (NaN3)
1% bovine serum albumin
5% sucrose - Immunogen
- Balb/c macrophage precursor cell hybrids.
- Fusion Partners
- Spleen cells from immunized rats were fused with cells of the Y3-Ag1.2.3 myeloma cell line.
- Max Ex/Em
-
Fluorophore Excitation Max (nm) Emission Max (nm) RPE 488nm laser 496 578 - Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
DO NOT FREEZE.
This product should be stored undiluted. This product is photosensitive and should be protected from light. Should this product contain a precipitate we recommend microcentrifugation before use.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Flow Cytometry | Neat | 1/5 |
How to Use the Spectraviewer
Watch the Tool Tutorial Video ▸- Start by selecting the application you are interested in, with the option to select an instrument from the drop down menu or create a customized instrument
- Select the fluorophores or fluorescent proteins you want to include in your panel to check compatibility
- Select the lasers and filters you wish to include
- Select combined or multi-laser view to visualize the spectra
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Rat IgG2a Negative Control:RPE | MCA1212PE | F | 100 Tests | Log in | |||
| List Price | Your Price | ||||||
| Log in | |||||||
| Description | Rat IgG2a Negative Control:RPE | ||||||
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Mouse Seroblock FcR | BUF041A | F | 0.1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse Seroblock FcR | ||||||
| Mouse Seroblock FcR | BUF041B | F | 0.5 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse Seroblock FcR | ||||||
References for Ly-6C antibody
-
Zhang, Y. & Bliska, J.B. (2010) YopJ-promoted cytotoxicity and systemic colonization are associated with high levels of murine interleukin-18, gamma interferon, and neutrophils in a live vaccine model of Yersinia pseudotuberculosis infection.
Infect Immun 78: 2329-41. -
Leenen, P.J. et al. (1990) Murine macrophage precursor characterization. II. Monoclonal antibodies against macrophage precursor antigens.
Eur J Immunol. 20 (1): 27-34. -
de Bruijn, M.F. et al. (1998) Bone marrow cellular composition in Listeria monocytogenes infected mice detected using ER-MP12 and ER-MP20 antibodies: a flow cytometric alternative to differential counting.
J Immunol Methods. 217 (1-2): 27-39. -
Schatteman, G.C. et al. (2010) Lin- Cells Mediate Tissue Repair by Regulating MCP-1/CCL-2.
Am J Pathol. 177: 2002-10. -
Baumeister, T. et al. (2003) Interleukin-3Ralpha+ myeloid dendritic cells and mast cells develop simultaneously from different bone marrow precursors in cultures with interleukin-3.
J Invest Dermatol. 121: 280-8. -
Devey, L. et al. (2009) Tissue-resident macrophages protect the liver from ischemia reperfusion injury via a heme oxygenase-1-dependent mechanism.
Mol Ther. 17: 65-72. -
Nikolic, T. et al. (2003) Developmental stages of myeloid dendritic cells in mouse bone marrow.
Int Immunol. 15: 515-24. -
Wynn, A.A. et al. (2001) Role of granulocyte/macrophage colony-stimulating factor in zymocel-induced hepatic granuloma formation.
Am J Pathol. 158 (1): 131-45.
View The Latest Product References
-
Lesokhin, A.M. et al. (2012) Monocytic CCR2+ Myeloid-Derived Suppressor Cells Promote Immune Escape by Limiting Activated CD8 T-cell Infiltration into the Tumor Microenvironment.
Cancer Res. 72: 876-86. -
Chan, J. et al. (1998) Macrophage lineage cells in inflammation: characterization by colony-stimulating factor-1 (CSF-1) receptor (c-Fms), ER-MP58, and ER-MP20 (Ly-6C) expression.
Blood. 92: 1423-31. -
van Rijt, L.S. et al. (2002) Allergen-induced accumulation of airway dendritic cells is supported by an increase in CD31(hi)Ly-6C(neg) bone marrow precursors in a mouse model of asthma.
Blood. 100: 3663-71. -
Arnardottir, H.H.et al. (2012) Dietary Fish Oil Decreases the Proportion of Classical Monocytes in Blood in Healthy Mice but Increases Their Proportion upon Induction of Inflammation.
J Nutr. 142: 803-8. -
Henkel, G. et al. (1999) Commitment to the monocytic lineage occurs in the absence of the transcription factor PU.1.
Blood. 93:2849-58. -
Bossaller, L. et al. (2013) Overexpression of membrane-bound fas ligand (CD95L) exacerbates autoimmune disease and renal pathology in pristane-induced lupus.
J Immunol. 191: 2104-14. -
Garcia, J.A. et al. (2013) Regulation of adaptive immunity by the fractalkine receptor during autoimmune inflammation.
J Immunol. 191: 1063-72. -
Stijlemans, B. et al. (2015) Murine Liver Myeloid Cell Isolation Protocol
BIO-PROTOCOL. 5 (10). -
Damya, L. et al. (2014) Purification of Tumor-Associated Macrophages (TAM) and Tumor-Associated Dendritic Cells (TADC)
BIO-PROTOCOL. 4 (22). -
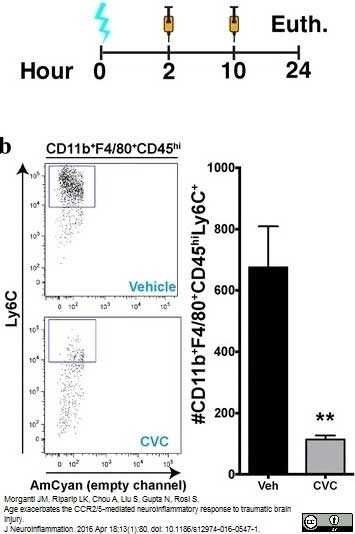
Morganti, J.M. et al. (2016) Age exacerbates the CCR2/5-mediated neuroinflammatory response to traumatic brain injury.
J Neuroinflammation. 13 (1): 80. -
Mooney, J.E. et al. (2010) Cellular plasticity of inflammatory myeloid cells in the peritoneal foreign body response.
Am J Pathol. 176 (1): 369-80. -
Iwasaki, Y. et al. (2011) In situ proliferation and differentiation of macrophages in dental pulp.
Cell Tissue Res. 346 (1): 99-109. -
Movahedi, K. et al. (2012) Nanobody-based targeting of the macrophage mannose receptor for effective in vivo imaging of tumor-associated macrophages.
Cancer Res. 72 (16): 4165-77. -
Ribechini, E. et al. (2009) Gr-1 antibody induces STAT signaling, macrophage marker expression and abrogation of myeloid-derived suppressor cell activity in BM cells.
Eur J Immunol. 39 (12): 3538-51. -
Bossaller, L. et al. (2016) TLR9 Deficiency Leads to Accelerated Renal Disease and Myeloid Lineage Abnormalities in Pristane-Induced Murine Lupus.
J Immunol. 197 (4): 1044-53. -
Barnes, M.A. et al. (2015) Macrophage migration inhibitory factor is required for recruitment of scar-associated macrophages during liver fibrosis.
J Leukoc Biol. 97 (1): 161-9. -
Ohnishi, K. et al. (2012) Immunohistochemical detection of possible cellular origin of hepatic histiocytic sarcoma in mice.
J Clin Exp Hematop. 52 (3): 171-7. -
Van den Bossche. J. et al. (2012) Claudin-1, claudin-2 and claudin-11 genes differentially associate with distinct types of anti-inflammatory macrophages in vitro and with parasite- and tumour-elicited macrophages in vivo.
Scand J Immunol. 75 (6): 588-98. -
Houthuys, E. et al. (2010) A method for the isolation and purification of mouse peripheral blood monocytes.
J Immunol Methods. 359 (1-2): 1-10. -
Greifenberg, V. et al. (2009) Myeloid-derived suppressor cell activation by combined LPS and IFN-gamma treatment impairs DC development.
Eur J Immunol. 39 (10): 2865-76. -
Cardona, S.M. et al. (2015) Disruption of Fractalkine Signaling Leads to Microglial Activation and Neuronal Damage in the Diabetic Retina.
ASN Neuro. 7 (5):1759091415608204. -
Waddell, A. et al. (2011) Colonic eosinophilic inflammation in experimental colitis is mediated by Ly6C(high) CCR2(+) inflammatory monocyte/macrophage-derived CCL11.
J Immunol. 186 (10): 5993-6003. -
Robbie, S.J. et al. (2016) Enhanced Ccl2-Ccr2 signaling drives more severe choroidal neovascularization with aging.
Neurobiol Aging. 40: 110-9. -
Cao, Y. et al. (2016) IL-1β differently stimulates proliferation and multinucleation of distinct mouse bone marrow osteoclast precursor subsets.
J Leukoc Biol. 100 (3): 513-23. -
Cao, Y. et al. (2017) TNF-α has both stimulatory and inhibitory effects on mouse monocyte-derived osteoclastogenesis.
J Cell Physiol. 232 (12): 3273-85. -
Khedoe, P.P.S.J. et al. (2017) Acute and chronic effects of treatment with mesenchymal stromal cells on LPS-induced pulmonary inflammation, emphysema and atherosclerosis development.
PLoS One. 12 (9): e0183741. -
Koohy, H. et al. (2018) Genome organization and chromatin analysis identify transcriptional downregulation of insulin-like growth factor signaling as a hallmark of aging in developing B cells.
Genome Biol. 19 (1): 126. -
Pluijmert, N.J. et al. (2020) Effects on cardiac function, remodeling and inflammation following myocardial ischemia-reperfusion injury or unreperfused myocardial infarction in hypercholesterolemic APOE*3-Leiden mice.
Sci Rep. 10 (1): 16601. -
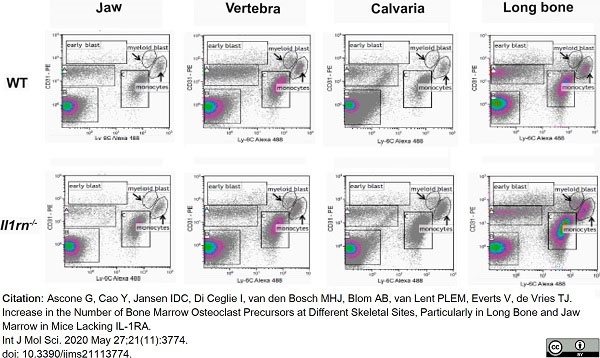
Ascone, G. et al. (2020) Increase in the Number of Bone Marrow Osteoclast Precursors at Different Skeletal Sites, Particularly in Long Bone and Jaw Marrow in Mice Lacking IL-1RA.
Int J Mol Sci. 21 (11): 3774. -
Pluijmert, N.J. et al. (2021) Phosphorylcholine antibodies restrict infarct size and left ventricular remodelling by attenuating the unreperfused post-ischaemic inflammatory response.
J Cell Mol Med. 25 (16): 7772-82. -
Njock, M-K. (2022) Endothelial extracellular vesicles promote tumour growth by tumour-associated macrophage reprogramming
J Extracell Vesicles 2022 Jun;11(6):e12228. -
Vainchtein, I.D. et al. (2023) Characterizing microglial gene expression in a model of secondary progressive multiple sclerosis.
Glia. 71 (3): 588-601. -
Mielczarek, O. et al. (2023) Intra- and interchromosomal contact mapping reveals the Igh locus has extensive conformational heterogeneity and interacts with B-lineage genes.
Cell Rep 42 (9):113074. -
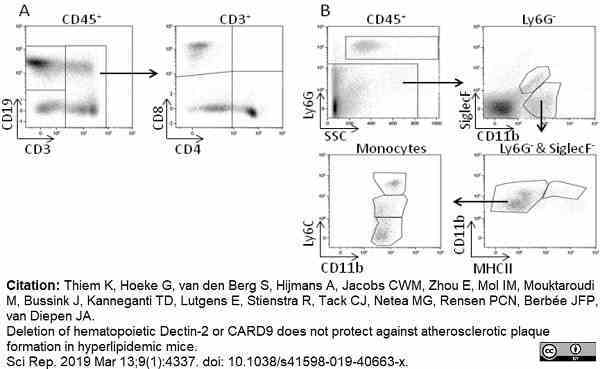
Thiem, K. et al. (2019) Deletion of hematopoietic Dectin-2 or CARD9 does not protect against atherosclerotic plaque formation in hyperlipidemic mice.
Sci Rep. 9 (1): 4337. -
Heissig, B. et al. (2022) siRNA against CD40 delivered via a fungal recognition receptor ameliorates murine acute graft-versus-host disease.
EJHaem. 3 (3): 849-61. -
Cardona, S.M. et al. (2018) Role of the Fractalkine Receptor in CNS Autoimmune Inflammation: New Approach Utilizing a Mouse Model Expressing the Human CX3CR1(I249/M280) Variant.
Front Cell Neurosci. 12: 365.
- Synonyms
- Lymphocyte Antigen 6C2
- RRID
- AB_2234744
- UniProt
- P0CW03
MCA2389PE
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Mouse ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up