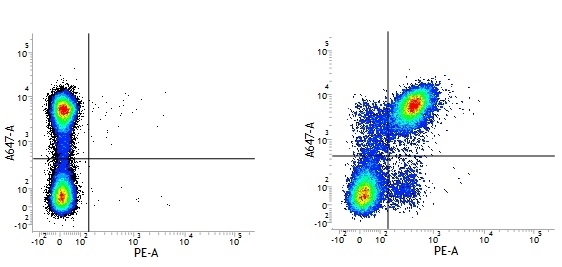
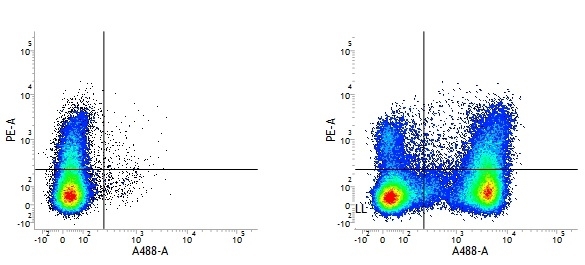
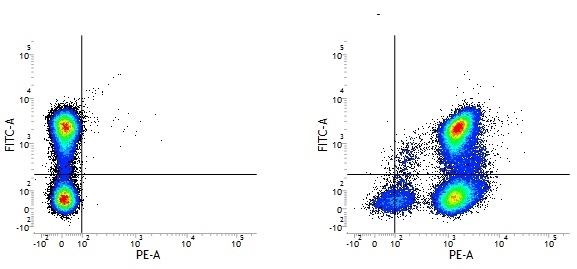
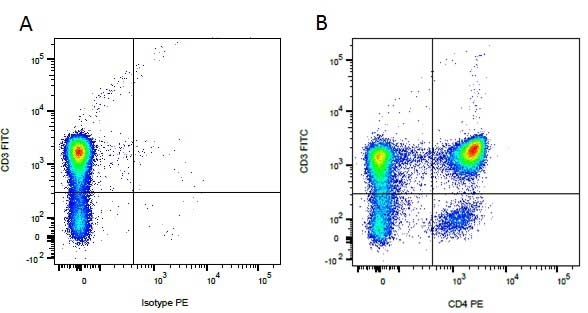
Rat IgG2a Negative Control antibody





Rat IgG2a Negative Control:RPE
- Product Type
- Negative/Isotype Control
- Isotype
- IgG2a
- Specificity
- Rat IgG2a Negative Control
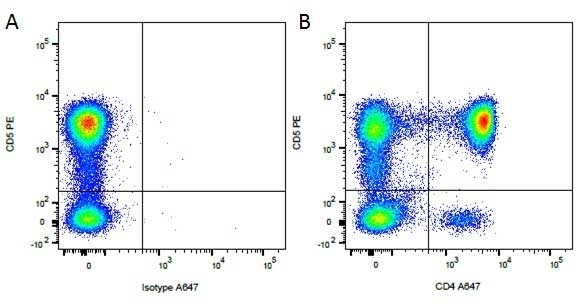
| Rat IgG2a Negative Control antibody is suitable for the assessment of the level of non-specific binding of rat IgG2a monoclonal antibodies to mouse cells. Test results indicate Rat IgG2a Negative Control antibody is also suitable for use as a negative control with canine cells. N.B. This antibody recognizes a human cell surface marker, and therefore is not suitable as a negative control in human cells or cell lines. |

|
- Target Species
- Negative Control
- Product Form
- Purified IgG conjugated to R. Phycoerythrin (RPE) - lyophilized
- Reconstitution
- Reconstitute with 1 ml distilled water
- Preparation
- Purified IgG prepared by affinity chromatography on Protein G from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- 0.09% sodium azide (NaN3)
1% bovine serum albumin
5% sucrose - Immunogen
- Human lymphocytes.
- Fusion Partners
- Spleen cells from immunized DA rats were fused with cells of the rat Y3/Ag1.2.3. myeloma cell line.
- Max Ex/Em
-
Fluorophore Excitation Max (nm) Emission Max (nm) RPE 488nm laser 496 578 - Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
DO NOT FREEZE.
This product should be stored undiluted. This product is photosensitive and should be protected from light. Should this product contain a precipitate we recommend microcentrifugation before use.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Flow Cytometry | * |
- Flow Cytometry
- Use 10μl of the suggested working dilution to label 106 cells in 100μl
How to Use the Spectraviewer
Watch the Tool Tutorial Video ▸- Start by selecting the application you are interested in, with the option to select an instrument from the drop down menu or create a customized instrument
- Select the fluorophores or fluorescent proteins you want to include in your panel to check compatibility
- Select the lasers and filters you wish to include
- Select combined or multi-laser view to visualize the spectra
References for Rat IgG2a Negative Control antibody
-
Stapleton, T.W. et al. (2000) Investigation of the regenerative capacity of an acellular porcine medial meniscus for tissue engineering applications.
Tissue Eng Part A. 17: 231-42. -
Guilloteau, L.A. et al. (2003) Nramp1 is not a major determinant in the control of Brucella melitensis infection in mice.
Infect Immun. 71: 621-8. -
Sumagin, R. et al. (2008) Leukocyte-endothelial cell interactions are linked to vascular permeability via ICAM-1-mediated signaling.
Am J Physiol Heart Circ Physiol. 295: H969-H977. -
McConnell, M.J. et al. (2009) H2-K(b) and H2-D(b) regulate cerebellar long-term depression and limit motor learning.
Proc Natl Acad Sci U S A. 106: 6784-9. -
Chiu, W.C. et al. (2011) Effects of dietary fish oil supplementation on cellular adhesion molecule expression and tissue myeloperoxidase activity in hypercholesterolemic mice with sepsis.
J Nutr Biochem. 20: 254-60. -
Schmidt, E.P. et al. (2012) The pulmonary endothelial glycocalyx regulates neutrophil adhesion and lung injury during experimental sepsis.
Nat Med. 18 (8): 1217-23. -
Park, S.W. et al. (2012) A1 adenosine receptor allosteric enhancer PD-81723 protects against renal ischemia-reperfusion injury.
Am J Physiol Renal Physiol. 303: F721-32. -
Rabadi, M. et al. (2016) Peptidyl arginine deiminase-4-deficient mice are protected against kidney and liver injury after renal ischemia and reperfusion.
Am J Physiol Renal Physiol. 311 (2): F437-49.
View The Latest Product References
-
Rabadi, M.M. et al. (2019) Peptidyl arginine deiminase-4 exacerbates ischemic AKI by finding NEMO.
Am J Physiol Renal Physiol. 316 (6): F1180-F1190. -
Han, S.J. et al. (2020) Renal proximal tubular NEMO plays a critical role in ischemic acute kidney injury.
JCI Insight. 5 (19): e139246. -
Ono, Y. et al. (2018) CD11c+ M1-like macrophages (Møs) but not CD206+ M2-like Mø are involved in folliculogenesis in mice ovary.
Sci Rep. 8 (1): 8171.
- RRID
- AB_322676
MCA1212PE
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Negative Control ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up