CD4 antibody | KEN-4






Mouse anti Rabbit CD4:FITC
- Product Type
- Monoclonal Antibody
- Clone
- KEN-4
- Isotype
- IgG2a
- Specificity
- CD4
| Mouse anti Rabbit CD4 antibody, clone KEN-4 recognizes the rabbit CD4 cell surface antigen, also known as T-cell surface antigen T4/Leu-3. Rabbit CD4 is a 434 amino acid, with an additional N-terminal signal peptide ~50 kDa cell surface single pass, type I transmembrane glycoprotein expressed by T helper cells. Mouse anti Rabbit CD4 antibody, clone KEN-4 blocks the allogeneic mixed lymphocyte reaction response. |
- Target Species
- Rabbit
- Species Cross-Reactivity
-
Target Species Cross Reactivity Brown Hare (Lepus europeus) - N.B. Antibody reactivity and working conditions may vary between species.
- Product Form
- Purified IgG conjugated to Fluorescein Isothiocyanate Isomer 1 (FITC) - liquid
- Preparation
- Purified IgG prepared by affinity chromatography on Protein A from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
0.09% Sodium Azide 1% Bovine Serum Albumin - Immunogen
- Rabbit thymocytes.
- Approx. Protein Concentrations
- IgG concentration 0.1 mg/ml
- Fusion Partners
- Spleen cells from immunized mice were fused with cells of the mouse PAI myeloma cell line.
- Max Ex/Em
-
Fluorophore Excitation Max (nm) Emission Max (nm) FITC 490 525 - Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended. This product is photosensitive and should be protected from light.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Flow Cytometry | Neat |
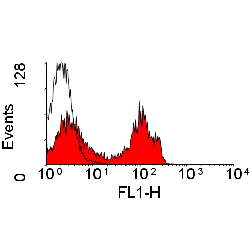
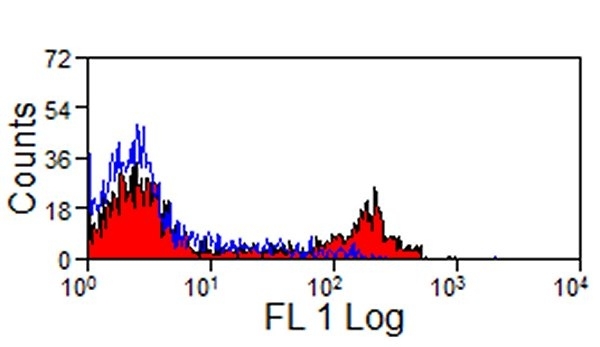
- Flow Cytometry
- Use 10ul of the suggested working dilution to label 106 cells or 100ul whole blood.
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Mouse IgG2a Negative Control:FITC | MCA929F | F | 100 Tests |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse IgG2a Negative Control:FITC | ||||||
Source Reference
-
Kotani, M. et al. (1993) Generation and characterization of monoclonal antibodies against rabbit CD4, CD5 and CD11a antigens.
J Immunol Methods. 157 (1-2): 241-52.
References for CD4 antibody
-
Perosa, F. and Dammacco, F. (1994) Anti-idiotypic monoclonal antibodies (mAb) to an anti-CD4 mAb induce CD4+ T cell depletion in rabbit.
Int J Clin Lab Res. 24: 208-12. -
Renaux, S. et al. (2003) Dynamics and responsiveness of T-lymphocytes in secondary lymphoid organs of rabbits developing immunity to Eimeria intestinalis.
Vet Parasitol. 110 (3-4): 181-95. -
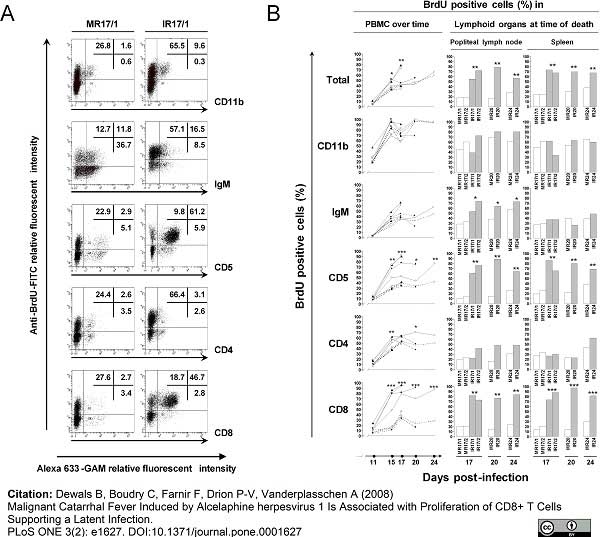
Dewals, B. et al. (2008) Malignant catarrhal fever induced by alcelaphine herpesvirus 1 is associated with proliferation of CD8+ T cells supporting a latent infection.
PLos ONE 3: e1627. -
Pakandl, M. et al. (2008) Dependence of the immune response to coccidiosis on the age of rabbit suckling.
Parasitol Res. 103 (6): 1265-71. -
Yang, J. et al. (2009) Expression and localization of rabbit B-cell activating factor (BAFF) and its specific receptor BR3 in cells and tissues of the rabbit immune system.
Dev Comp Immunol. 33 (5): 697-708. -
Chentoufi, A.A. et al. (2010) A novel HLA (HLA-A*0201) transgenic rabbit model for preclinical evaluation of human CD8+ T cell epitope-based vaccines against ocular herpes.
J Immunol. 184: 2561-71. -
Rütgen, B.C. et al. (2014) Exploratory assessment of CD4+ T lymphocytes in brown hares (Lepus europeus) using a cross-reactive anti-rabbit CD4 antibody.
Vet Immunol Immunopathol. 161 (1-2): 108-15. -
Parameswaran, N. et al. (2014) The A2 gene of alcelaphine herpesvirus-1 is a transcriptional regulator affecting cytotoxicity in virus-infected T cells but is not required for malignant catarrhal fever induction in rabbits.
Virus Res. 188: 68-80.
View The Latest Product References
-
Boutard, B. et al. (2015) The α2,3-sialyltransferase encoded by myxoma virus is a virulence factor that contributes to immunosuppression.
PLoS One. 10 (2): e0118806. -
Khan, A.A. et al. (2015) Therapeutic immunization with a mixture of herpes simplex virus 1 glycoprotein D-derived “asymptomatic” human CD8+ T-cell epitopes decreases spontaneous ocular shedding in latently infected HLA transgenic rabbits: association with low frequency of local PD-1+ TIM-3+ CD8+ exhausted T cells.
J Virol. 89 (13): 6619-32. -
Myster, F. et al. (2015) Viral semaphorin inhibits dendritic cell phagocytosis and migration but is not essential for gammaherpesvirus-induced lymphoproliferation in malignant catarrhal fever.
J Virol. 89 (7): 3630-47. -
Beghelli, D et al. (2016) Effects of Oregano (Origanum vulgare L.) and Rosemary (Rosmarinus officinalis L.) Aqueous Extracts On in vitro Rabbit Immune Responses
MOJ Immunology. 4 (4) [Epub ahead of print]. -
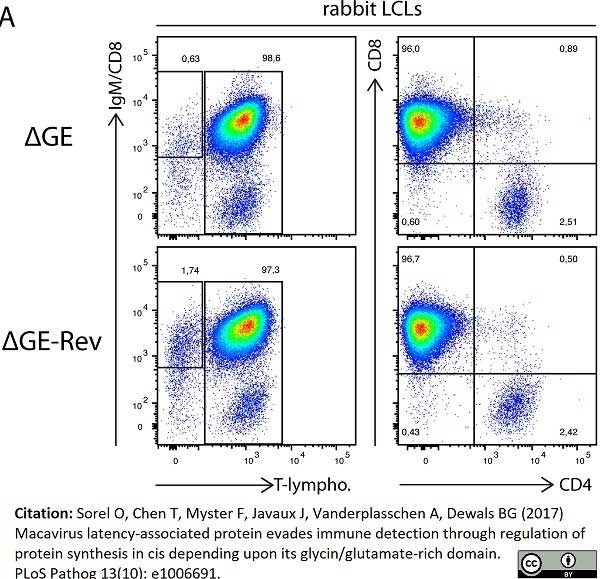
Sorel, O. et al. (2017) Macavirus latency-associated protein evades immune detection through regulation of protein synthesis in cis depending upon its glycin/glutamate-rich domain.
PLoS Pathog. 13 (10): e1006691. -
Penadés, M. et al. (2018) Long-term implications of feed energy source in different genetic types of reproductive rabbit females. II. Immunologic status.
Animal. 12 (9): 1877-85. -
Jeklova, E. et al. (2020) Characterization of humoral and cell-mediated immunity in rabbits orally infected with Encephalitozoon cuniculi..
Vet Res. 51 (1): 79. -
Niedźwiedzka-Rystwej, P. et al. (2020) B and T lymphocytes in rabbits change according to the sex and throughout the year.
Pol J Vet Sci. 23 (1): 37-42. -
Muñoz-Silvestre, A. et al. (2020) Pathogenesis of Intradermal Staphylococcal Infections: Rabbit Experimental Approach to Natural Staphylococcus aureus Skin Infections.
Am J Pathol. 190 (6): 1188-1210. -
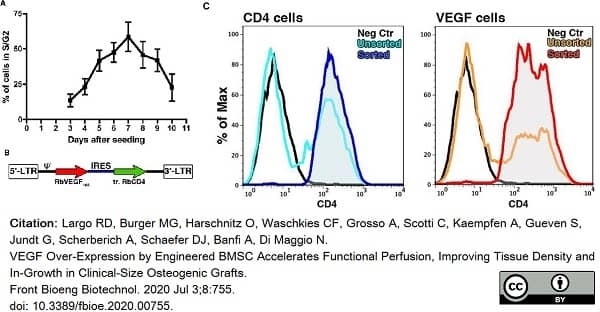
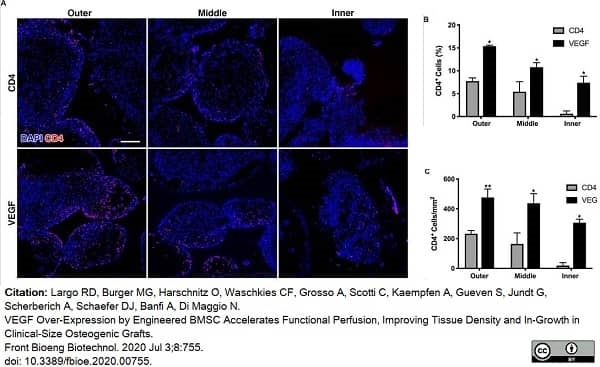
Largo, R.D. et al. (2020) VEGF Over-Expression by Engineered BMSC Accelerates Functional Perfusion, Improving Tissue Density and In-Growth in Clinical-Size Osteogenic Grafts.
Front Bioeng Biotechnol. 8: 755. -
Niedźwiedzka-Rystwej, P. et al. (2022) Reactivity of selected markers of innate and adaptive immunity in rabbits experimentally infected with antigenic variants of RHD (Lagovirus europaeus/GI.1a).
Vet Res Commun. 46 (1): 233-42. -
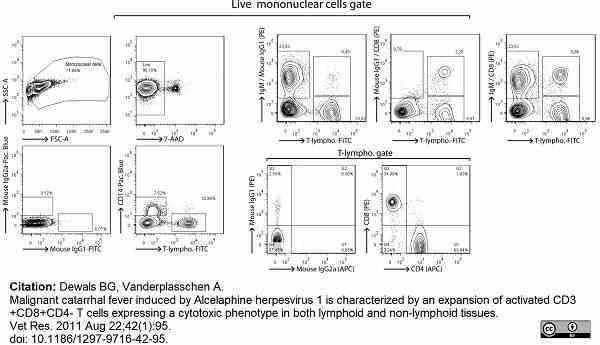
Dewals, B.G. & Vanderplasschen, A. (2011) Malignant catarrhal fever induced by Alcelaphine herpesvirus 1 is characterized by an expansion of activated CD3+CD8+CD4- T cells expressing a cytotoxic phenotype in both lymphoid and non-lymphoid tissues.
Vet Res. 42 (1): 95. -
Li, H. et al. (2011) Characterization of ovine herpesvirus 2-induced malignant catarrhal fever in rabbits.
Vet Microbiol. 150 (3-4): 270-7.
- RRID
- AB_2075555
- UniProt
- P46630
- Entrez Gene
- CD4
- GO Terms
- GO:0007155 cell adhesion
- GO:0016021 integral to membrane
- GO:0006955 immune response
- GO:0045058 T cell selection
MCA799F
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Rabbit ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up