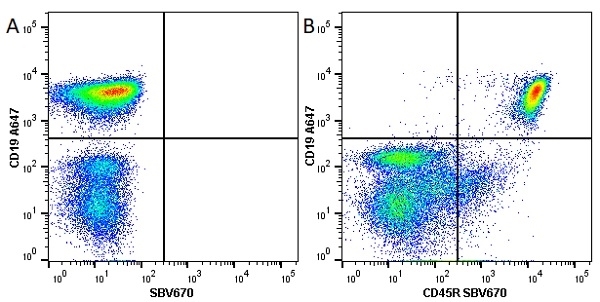
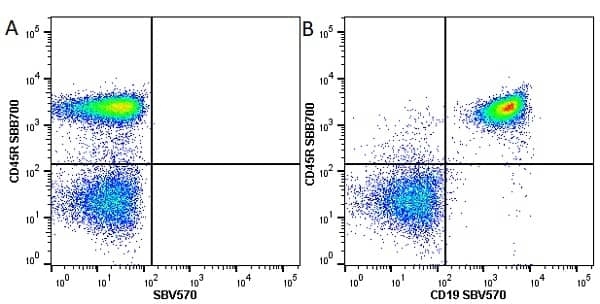
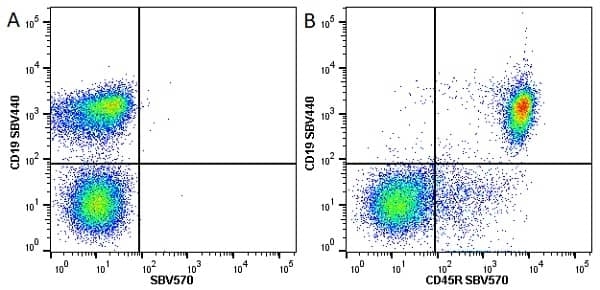
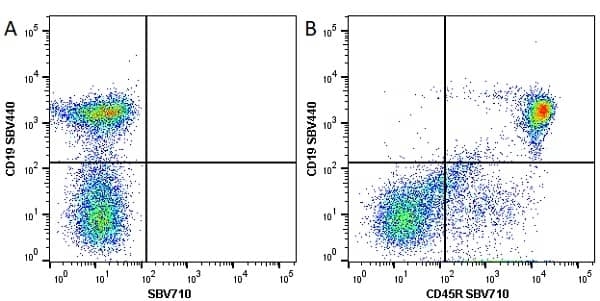
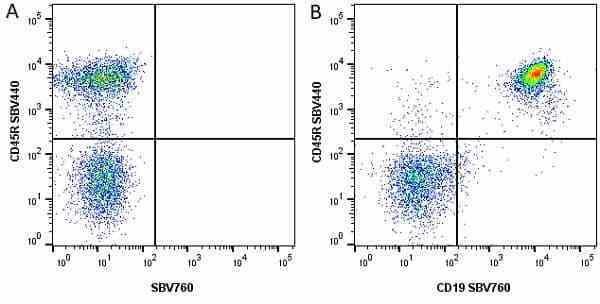
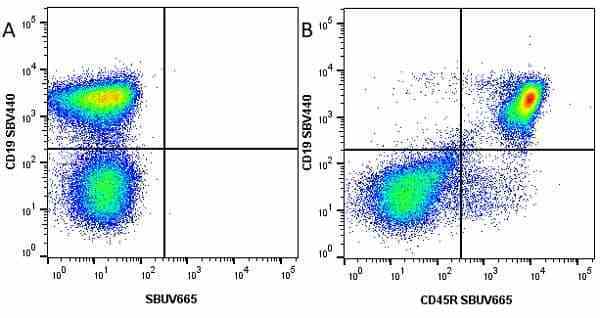
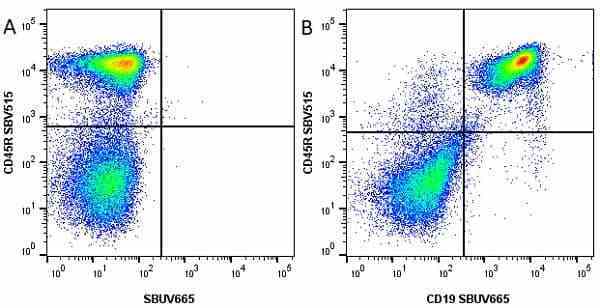
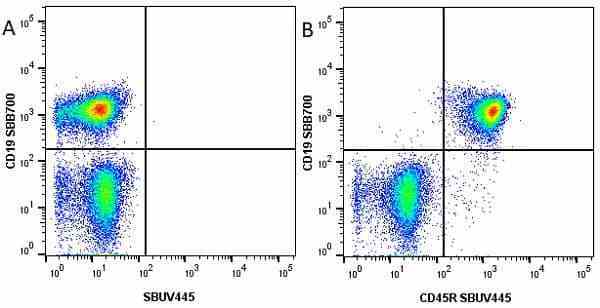
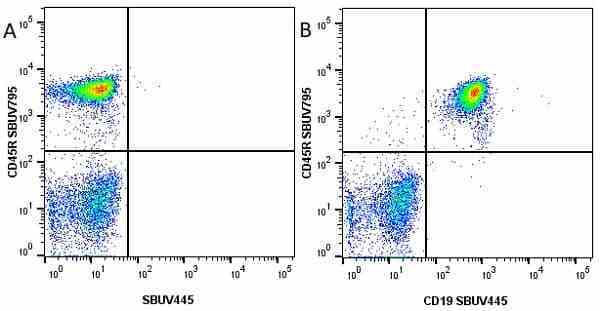
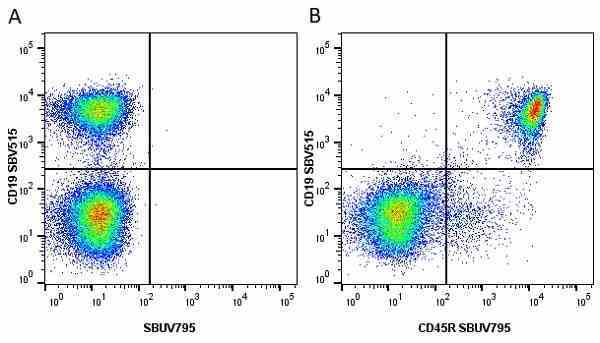
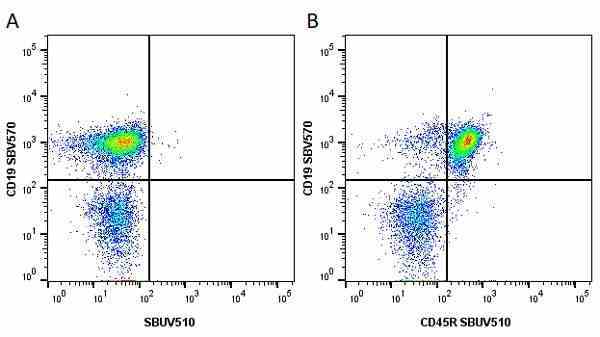
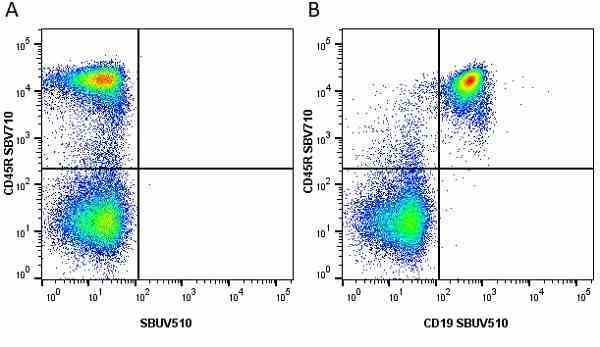
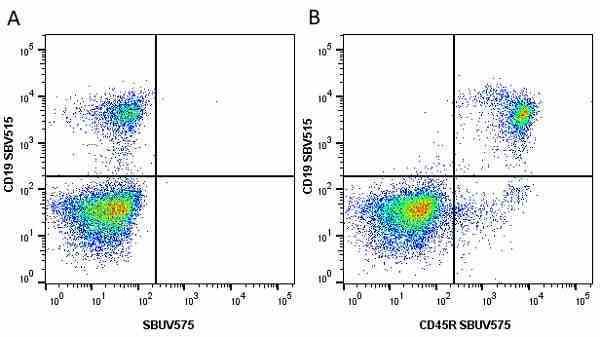
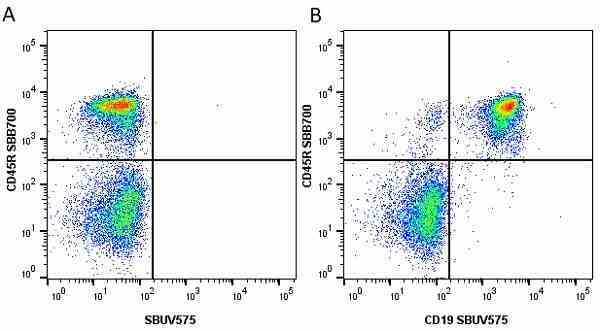
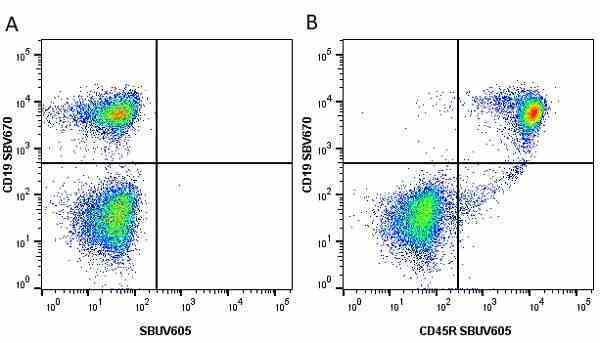
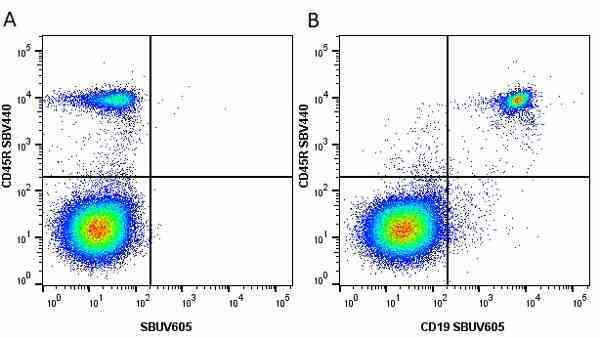
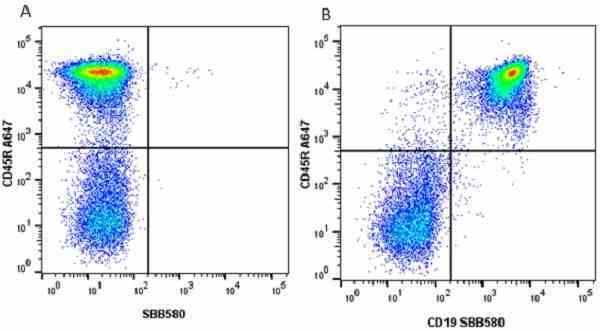
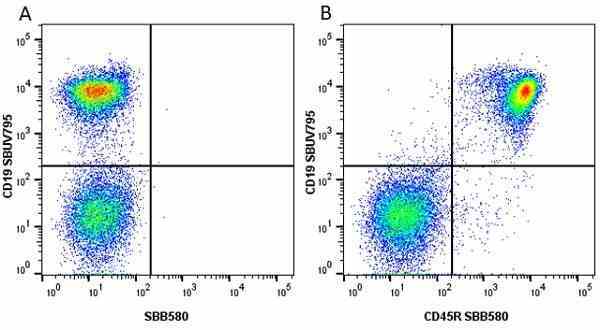
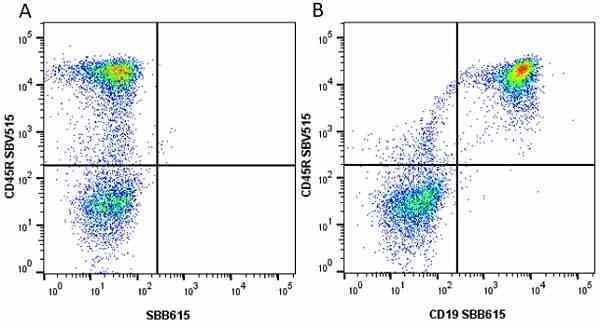
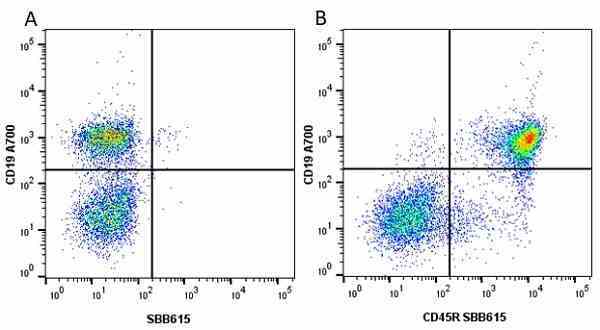
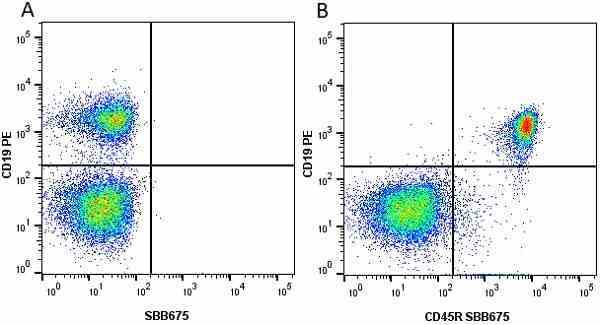
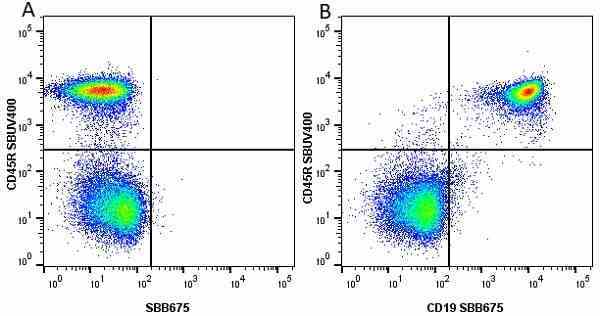
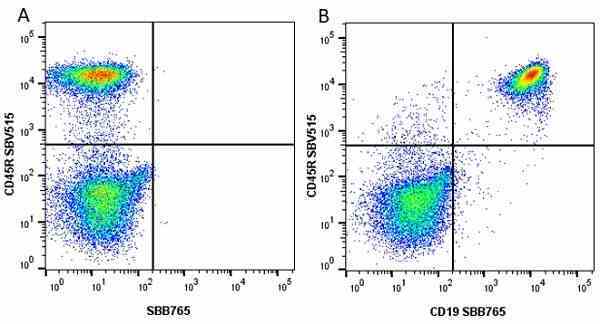
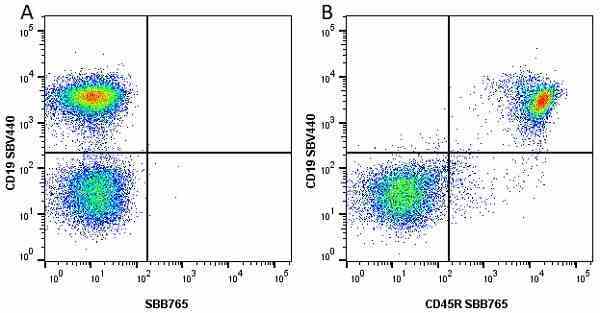
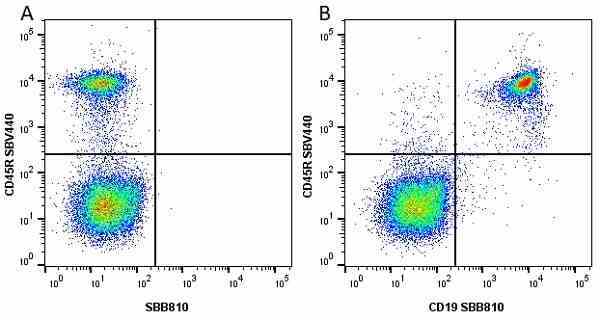
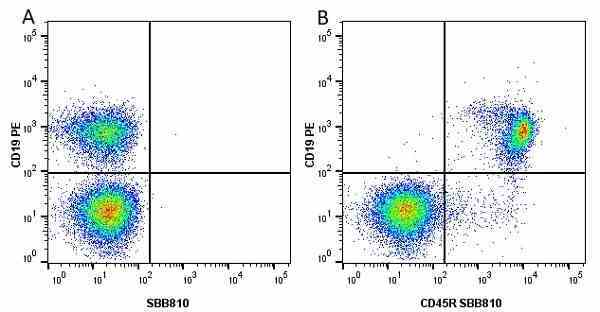
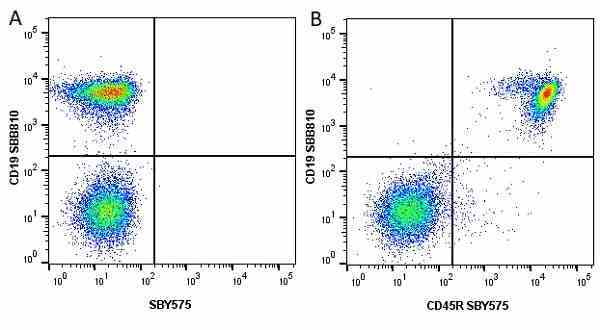
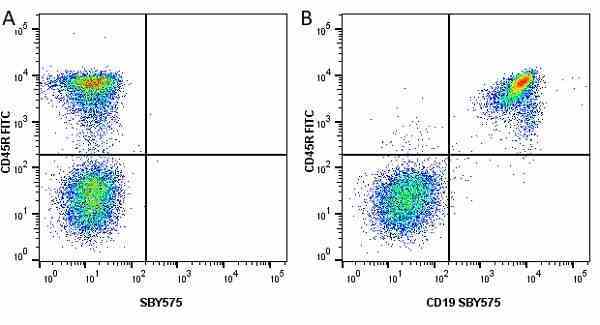
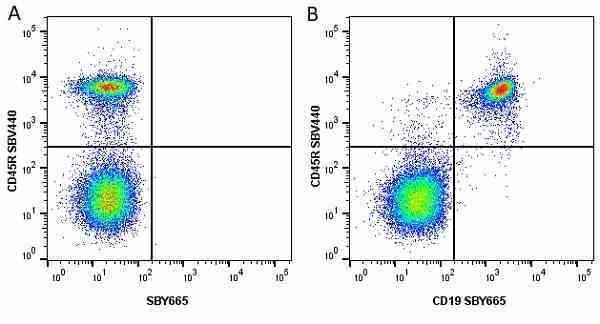
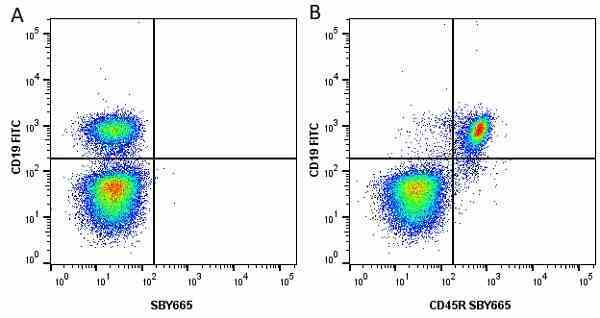
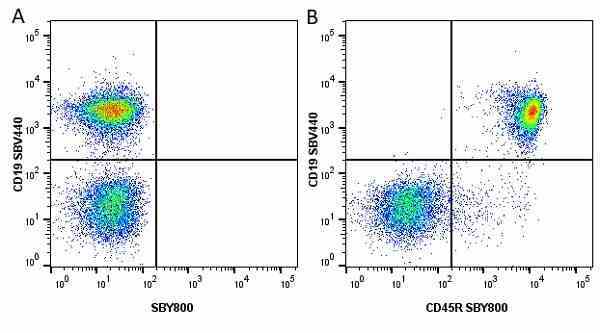
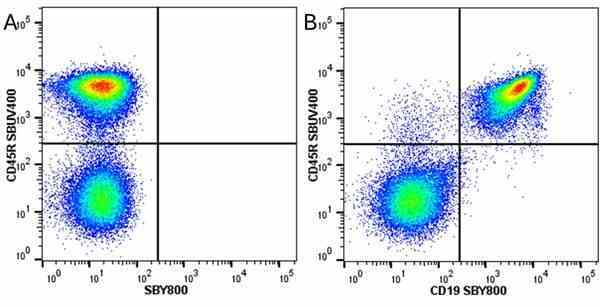
CD45R antibody | RA3-6B2





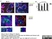





















































Rat anti Mouse CD45R:StarBright UltraViolet 795
- Product Type
- Monoclonal Antibody
- Clone
- RA3-6B2
- Isotype
- IgG2a
- Specificity
- CD45R
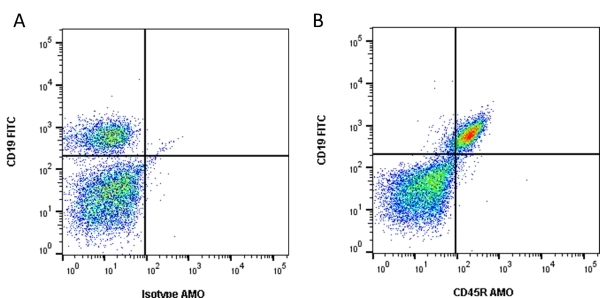
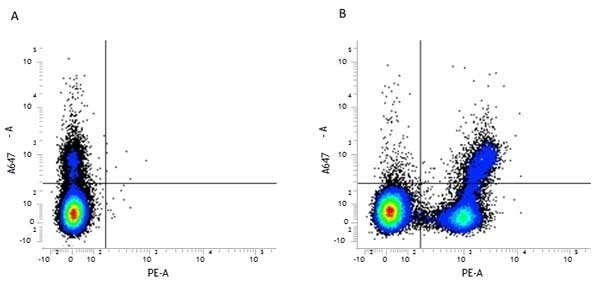
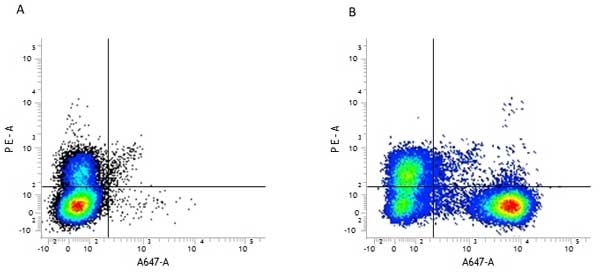
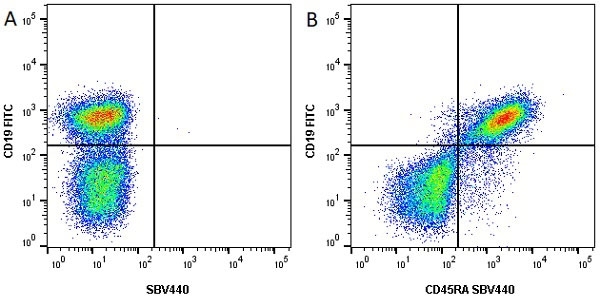
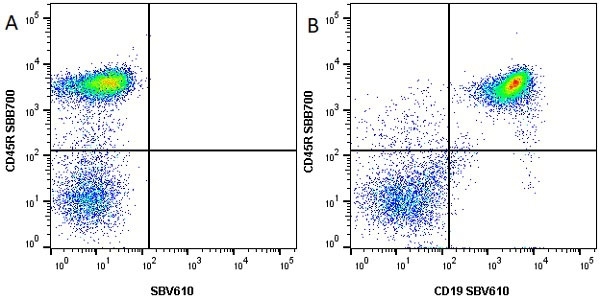
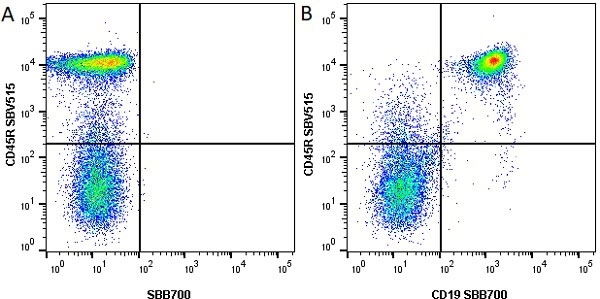
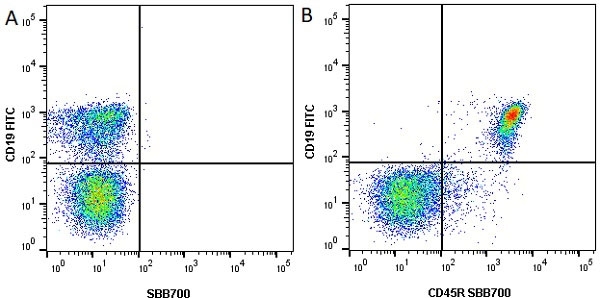
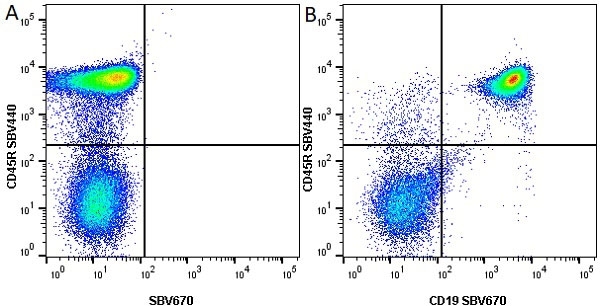
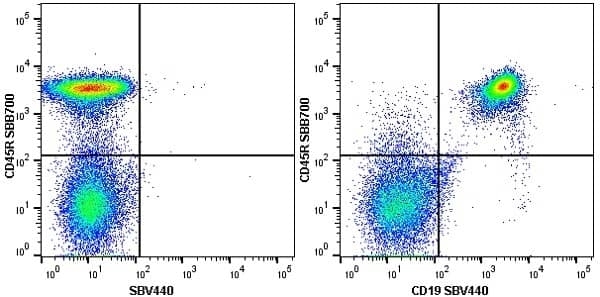
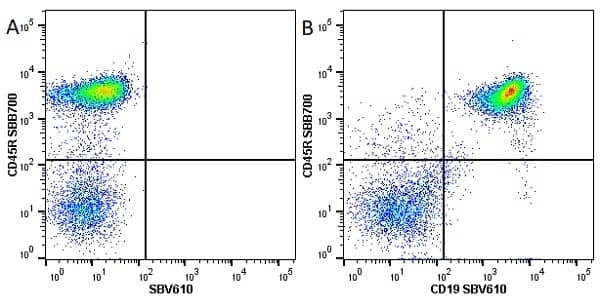
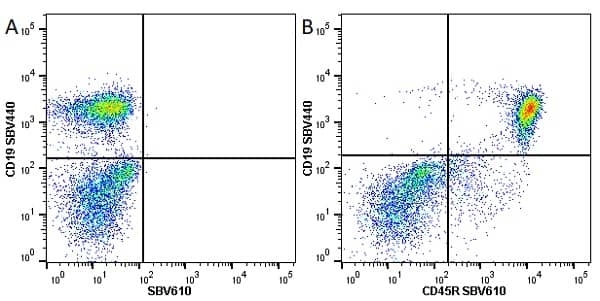
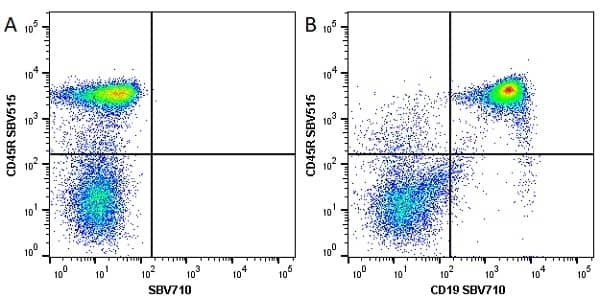
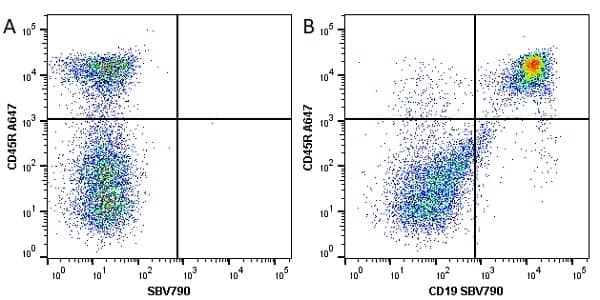
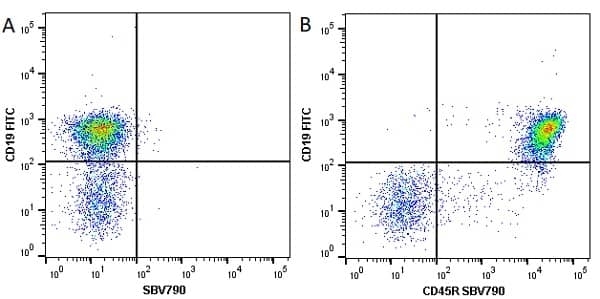
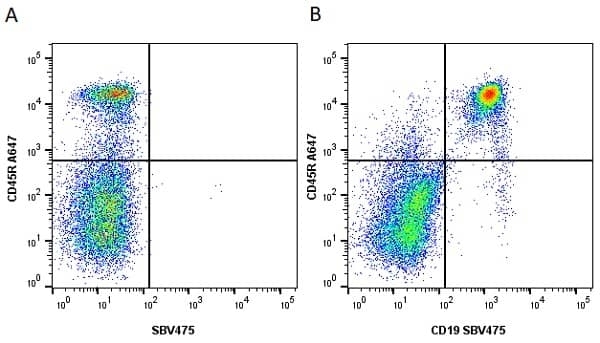
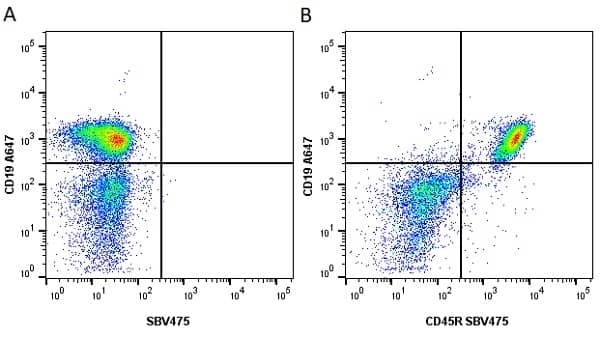
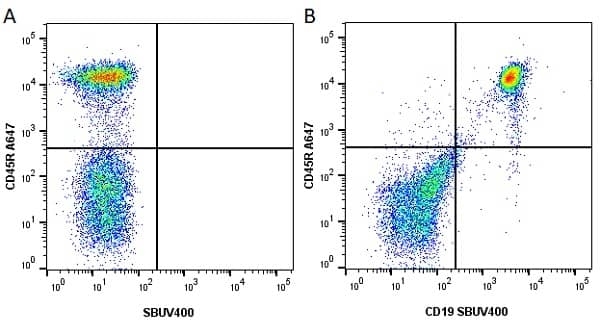
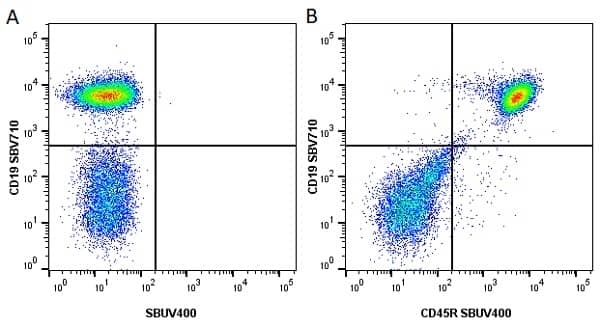
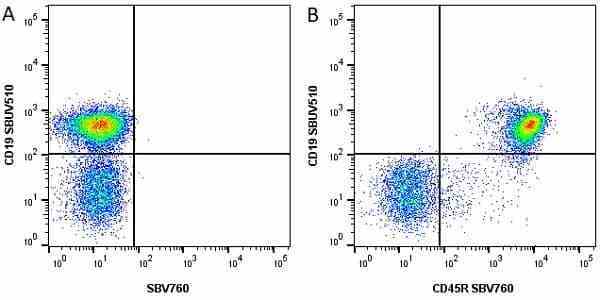
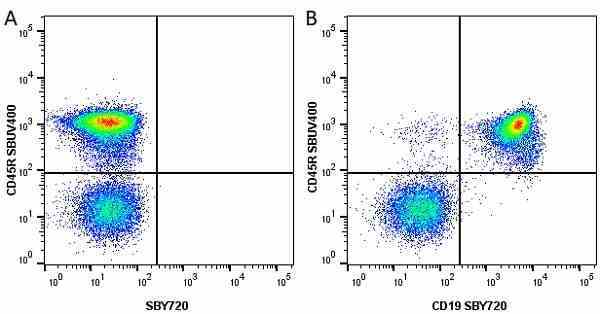
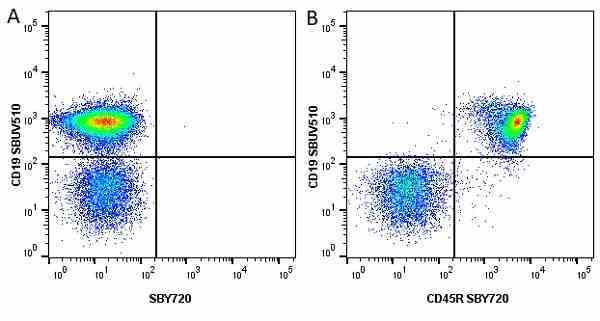
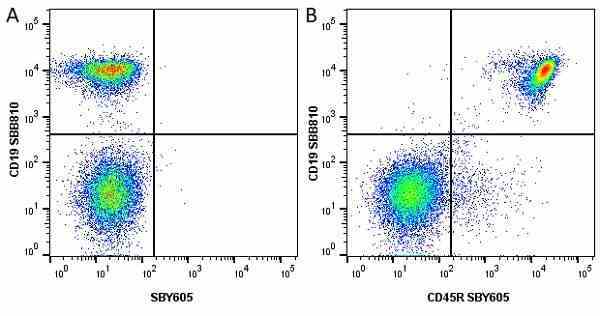
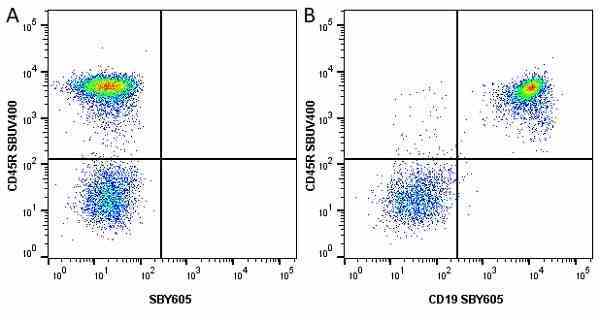
| Rat anti Mouse CD45R antibody, clone RA3-6B2 recognizes murine CD45R, a form of the CD45 antigen expressed by B cells and lytically active subsets of NK cells and non-MHC restricted CTL's. Rat anti Mouse CD45R antibody, clone RA3-6B2 immunoprecipitates the high molecular weight form of CD45 (220 kDa). Rat anti Mouse CD45R antibody, clone RA3-6B2 is suitable for plp fixed paraffin embedded tissues (Whiteland et al.1995). |
- Target Species
- Mouse
- Species Cross-Reactivity
-
Target Species Cross Reactivity Human Cat - N.B. Antibody reactivity and working conditions may vary between species.
- Product Form
- Purified IgG conjugated to StarBright UltraViolet 795 - liquid
- Preparation
- Purified IgG prepared by affinity chromatography on Protein G from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- 0.09% sodium azide (NaN3)
1% bovine serum albumin
0.1% Pluronic F68
0.1% PEG 3350
0.05% Tween 20 - Immunogen
- Murine leukemia-induced pre-B tumor cells (RAW112)
- Fusion Partners
- Spleen cells from immunized Lewis rats were fused with cells of the rat S194/5 XX0.BU-1 myeloma cell line
- Max Ex/Em
-
Fluorophore Excitation Max (nm) Emission Max (nm) StarBright UltraViolet 795 340 792 - Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
- Acknowledgements
- This product is covered by U.S. Patent No. 10,150,841 and related U.S. and foreign counterparts
This product should be stored undiluted.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Flow Cytometry | Neat |
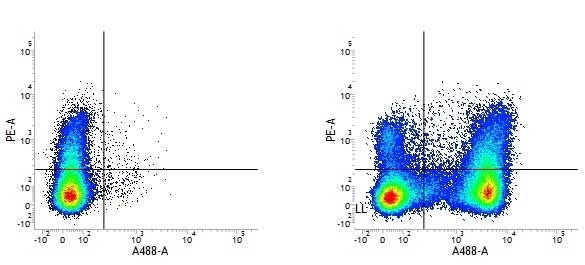
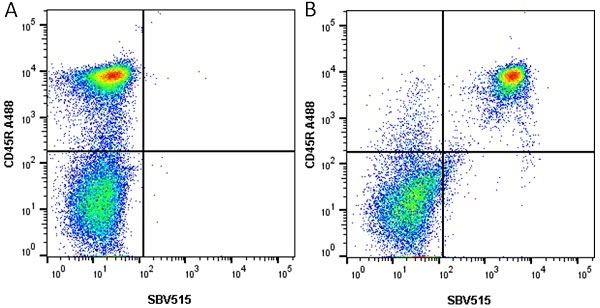
- Flow Cytometry
- Use 5μl of the suggested working dilution to label 106 cells in 100μl. Best practices suggest a 5 minutes centrifugation at 6,000g prior to sample application.
How to Use the Spectraviewer
Watch the Tool Tutorial Video ▸- Start by selecting the application you are interested in, with the option to select an instrument from the drop down menu or create a customized instrument
- Select the fluorophores or fluorescent proteins you want to include in your panel to check compatibility
- Select the lasers and filters you wish to include
- Select combined or multi-laser view to visualize the spectra
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Mouse Seroblock FcR | BUF041A | F | 0.1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse Seroblock FcR | ||||||
| Mouse Seroblock FcR | BUF041B | F | 0.5 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse Seroblock FcR | ||||||
Source Reference
-
Coffman, R.L. (1982) Surface antigen expression and immunoglobulin gene rearrangement during mouse pre-B cell development.
Immunol Rev. 69: 5-23.
References for CD45R antibody
-
Holmes, K.L. et al. (1986) Analysis of neoplasms induced by Cas-Br-M MuLV tumor extracts.
J Immunol. 137 (2): 679-88. -
Spangrude, G.J. et al. (1988) Purification and characterization of mouse hematopoietic stem cells.
Science. 241: 58-62. -
Spangrude, G.J. et al. (1988) Two rare populations of mouse Thy-1lo bone marrow cells repopulate the thymus.
J Exp Med. 167 (5): 1671-83. -
Whiteland, J.L. et al. (1995) Immunohistochemical detection of T-cell subsets and other leukocytes in paraffin-embedded rat and mouse tissues with monoclonal antibodies.
J Histochem Cytochem. 43 (3): 313-20. -
Hawke, S. et al. (1998) Long-term persistence of activated cytotoxic T lymphocytes after viral infection of the central nervous system.
J Exp Med. 187: 1575-82. -
Rosmalen, J.G. et al. (2000) Subsets of macrophages and dendritic cells in nonobese diabetic mouse pancreatic inflammatory infiltrates: correlation with the development of diabetes.
Lab Invest. 80 (1): 23-30. -
Perry, M.J. et al. (2000) Effects of high-dose estrogen on murine hematopoietic bone marrow precede those on osteogenesis.
Am J Physiol Endocrinol Metab. 279: E1159-65. -
Stevenson, P.G. et al. (2002) Uncoupling of virus-induced inflammation and anti-viral immunity in the brain parenchyma.
J Gen Virol. 83: 1735-43.
View The Latest Product References
-
Straubinger, R.K. et al. (2003) Quantitative evaluation of inflammatory and immune responses in the early stages of chronic Helicobacter pylori infection.
Infect Immun. 71: 2693-703. -
Shulga-Morskaya, S. et al. (2004) B cell-activating factor belonging to the TNF family acts through separate receptors to support B cell survival and T cell-independent antibody formation.
J Immunol. 173 (4): 2331-41. -
Gengozian, N. et al. (2005) Characterization of a monoclonal antibody identifying a CD45RA antigen on feline leukocytes.
Vet Immunol Immunopathol. 108: 253-64. -
Herrmann, I. et al. (2006) Streptococcus pneumoniae Infection aggravates experimental autoimmune encephalomyelitis via Toll-like receptor 2.
Infect Immun. 74: 4841-8. -
Itoh, T. et al. (2007) Ddb2 is a haploinsufficient tumor suppressor and controls spontaneous germ cell apoptosis.
Hum Mol Genet. 16: 1578-86. -
McGill, J. et al. (2009) Fetal exposure to ethanol has long-term effects on the severity of influenza virus infections.
J Immunol. 182: 7803-8 -
Ankeny, D.P. et al. (2009) B cells produce pathogenic antibodies and impair recovery after spinal cord injury in mice.
J Clin Invest. 119: 2990-9. -
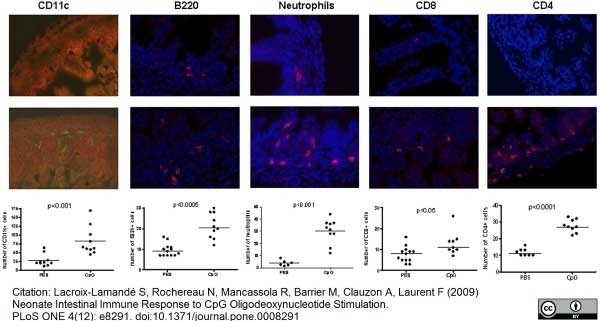
Lacroix-Lamande, S. et al. (2009) Neonate intestinal immune response to CpG oligodeoxynucleotide stimulation.
PLoS One. 4: e8291. -
Lundqvist, J. et al. (2010) Concomitant infection decreases the malaria burden but escalates relapsing fever borreliosis.
Infect Immun. 78 (5): 1924-30. -
Giuriato, S. et al. (2010) Conditional TPM3-ALK and NPM-ALK transgenic mice develop reversible ALK-positive early B-cell lymphoma/leukemia.
Blood. 115: 4061-70. -
Kleiter, I. et al. (2010) Smad7 in T cells drives T helper 1 responses in multiple sclerosis and experimental autoimmune encephalomyelitis.
Brain. 133: 1067-81. -
Nakaya, T. et al. (2010) Critical role of Pcid2 in B cell survival through the regulation of MAD2 expression.
J Immunol. 185: 5180-7. -
Soejima, M. et al. (2011) Role of innate immunity in a murine model of histidyl-transfer RNA synthetase (Jo-1)-mediated myositis.
Arthritis Rheum. 63: 479-87. -
Bertilaccio, M.T. et al. (2011) Lack of TIR8/SIGIRR triggers progression of chronic lymphocytic leukemia in mouse models.
Blood. 118: 660-9. -
Zhou, Z. et al. (2011) Autoreactive marginal zone B cells enter the follicles and interact with CD4+ T cells in lupus-prone mice.
BMC Immunol. 2011; 12:7. -
Fanning, S. et al. (2012) Bifidobacterial surface-exopolysaccharide facilitates commensal-host interaction through immune modulation and pathogen protection.
Proc Natl Acad Sci U S A. 109 (6): 2108-13. -
Ruf, M.T. et al. (2012) Chemotherapy-Associated Changes of Histopathological Features of Mycobacterium ulcerans Lesions in a Buruli Ulcer Mouse Model.
Antimicrob Agents Chemother. 56: 687-96. -
Carpenter, R.S. et al. (2015) Traumatic spinal cord injury in mice with human immune systems.
Exp Neurol. 271: 432-44. -
Lastrucci, C. et al. (2015) Molecular and cellular profiles of the resolution phase in a damage-associated molecular pattern (DAMP)-mediated peritonitis model and revelation of leukocyte persistence in peritoneal tissues.
FASEB J. 29 (5): 1914-29. -
Gibson-Corley, K.N. et al. (2016) A method for histopathological study of the multifocal nature of spinal cord lesions in murine experimental autoimmune encephalomyelitis.
PeerJ. 4: e1600. -
Thiele Née Schrewe, L. et al. (2020) Functional relevance of the multi-drug transporter abcg2 on teriflunomide therapy in an animal model of multiple sclerosis.
J Neuroinflammation. 17 (1): 9. -
Allen, A.C. et al. (2021) Parallel in vivo. experimental evolution reveals that increased stress resistance was key for the emergence of persistent tuberculosis bacilli.
Nat Microbiol. 6 (8): 1082-93. -
Chanut, F.J.A. et al. (2021) Conditioning Regimens in Long-Term Pre-Clinical Studies to Support Development of Ex Vivo Gene Therapy: Review of Nonproliferative and Proliferative Changes.
Hum Gene Ther. 32 (1-2): 66-76. -
Jaensch, S.M. et al. (2022) Clinicopathologic and immunophenotypic features in dogs with presumptive large granular lymphocyte leukaemia.
Aust Vet J. 100 (11): 527-32. -
Roca, C.P. et al. (2023) A cross entropy test allows quantitative statistical comparison of t-SNE and UMAP representations
Cell Reports Methods. 3 (1): 100390. -
Kohlmeyer, J.L. et al. (2023) CDK4/6-MEK Inhibition in MPNSTs Causes Plasma Cell Infiltration, Sensitization to PD-L1 Blockade, and Tumor Regression.
Clin Cancer Res. 29 (17): 3484-97.
- Synonyms
- B220
- LY-5
- UniProt
- P06800
- Entrez Gene
- Ptprc
- GO Terms
- GO:0000187 activation of MAPK activity
- GO:0001915 negative regulation of T cell mediated cytotoxicity
- GO:0001916 positive regulation of T cell mediated cytotoxicity
- GO:0001960 negative regulation of cytokine-mediated signaling pathway
- GO:0002925 positive regulation of humoral immune response mediated by circulating immunoglobulin
- GO:0051726 regulation of cell cycle
- GO:0004725 protein tyrosine phosphatase activity
- GO:0005887 integral to plasma membrane
- GO:0005925 focal adhesion
- View More GO Terms
- GO:0006469 negative regulation of protein kinase activity
- GO:0006470 protein dephosphorylation
- GO:0007159 leukocyte cell-cell adhesion
- GO:0008201 heparin binding
- GO:0009897 external side of plasma membrane
- GO:0019901 protein kinase binding
- GO:0030183 B cell differentiation
- GO:0030890 positive regulation of B cell proliferation
- GO:0031953 negative regulation of protein autophosphorylation
- GO:0034113 heterotypic cell-cell adhesion
- GO:0042098 T cell proliferation
- GO:0042100 B cell proliferation
- GO:0043065 positive regulation of apoptosis
- GO:0043395 heparan sulfate proteoglycan binding
- GO:0043410 positive regulation of MAPKKK cascade
- GO:0045059 positive thymic T cell selection
- GO:0045060 negative thymic T cell selection
- GO:0045121 membrane raft
- GO:0045577 regulation of B cell differentiation
- GO:0045588 positive regulation of gamma-delta T cell differentiation
- GO:0046641 positive regulation of alpha-beta T cell proliferation
- GO:0048304 positive regulation of isotype switching to IgG isotypes
- GO:0050732 negative regulation of peptidyl-tyrosine phosphorylation
- GO:0050852 T cell receptor signaling pathway
- GO:0050853 B cell receptor signaling pathway
- GO:0050855 regulation of B cell receptor signaling pathway
- GO:0050857 positive regulation of antigen receptor-mediated signaling pathway
- GO:0051209 release of sequestered calcium ion into cytosol
- GO:0051607 defense response to virus
MCA1258SBUV795
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Mouse ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up