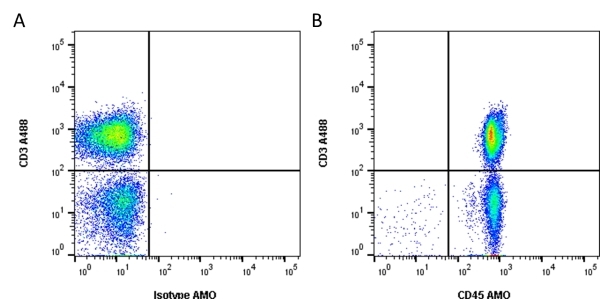
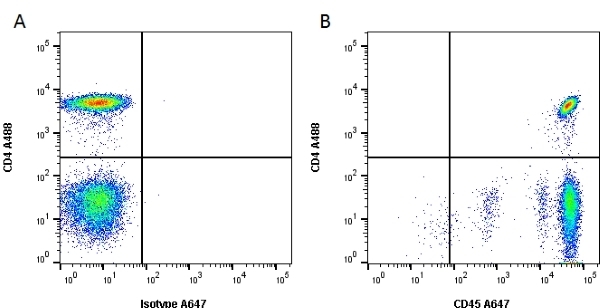
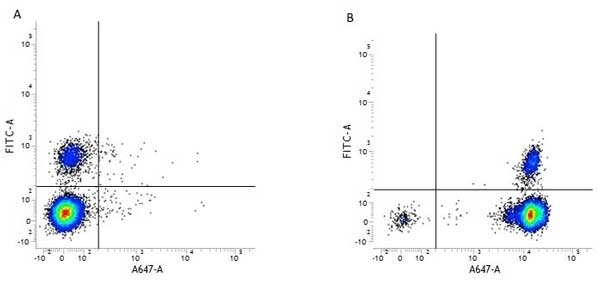
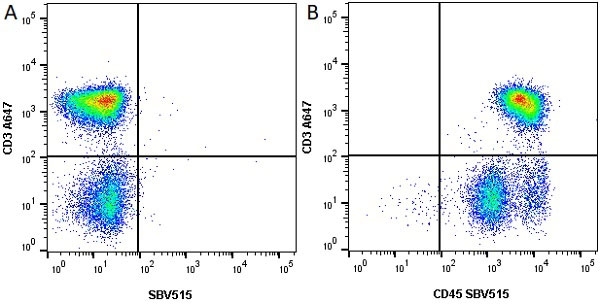
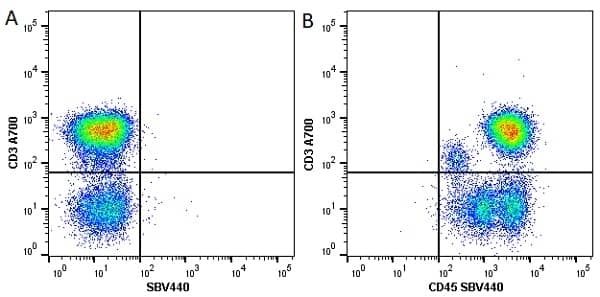
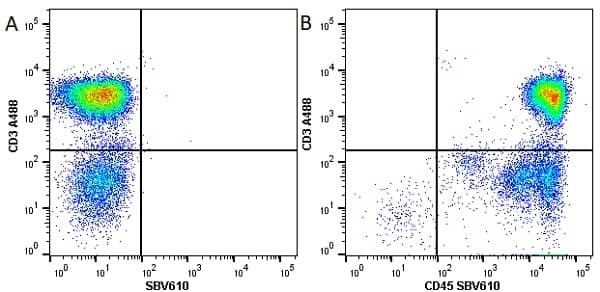
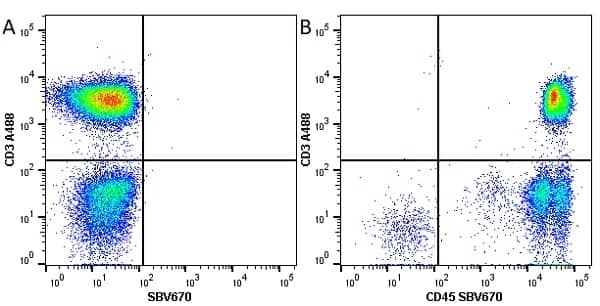
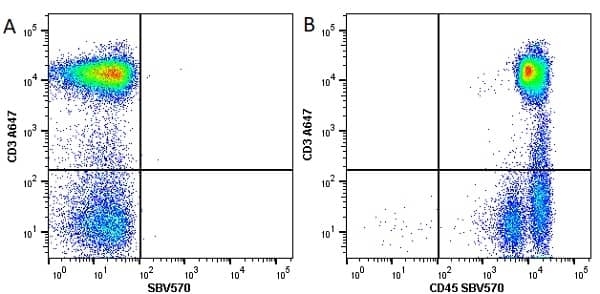
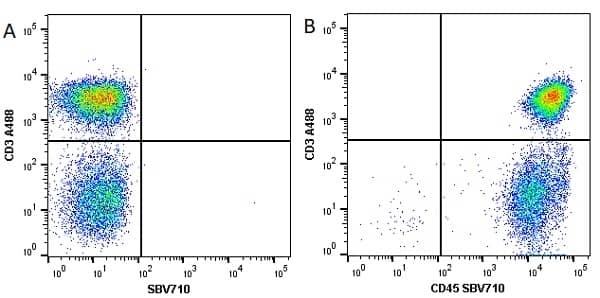
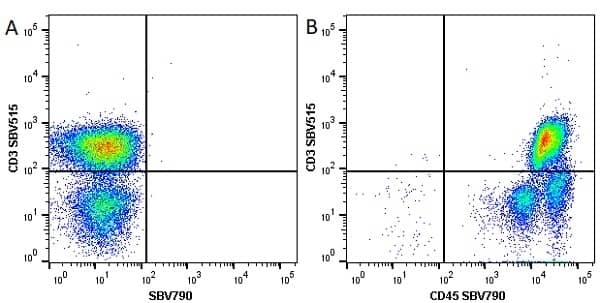
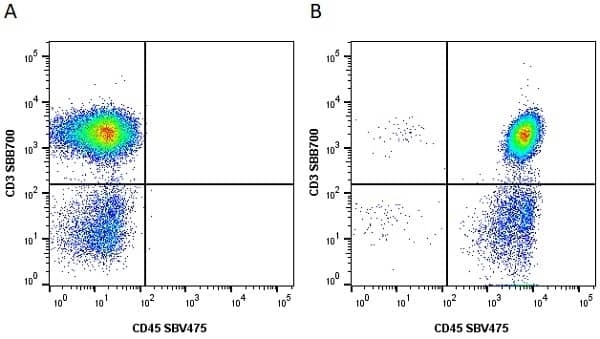
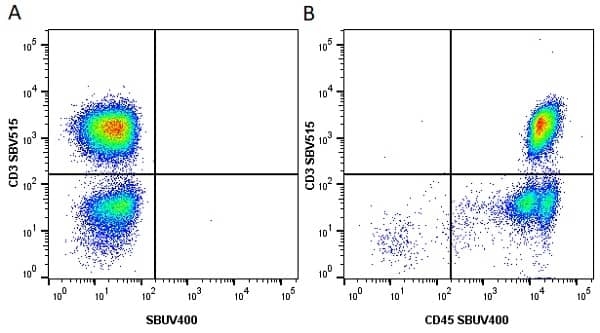
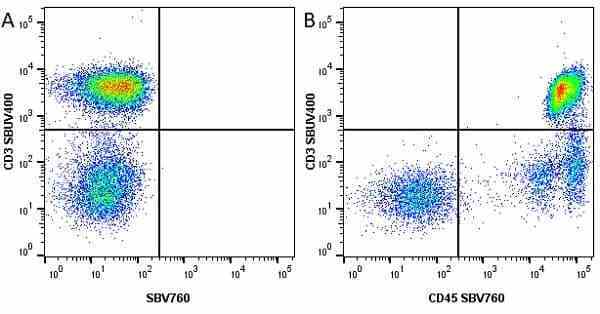
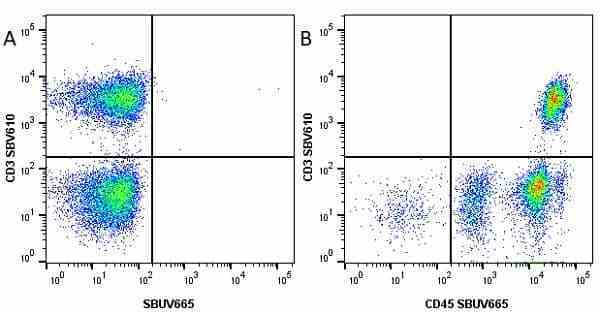
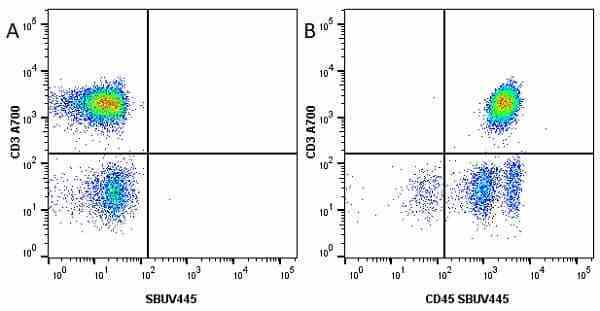
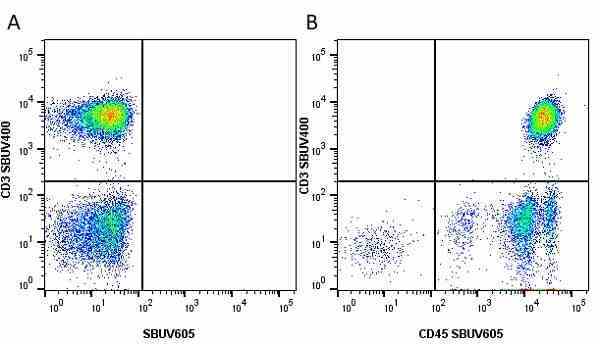
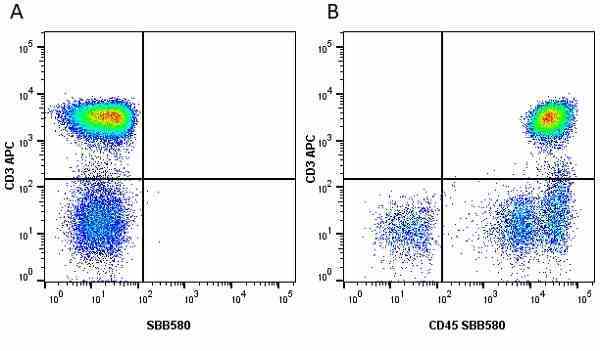
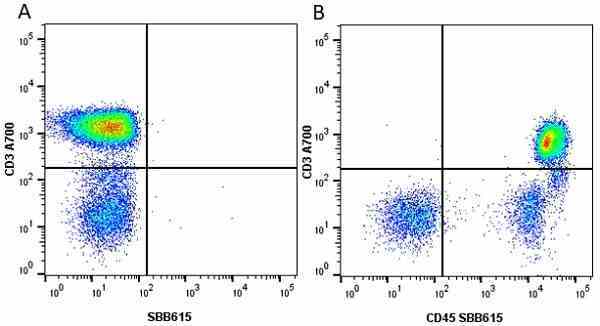
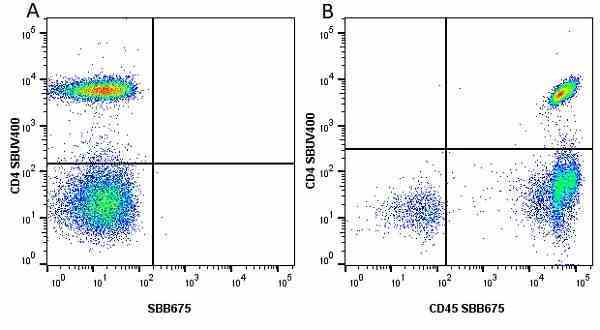
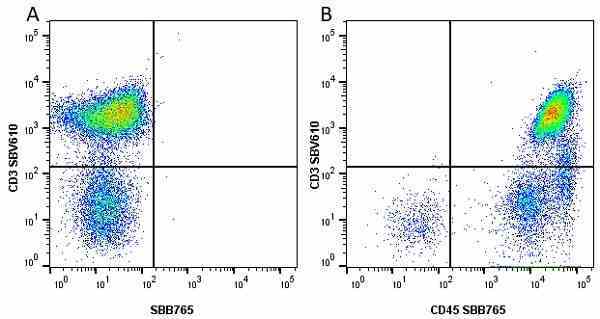
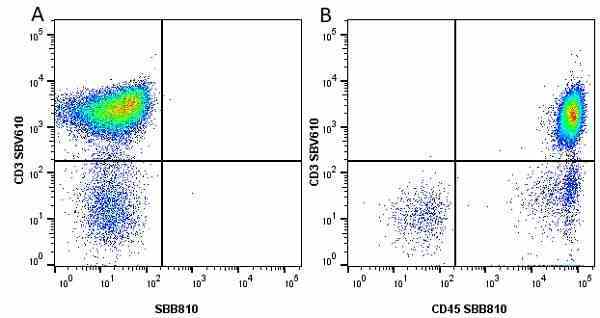
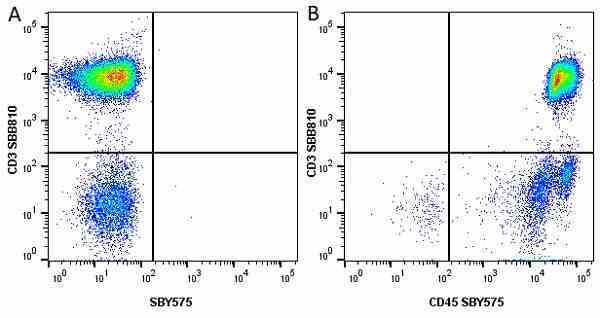
CD45 antibody | F10-89-4







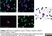



































Mouse anti Human CD45:StarBright Violet 475
- Product Type
- Monoclonal Antibody
- Clone
- F10-89-4
- Isotype
- IgG2a
- Specificity
- CD45
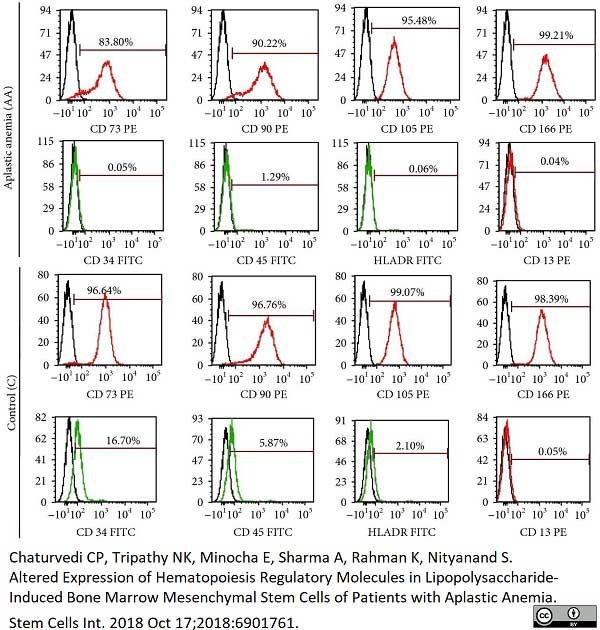
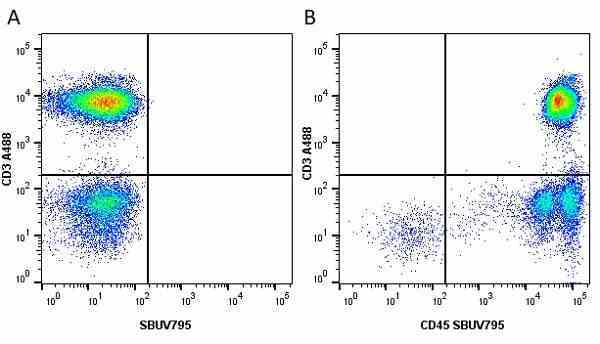
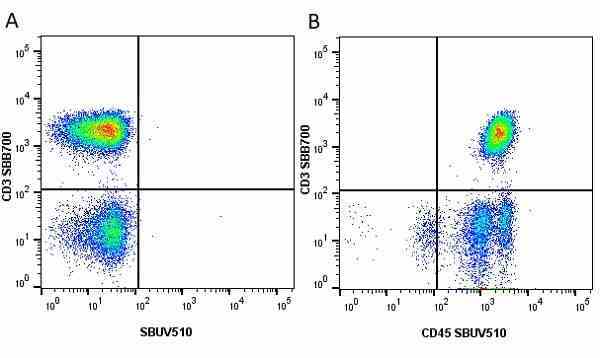
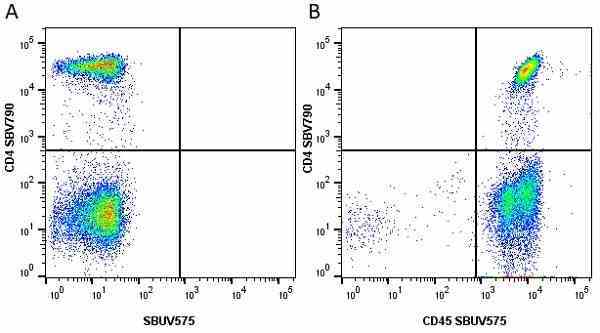
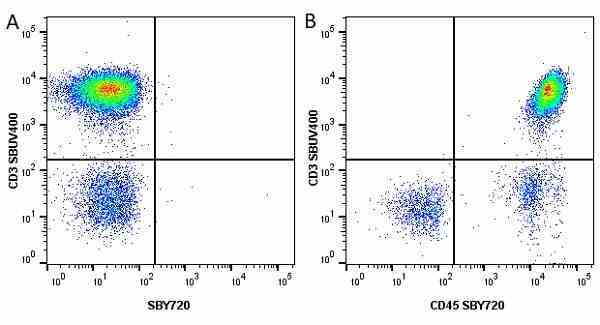
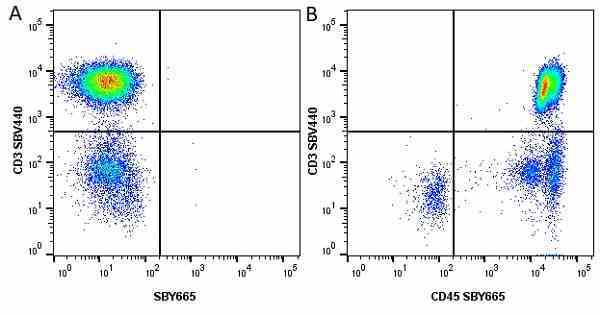
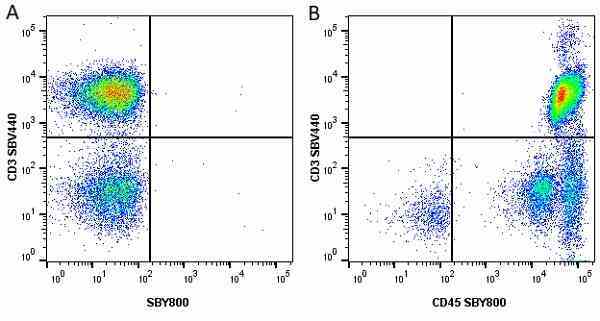
| Mouse anti Human CD45 antibody, clone F10-89-4 recognizes the human CD45 cell surface antigen, also known as leucocyte common antigen (LCA). CD45 is a complex molecule existing in a number of isoforms. Antibodies recognizing a common epitope on all of these isoforms are termed CD45 whilst those recognizing only individual isoforms are termed CD45RA or CD45RO etc. Mouse anti Human CD45 antibody, clone F10-89-4 reacts with all forms of CD45 expressed by all haematopoietic cells, except erythrocytes, having a higher level of expression on lymphocytes than on granulocytes. It is routinely tested in flow cytometry on human peripheral blood leucocytes. Mouse anti Human CD45 antibody, clone F10-89-4, has been validated for use on the Genesis Cell Isolation System with the CelSelect SlideTM technology. |

|
- Target Species
- Human
- Product Form
- Purified IgG conjugated to StarBright Violet 475 - liquid
- Preparation
- Purified IgG prepared by affinity chromatography on Protein A from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- 0.09% Sodium Azide (NaN3)
1% Bovine Serum Albumin
0.1% Pluronic F68
0.1% PEG 3350
0.05% Tween 20 - Immunogen
- Human T lymphocytes.
- Fusion Partners
- Spleen cells from immunized BALB/c mice were fused with cells of the mouse NS-1 myeloma cell line.
- Max Ex/Em
-
Fluorophore Excitation Max (nm) Emission Max (nm) StarBright Violet 475 405 479 - Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
- Acknowledgements
- This product is covered by U.S. Patent No. 10,150,841 and related U.S. and foreign counterparts
This product should be stored undiluted.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Flow Cytometry | Neat |
- Flow Cytometry
- Use 5ul of the suggested working dilution to label 106 cells in 100ul. Best practices suggest a 5 minutes centrifugation at 6,000g prior to sample application.
How to Use the Spectraviewer
Watch the Tool Tutorial Video ▸- Start by selecting the application you are interested in, with the option to select an instrument from the drop down menu or create a customized instrument
- Select the fluorophores or fluorescent proteins you want to include in your panel to check compatibility
- Select the lasers and filters you wish to include
- Select combined or multi-laser view to visualize the spectra
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Human Seroblock | BUF070A | F | 50 Test | Log in | |||
| List Price | Your Price | ||||||
| Log in | |||||||
| Description | Human Seroblock | ||||||
| Human Seroblock | BUF070B | F | 200 Test | Log in | |||
| List Price | Your Price | ||||||
| Log in | |||||||
| Description | Human Seroblock | ||||||
Source Reference
-
Dalchau, R. et al. (1980) Monoclonal antibody to a human leukocyte-specific membrane glycoprotein probably homologous to the leukocyte-common (L-C) antigen of the rat.
Eur J Immunol. 10 (10): 737-44.
References for CD45 antibody
-
Quenby, S et al. (1999) Pre-implantation endometrial leukocytes in women with recurrent miscarriage.
Human Reprod. 14(9):2386-2391. -
Hauser, P.V. et al. (2010) Stem cells derived from human amniotic fluid contribute to acute kidney injury recovery.
Am J Pathol. 177: 2011-21. -
Mallam, E. et al. (2010) Characterization of in vitro expanded bone marrow-derived mesenchymal stem cells from patients with multiple sclerosis.
Mult Scler. 16: 909-18. -
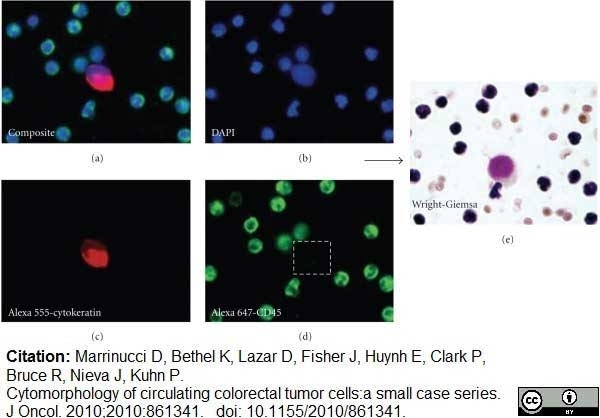
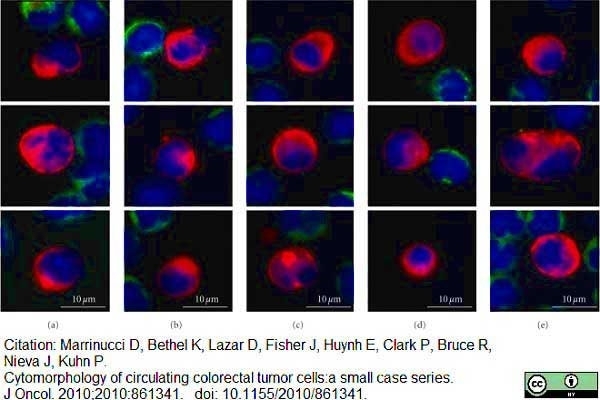
Marrinucci, D. et al. (2010) Cytomorphology of circulating colorectal tumor cells:a small case series.
J Oncol. 2010: 861341. -
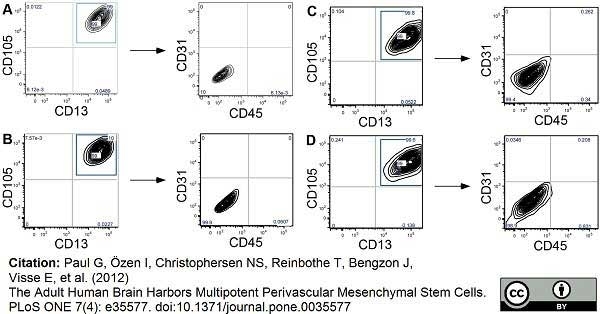
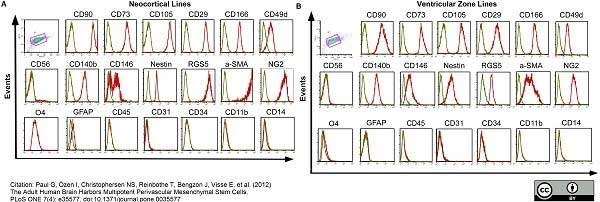
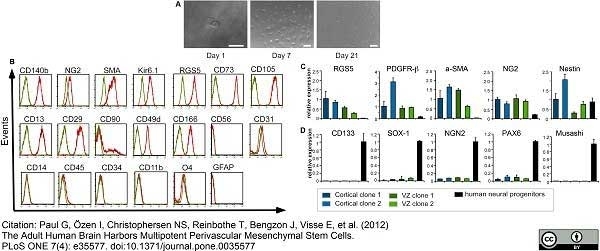
Paul, G. et al. (2012) The adult human brain harbors multipotent perivascular mesenchymal stem cells.
PLoS One. 7: e35577. -
De Schauwer, C. et al. (2012) In search for cross-reactivity to immunophenotype equine mesenchymal stromal cells by multicolor flow cytometry.
Cytometry A. 81 (4): 312-23. -
Kazane, S.A. et al. (2012) Site-specific DNA-antibody conjugates for specific and sensitive immuno-PCR.
Proc Natl Acad Sci U S A. 109: 3731-6. -
Spaas, J.H. et al. (2013) Culture and characterisation of equine peripheral blood mesenchymal stromal cells.
Vet J. 195 (1): 107-13.
View The Latest Product References
-
Sadarangani, A. et al. (2015) GLI2 inhibition abrogates human leukemia stem cell dormancy.
J Transl Med. 13: 98. -
Gunawardene, P. et al. (2015) Association Between Circulating Osteogenic Progenitor Cells and Disability and Frailty in Older Persons: The Nepean Osteoporosis and Frailty Study.
J Gerontol A Biol Sci Med Sci. pii: glv190. -
Mohamed Suhaimi, N.A. et al. (2015) Non-invasive sensitive detection of KRAS and BRAF mutation in circulating tumor cells of colorectal cancer patients.
Mol Oncol. 9 (4): 850-60. -
Ruiz, C. et al. (2015) Limited genomic heterogeneity of circulating melanoma cells in advanced stage patients.
Phys Biol. 12 (1): 016008. -
Gogoi P et al. (2016) Development of an Automated and Sensitive Microfluidic Device for Capturing and Characterizing Circulating Tumor Cells (CTCs) from Clinical Blood Samples.
PLoS One. 11 (1): e0147400. -
Gomiero, C. et al. (2016) Tenogenic induction of equine mesenchymal stem cells by means of growth factors and low-level laser technology.
Vet Res Commun. 40 (1): 39-48. -
Bianchessi, M. et al. (2016) Effect of Fibroblast Growth Factor 2 on Equine Synovial Fluid Chondroprogenitor Expansion and Chondrogenesis.
Stem Cells Int. 2016: 9364974. -
Branly, T. et al. (2017) Characterization and use of Equine Bone Marrow Mesenchymal Stem Cells in Equine Cartilage Engineering. Study of their Hyaline Cartilage Forming Potential when Cultured under Hypoxia within a Biomaterial in the Presence of BMP-2 and TGF-β1.
Stem Cell Rev Rep. 13 (5): 611-30. -
GarikipatiV, N.S. et al. (2018) Isolation and characterization of mesenchymal stem cells from human fetus heart.
PLoS One. 13 (2): e0192244. -
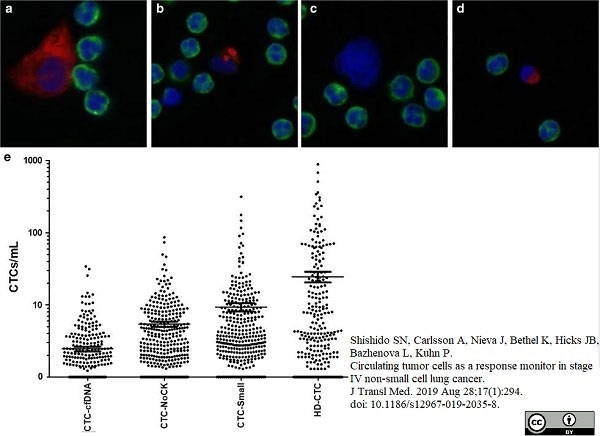
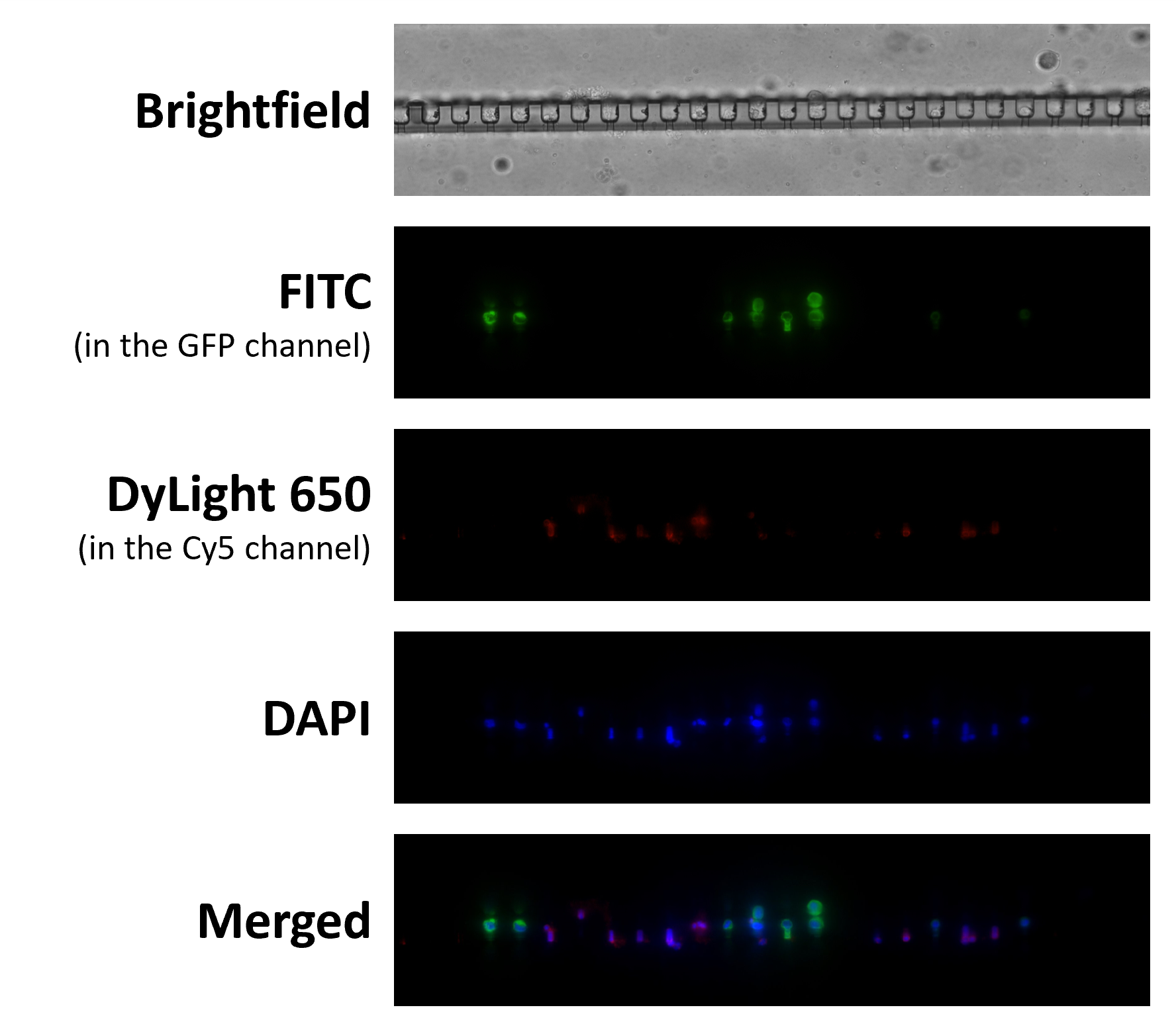
Shishido, S.N. et al. (2019) Circulating tumor cells as a response monitor in stage IV non-small cell lung cancer.
J Transl Med. 17 (1): 294. -
Welter, L. et al. (2020) Treatment response and tumor evolution: lessons from an extended series of multianalyte liquid biopsies in a metastatic breast cancer patient.
Cold Spring Harb Mol Case Stud. 6 (6): a005819. -
Ndacayisaba, L.J. et al. (2022) Enrichment-Free Single-Cell Detection and Morphogenomic Profiling of Myeloma Patient Samples to Delineate Circulating Rare Plasma Cell Clones
Curr Oncol. 29 (5): 2954-72. -
Shishido, S.N. et al. (2022) Liquid Biopsy Landscape in Patients with Primary Upper Tract Urothelial Carcinoma.
Cancers (Basel). 14 (12): 3007. -
Chai, S. et al. (2022) Identification of epithelial and mesenchymal circulating tumor cells in clonal lineage of an aggressive prostate cancer case.
NPJ Precis Oncol. 6 (1): 41. -
Zhu, J. et al. (2022) Sequential Method for Analysis of CTCs and Exosomes from the Same Sample of Patient Blood.
ACS Omega. 7 (42): 37581-88. -
Setayesh, S.M. et al. (2022) Multianalyte liquid biopsy to aid the diagnostic workup of breast cancer.
NPJ Breast Cancer. 8 (1): 112. -
Ndacayisaba, L.J. et al. (2022) Characterization of BCMA Expression in Circulating Rare Single Cells of Patients with Plasma Cell Neoplasms.
Int J Mol Sci. 23 (21): 13427. -
Qi, E. et al. (2023) Investigation of liquid biopsy analytes in peripheral blood of individuals after SARS-CoV-2 infection.
EBioMedicine. 90: 104519. -
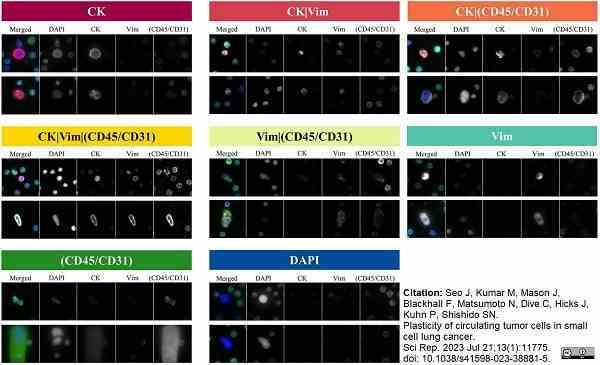
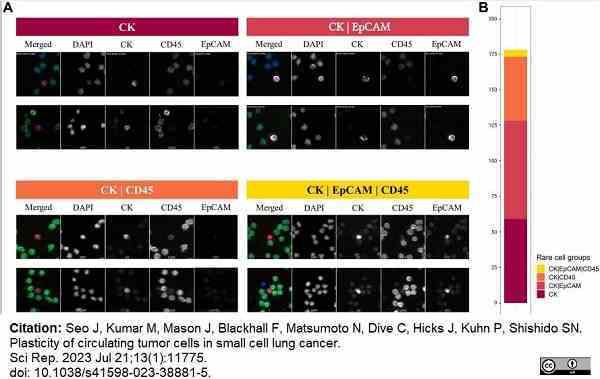
Seo, J. et al. (2023) Plasticity of circulating tumor cells in small cell lung cancer.
Sci Rep. 13 (1): 11775. -
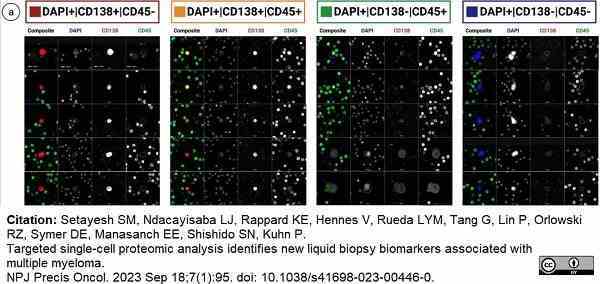
Setayesh, S.M. et al. (2023) Targeted single-cell proteomic analysis identifies new liquid biopsy biomarkers associated with multiple myeloma.
NPJ Precis Oncol. 7 (1): 95. -
Welter, L. et al. (2023) Cell State and Cell Type: Deconvoluting Circulating Tumor Cell Populations in Liquid Biopsies by Multi-Omics.
Cancers (Basel). 15 (15): 3949. -
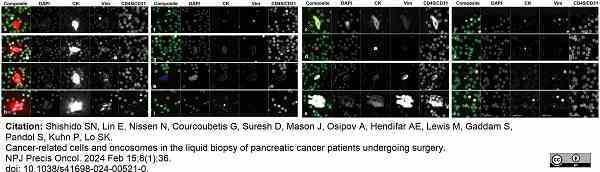
Shishido, S.N. et al. (2024) Cancer-related cells and oncosomes in the liquid biopsy of pancreatic cancer patients undergoing surgery.
NPJ Precis Oncol. 8 (1): 36.
- Synonyms
- LCA
- UniProt
- P08575
- Entrez Gene
- PTPRC
- GO Terms
- GO:0001915 negative regulation of T cell mediated cytotoxicity
- GO:0001960 negative regulation of cytokine-mediated signaling pathway
- GO:0002378 immunoglobulin biosynthetic process
- GO:0005001 transmembrane receptor protein tyrosine phosphatase activity
- GO:0005887 integral to plasma membrane
- GO:0005925 focal adhesion
- GO:0006469 negative regulation of protein kinase activity
- GO:0006470 protein dephosphorylation
- GO:0007411 axon guidance
- View More GO Terms
- GO:0019901 protein kinase binding
- GO:0030890 positive regulation of B cell proliferation
- GO:0033261 regulation of S phase
- GO:0042100 B cell proliferation
- GO:0045121 membrane raft
- GO:0050852 T cell receptor signaling pathway
- GO:0050853 B cell receptor signaling pathway
- GO:0050857 positive regulation of antigen receptor-mediated signaling pathway
- GO:0051209 release of sequestered calcium ion into cytosol
- GO:0051607 defense response to virus
MCA87SBV475
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Human ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up