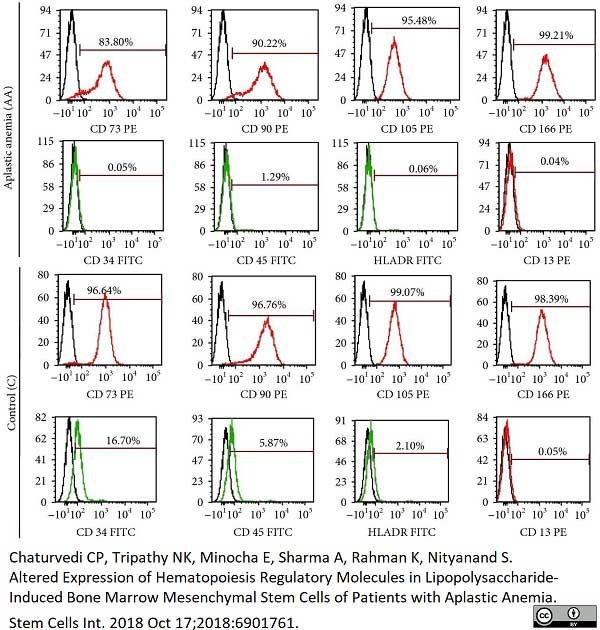
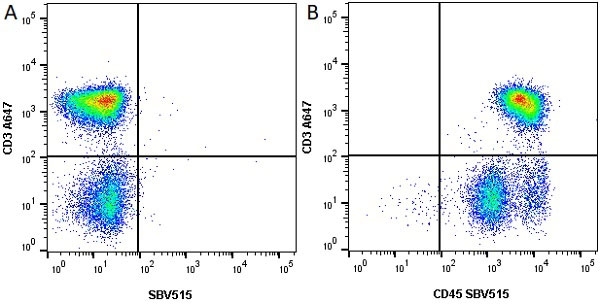
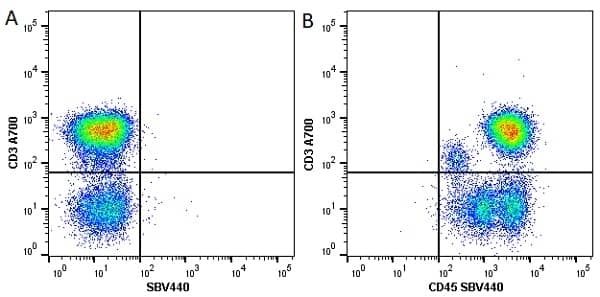
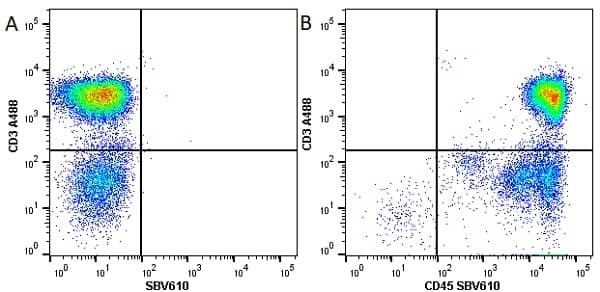
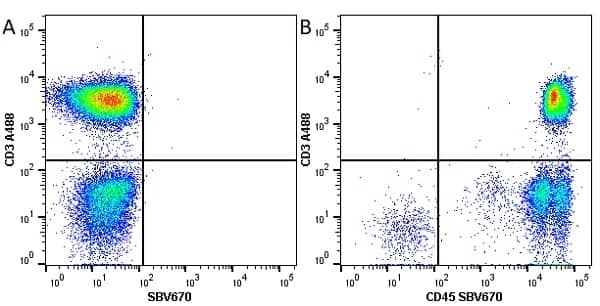
CD45 antibody | F10-89-4







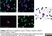



































Mouse anti Human CD45:Alexa Fluor® 647
- Product Type
- Monoclonal Antibody
- Clone
- F10-89-4
- Isotype
- IgG2a
- Specificity
- CD45
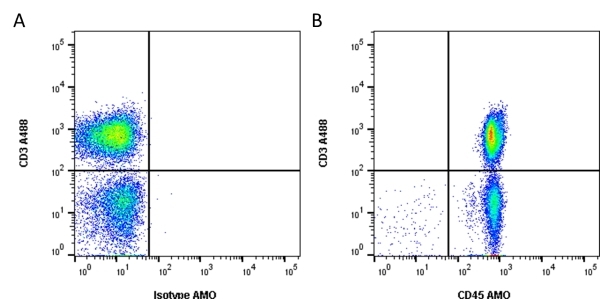
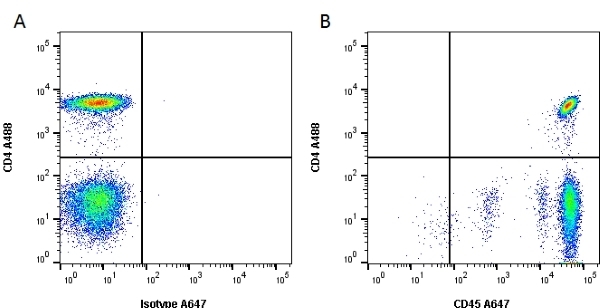
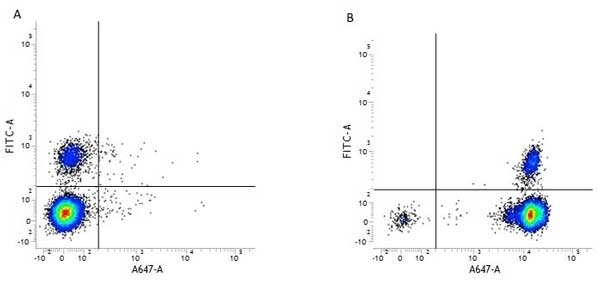
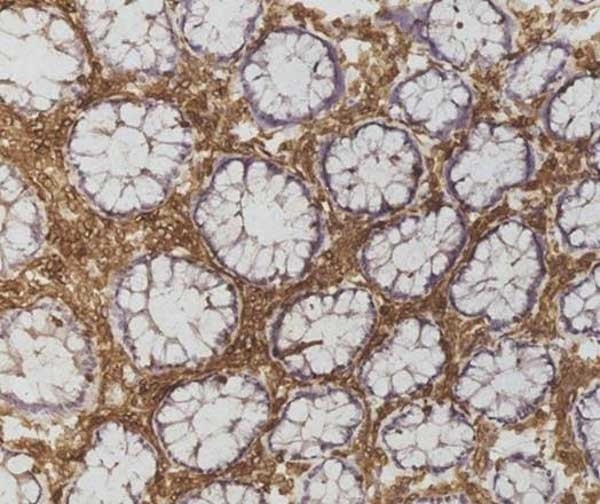
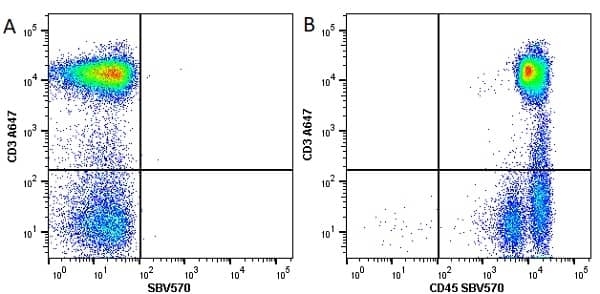
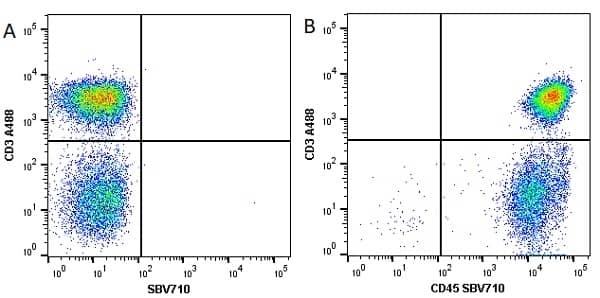
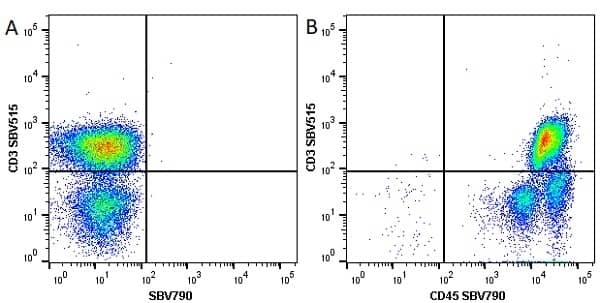
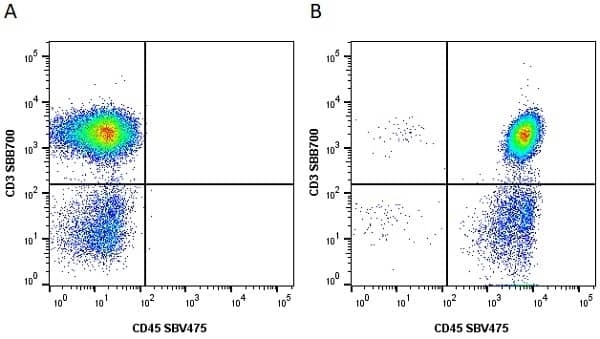
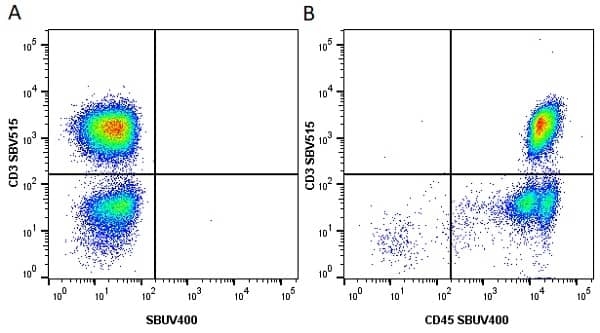
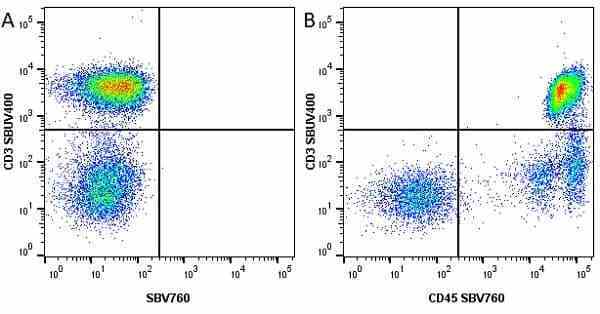
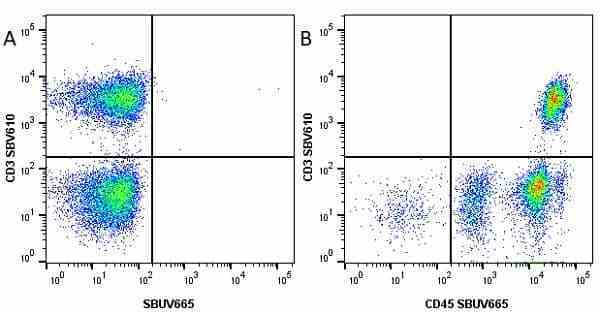
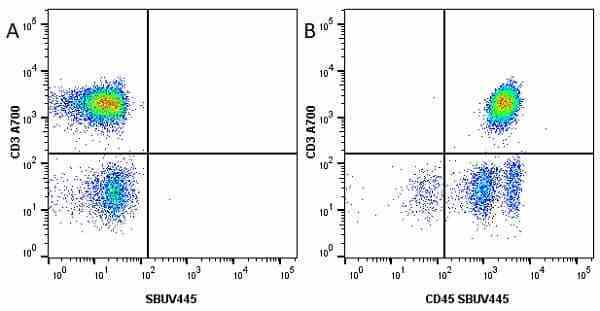
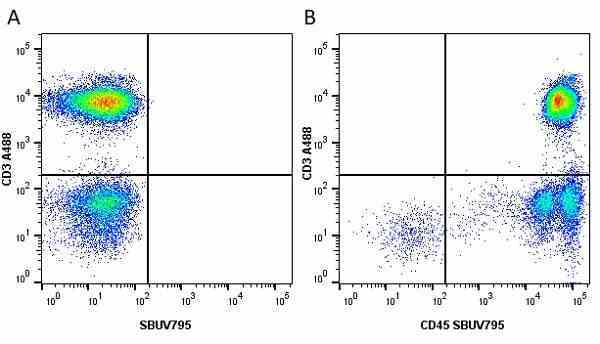
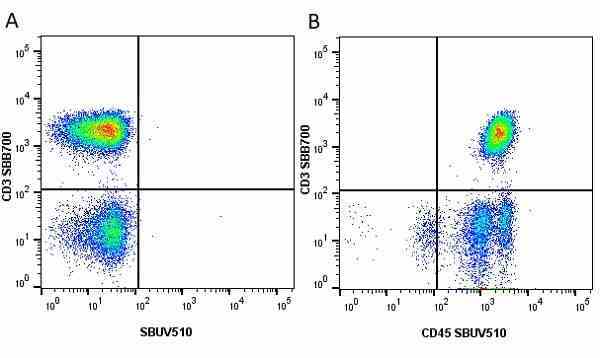
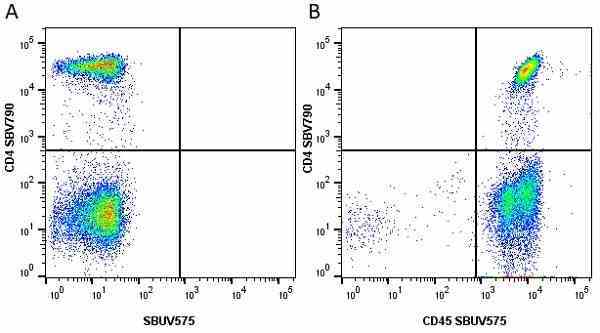
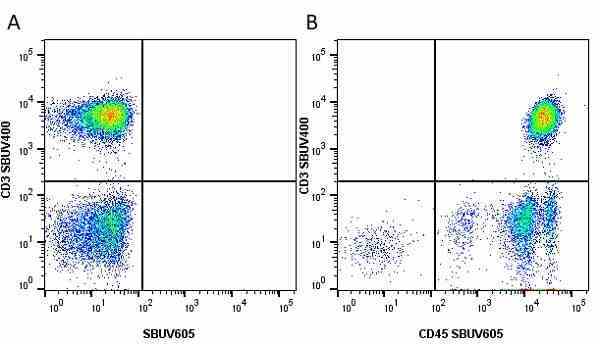
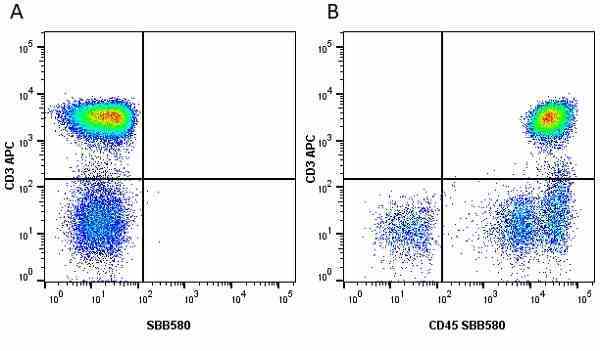
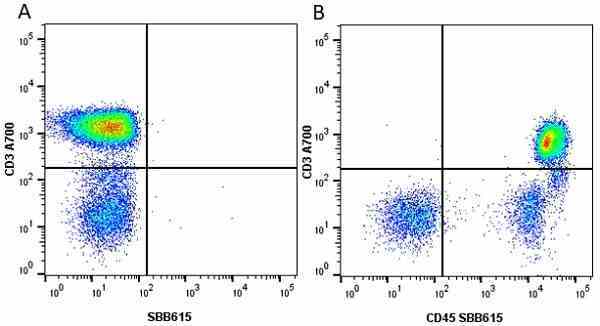
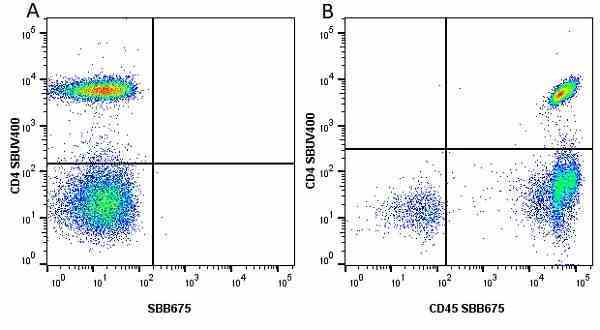
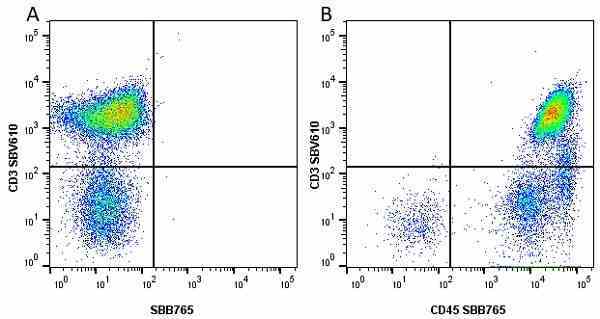
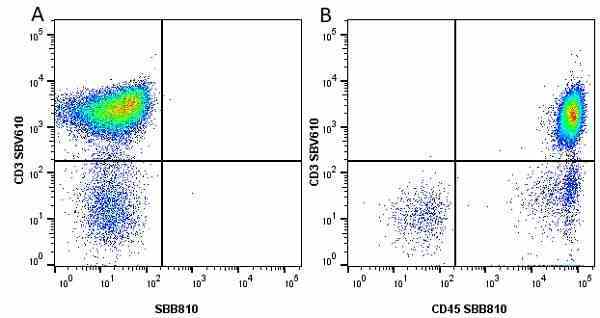
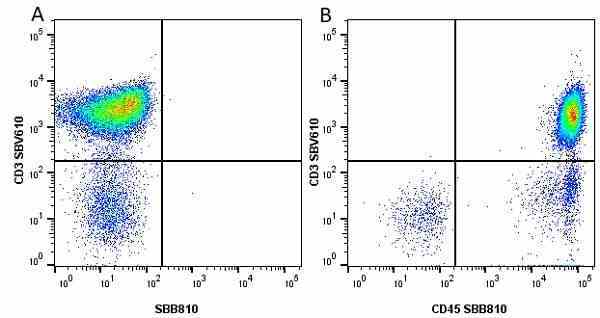
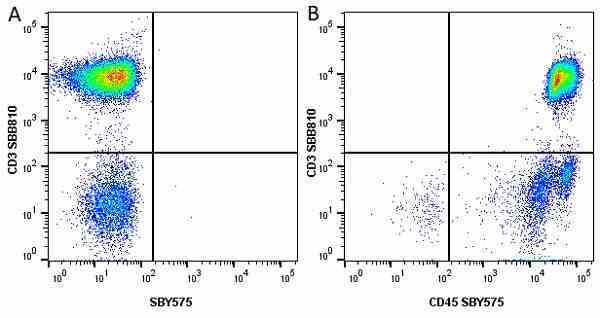
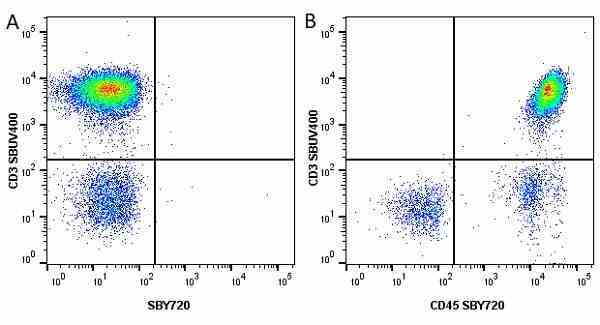
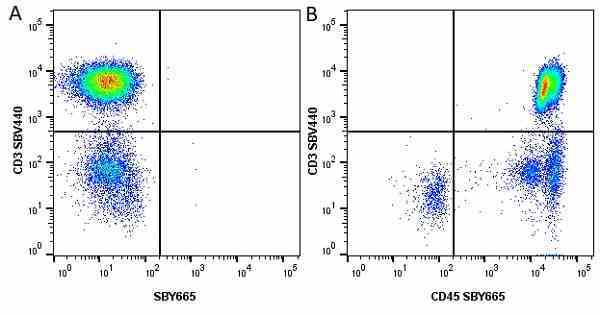
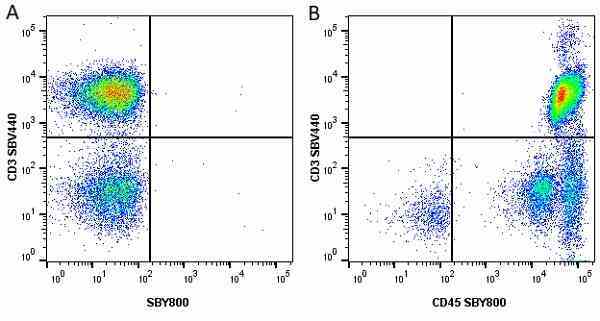
| Mouse anti Human CD45 antibody, clone F10-89-4 recognizes the human CD45 cell surface antigen, also known as leucocyte common antigen (LCA). CD45 is a complex molecule existing in a number of isoforms. Antibodies recognizing a common epitope on all of these isoforms are termed CD45 whilst those recognizing only individual isoforms are termed CD45RA or CD45RO etc. Mouse anti Human CD45 antibody, clone F10-89-4 reacts with all forms of CD45 expressed by all haematopoietic cells, except erythrocytes, having a higher level of expression on lymphocytes than on granulocytes. It is routinely tested in flow cytometry on human peripheral blood leucocytes. Mouse anti Human CD45 antibody, clone F10-89-4, has been validated for use on the Genesis Cell Isolation System with the CelSelect SlideTM technology. |

|
- Target Species
- Human
- Product Form
- Purified IgG conjugated to Alexa Fluor® 647 - liquid
- Preparation
- Purified IgG prepared by affinity chromatography on Protein A from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
0.09% Sodium Azide 1% Bovine Serum Albumin - Immunogen
- Human T lymphocytes.
- Approx. Protein Concentrations
- IgG concentration 0.05 mg/ml
- Fusion Partners
- Spleen cells from immunized BALB/c mice were fused with cells of the mouse NS-1 myeloma cell line.
- Max Ex/Em
-
Fluorophore Excitation Max (nm) Emission Max (nm) Alexa Fluor®647 650 665 - Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
- Acknowledgements
- This product is provided under an intellectual property licence from Life Technologies Corporation. The transfer of this product is contingent on the buyer using the purchase product solely in research, excluding contract research or any fee for service research, and the buyer must not sell or otherwise transfer this product or its components for (a) diagnostic, therapeutic or prophylactic purposes; (b) testing, analysis or screening services, or information in return for compensation on a per-test basis; (c) manufacturing or quality assurance or quality control, or (d) resale, whether or not resold for use in research. For information on purchasing a license to this product for purposes other than as described above, contact Life Technologies Corporation, 5791 Van Allen Way, Carlsbad CA 92008 USA or outlicensing@thermofisher.com
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended. This product is photosensitive and should be protected from light.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Flow Cytometry | Neat | 1/2 Pack Size: 100 Tests/1ml Neat Pack Size: 25 Tests/0.25ml |
- Flow Cytometry
- Use 10ul of the suggested working dilution to label 106 cells or 100ul whole blood.
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Mouse IgG2a Negative Control:Alexa Fluor® 647 | MCA929A647 | F | 100 Tests/1ml |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse IgG2a Negative Control:Alexa Fluor® 647 | ||||||
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Human Seroblock | BUF070A | F | 50 Test | Log in | |||
| List Price | Your Price | ||||||
| Log in | |||||||
| Description | Human Seroblock | ||||||
| Human Seroblock | BUF070B | F | 200 Test | Log in | |||
| List Price | Your Price | ||||||
| Log in | |||||||
| Description | Human Seroblock | ||||||
Source Reference
-
Dalchau, R. et al. (1980) Monoclonal antibody to a human leukocyte-specific membrane glycoprotein probably homologous to the leukocyte-common (L-C) antigen of the rat.
Eur J Immunol. 10 (10): 737-44.
References for CD45 antibody
-
Quenby, S et al. (1999) Pre-implantation endometrial leukocytes in women with recurrent miscarriage.
Human Reprod. 14(9):2386-2391. -
Hauser, P.V. et al. (2010) Stem cells derived from human amniotic fluid contribute to acute kidney injury recovery.
Am J Pathol. 177: 2011-21. -
Mallam, E. et al. (2010) Characterization of in vitro expanded bone marrow-derived mesenchymal stem cells from patients with multiple sclerosis.
Mult Scler. 16: 909-18. -
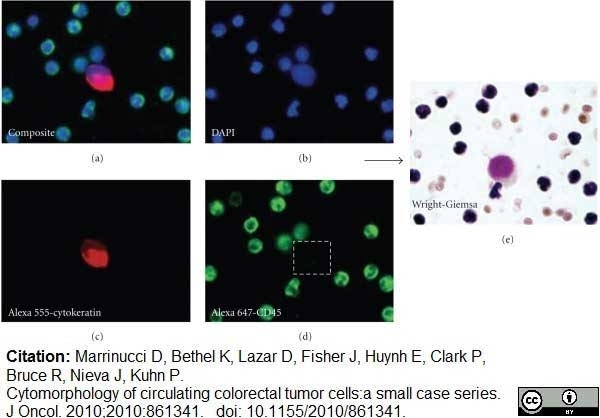
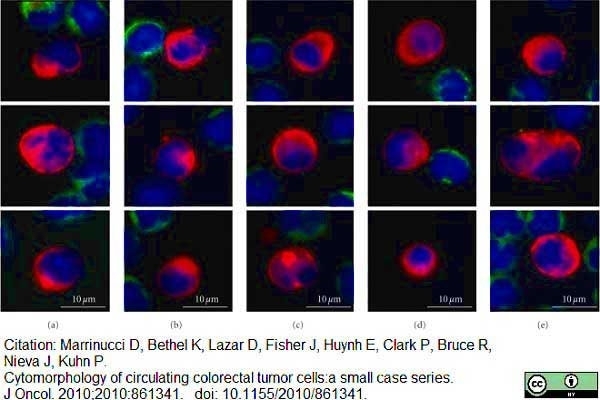
Marrinucci, D. et al. (2010) Cytomorphology of circulating colorectal tumor cells:a small case series.
J Oncol. 2010: 861341. -
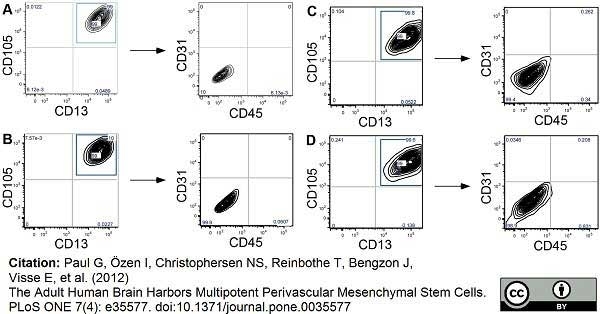
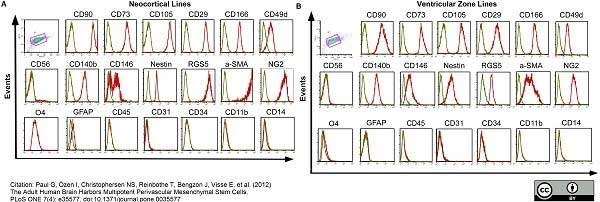
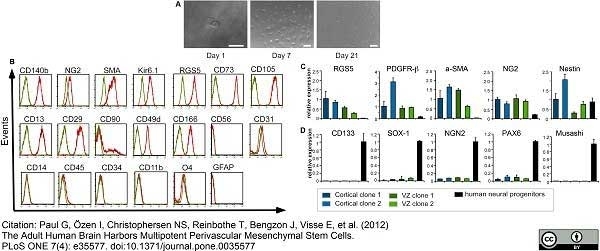
Paul, G. et al. (2012) The adult human brain harbors multipotent perivascular mesenchymal stem cells.
PLoS One. 7: e35577. -
De Schauwer, C. et al. (2012) In search for cross-reactivity to immunophenotype equine mesenchymal stromal cells by multicolor flow cytometry.
Cytometry A. 81 (4): 312-23. -
Kazane, S.A. et al. (2012) Site-specific DNA-antibody conjugates for specific and sensitive immuno-PCR.
Proc Natl Acad Sci U S A. 109: 3731-6. -
Spaas, J.H. et al. (2013) Culture and characterisation of equine peripheral blood mesenchymal stromal cells.
Vet J. 195 (1): 107-13.
View The Latest Product References
-
Sadarangani, A. et al. (2015) GLI2 inhibition abrogates human leukemia stem cell dormancy.
J Transl Med. 13: 98. -
Gunawardene, P. et al. (2015) Association Between Circulating Osteogenic Progenitor Cells and Disability and Frailty in Older Persons: The Nepean Osteoporosis and Frailty Study.
J Gerontol A Biol Sci Med Sci. pii: glv190. -
Mohamed Suhaimi, N.A. et al. (2015) Non-invasive sensitive detection of KRAS and BRAF mutation in circulating tumor cells of colorectal cancer patients.
Mol Oncol. 9 (4): 850-60. -
Ruiz, C. et al. (2015) Limited genomic heterogeneity of circulating melanoma cells in advanced stage patients.
Phys Biol. 12 (1): 016008. -
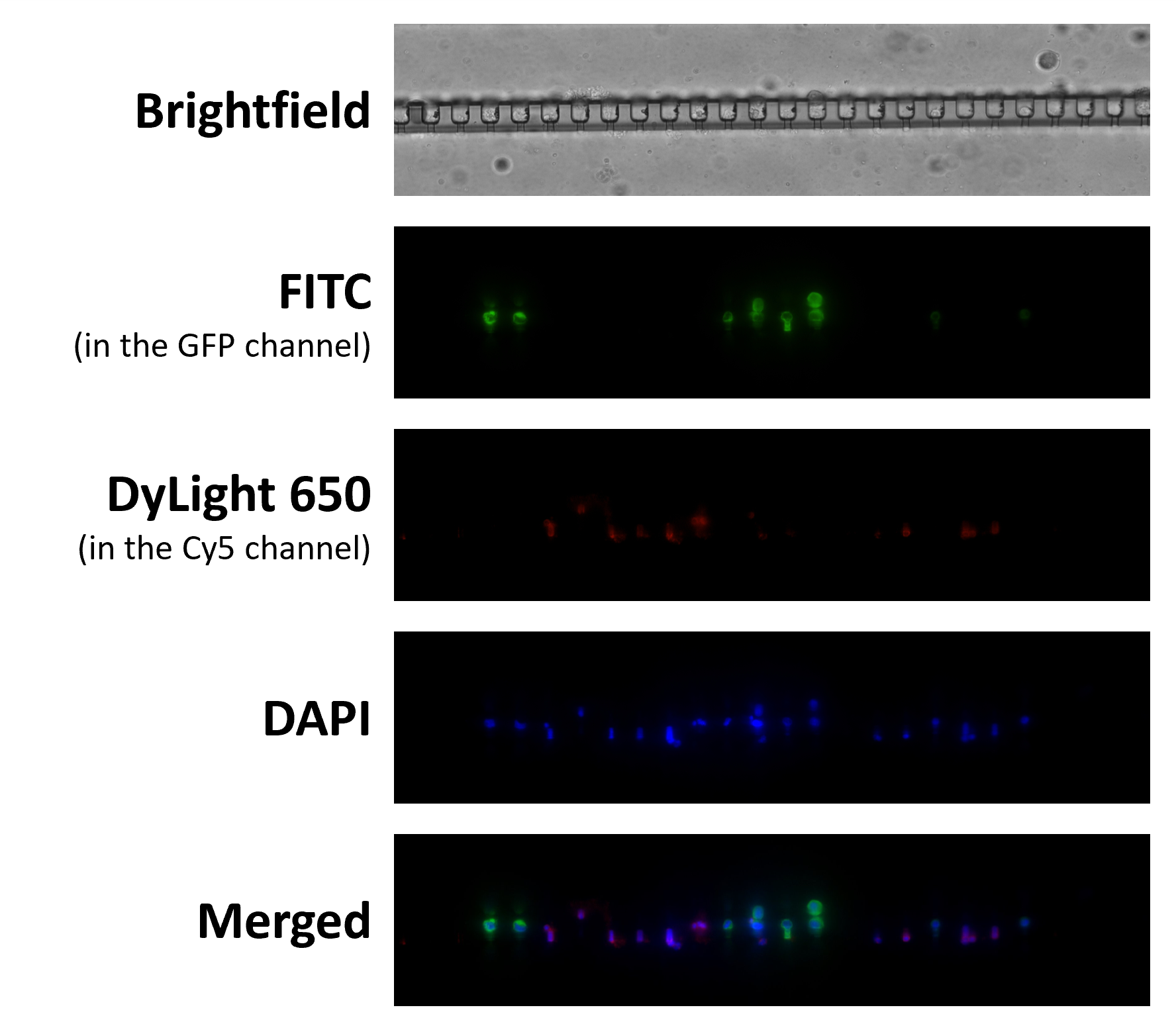
Gogoi P et al. (2016) Development of an Automated and Sensitive Microfluidic Device for Capturing and Characterizing Circulating Tumor Cells (CTCs) from Clinical Blood Samples.
PLoS One. 11 (1): e0147400. -
Gomiero, C. et al. (2016) Tenogenic induction of equine mesenchymal stem cells by means of growth factors and low-level laser technology.
Vet Res Commun. 40 (1): 39-48. -
Bianchessi, M. et al. (2016) Effect of Fibroblast Growth Factor 2 on Equine Synovial Fluid Chondroprogenitor Expansion and Chondrogenesis.
Stem Cells Int. 2016: 9364974. -
Branly, T. et al. (2017) Characterization and use of Equine Bone Marrow Mesenchymal Stem Cells in Equine Cartilage Engineering. Study of their Hyaline Cartilage Forming Potential when Cultured under Hypoxia within a Biomaterial in the Presence of BMP-2 and TGF-β1.
Stem Cell Rev Rep. 13 (5): 611-30. -
GarikipatiV, N.S. et al. (2018) Isolation and characterization of mesenchymal stem cells from human fetus heart.
PLoS One. 13 (2): e0192244. -
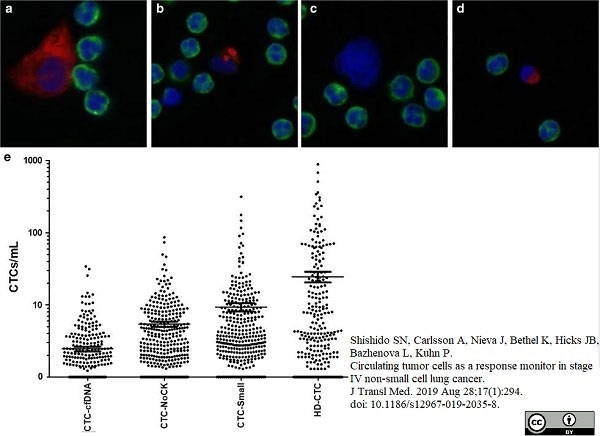
Shishido, S.N. et al. (2019) Circulating tumor cells as a response monitor in stage IV non-small cell lung cancer.
J Transl Med. 17 (1): 294. -
Welter, L. et al. (2020) Treatment response and tumor evolution: lessons from an extended series of multianalyte liquid biopsies in a metastatic breast cancer patient.
Cold Spring Harb Mol Case Stud. 6 (6): a005819. -
Ndacayisaba, L.J. et al. (2022) Enrichment-Free Single-Cell Detection and Morphogenomic Profiling of Myeloma Patient Samples to Delineate Circulating Rare Plasma Cell Clones
Curr Oncol. 29 (5): 2954-72. -
Shishido, S.N. et al. (2022) Liquid Biopsy Landscape in Patients with Primary Upper Tract Urothelial Carcinoma.
Cancers (Basel). 14 (12): 3007. -
Chai, S. et al. (2022) Identification of epithelial and mesenchymal circulating tumor cells in clonal lineage of an aggressive prostate cancer case.
NPJ Precis Oncol. 6 (1): 41. -
Zhu, J. et al. (2022) Sequential Method for Analysis of CTCs and Exosomes from the Same Sample of Patient Blood.
ACS Omega. 7 (42): 37581-88. -
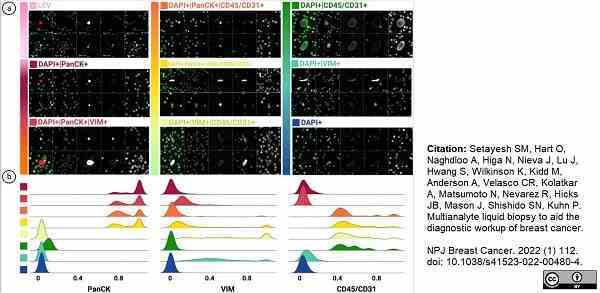
Setayesh, S.M. et al. (2022) Multianalyte liquid biopsy to aid the diagnostic workup of breast cancer.
NPJ Breast Cancer. 8 (1): 112. -
Ndacayisaba, L.J. et al. (2022) Characterization of BCMA Expression in Circulating Rare Single Cells of Patients with Plasma Cell Neoplasms.
Int J Mol Sci. 23 (21): 13427. -
Qi, E. et al. (2023) Investigation of liquid biopsy analytes in peripheral blood of individuals after SARS-CoV-2 infection.
EBioMedicine. 90: 104519. -
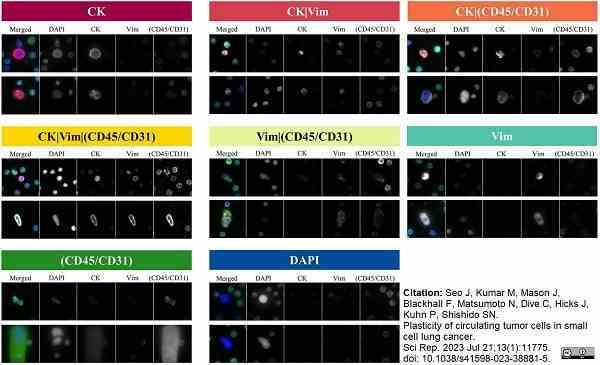
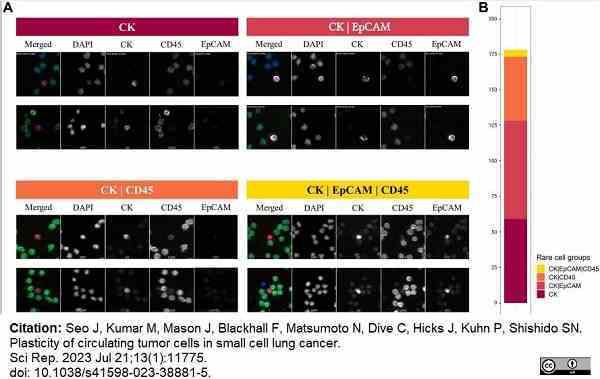
Seo, J. et al. (2023) Plasticity of circulating tumor cells in small cell lung cancer.
Sci Rep. 13 (1): 11775. -
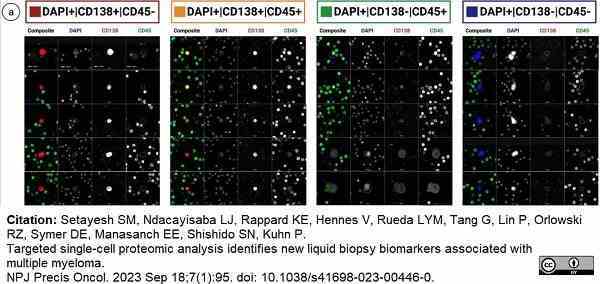
Setayesh, S.M. et al. (2023) Targeted single-cell proteomic analysis identifies new liquid biopsy biomarkers associated with multiple myeloma.
NPJ Precis Oncol. 7 (1): 95. -
Welter, L. et al. (2023) Cell State and Cell Type: Deconvoluting Circulating Tumor Cell Populations in Liquid Biopsies by Multi-Omics.
Cancers (Basel). 15 (15): 3949. -
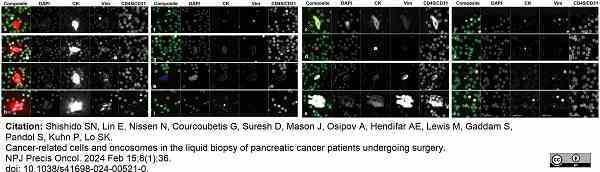
Shishido, S.N. et al. (2024) Cancer-related cells and oncosomes in the liquid biopsy of pancreatic cancer patients undergoing surgery.
NPJ Precis Oncol. 8 (1): 36.
- Synonyms
- LCA
- RRID
- AB_324730
- UniProt
- P08575
- Entrez Gene
- PTPRC
- GO Terms
- GO:0001915 negative regulation of T cell mediated cytotoxicity
- GO:0001960 negative regulation of cytokine-mediated signaling pathway
- GO:0002378 immunoglobulin biosynthetic process
- GO:0005001 transmembrane receptor protein tyrosine phosphatase activity
- GO:0005887 integral to plasma membrane
- GO:0005925 focal adhesion
- GO:0006469 negative regulation of protein kinase activity
- GO:0006470 protein dephosphorylation
- GO:0007411 axon guidance
- View More GO Terms
- GO:0019901 protein kinase binding
- GO:0030890 positive regulation of B cell proliferation
- GO:0033261 regulation of S phase
- GO:0042100 B cell proliferation
- GO:0045121 membrane raft
- GO:0050852 T cell receptor signaling pathway
- GO:0050853 B cell receptor signaling pathway
- GO:0050857 positive regulation of antigen receptor-mediated signaling pathway
- GO:0051209 release of sequestered calcium ion into cytosol
- GO:0051607 defense response to virus
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Human ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up