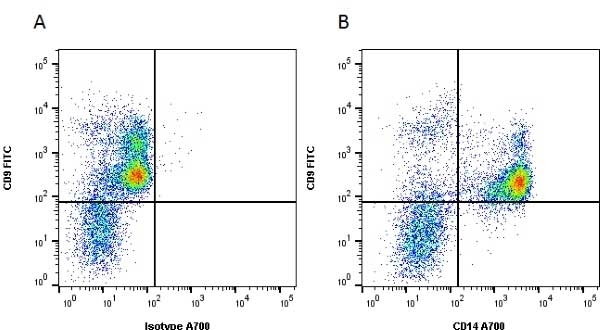
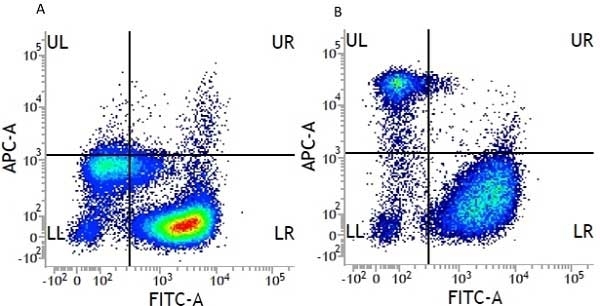
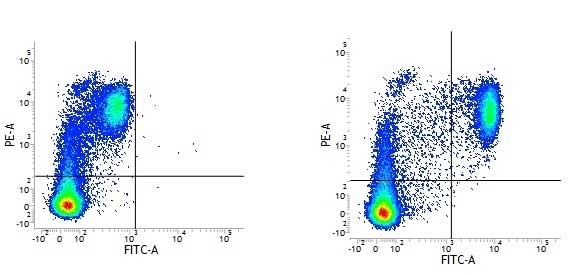
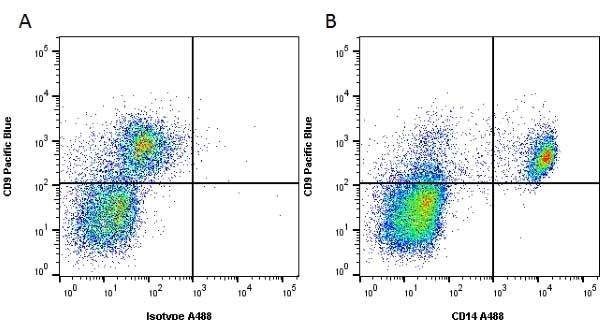
CD14 antibody | UCHM1





Mouse anti Human CD14:FITC
- Product Type
- Monoclonal Antibody
- Clone
- UCHM1
- Isotype
- IgG2a
- Specificity
- CD14
| Mouse anti Human CD14 antibody, clone UCHM1 recognizes a cell surface antigen of ~55 kDa, known as CD14.The CD14 molecule is found predominantly on monocytes and macrophages in flow cytometry, it is less strongly expressed on granulocytes, and is absent from stem cells and myeloid cells of very early differentiation states. In immunohistology the CD14 molecule is found to be present on Langerhans cells, follicular dendritic cells, histiocytes and high endothelial venules. Antibodies to the CD14 molecule are known to induce oxidative burst formation. In tonsil tissue sections UCHM1 gives positive staining reactions with monocytic cells, the interfollicular tissue macrophages seen under the capsule, and dendritic reticulum cells. Skin Langerhans cells are always negative (Hogg et al. 1984). UCHM1 also reacts with Kupffer cells and sinus lining cells on the liver. |
- Target Species
- Human
- Species Cross-Reactivity
-
Target Species Cross Reactivity Cynomolgus monkey Rhesus Monkey Fish Trout - N.B. Antibody reactivity and working conditions may vary between species.
- Product Form
- Purified IgG conjugated to Fluorescein Isothiocyanate Isomer 1 (FITC) - liquid
- Preparation
- Purified IgG prepared by affinity chromatography on Protein A from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
0.09% Sodium Azide 1% Bovine Serum Albumin - Immunogen
- Human Thymocytes followed by peripheral blood mononuclear cells.
- Approx. Protein Concentrations
- IgG concentration 0.1mg/ml
- Fusion Partners
- Spleen cells from immunized BALB/c mice were fused with cells from the NS1-Ag4/1 mouse myeloma line.
- Max Ex/Em
-
Fluorophore Excitation Max (nm) Emission Max (nm) FITC 490 525 - Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended. This product is photosensitive and should be protected from light.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Flow Cytometry | Neat | 1/10 |
- Flow Cytometry
- Use 10ul of the suggested working dilution to label 106 cells or 100ul whole blood
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Mouse IgG2a Negative Control:FITC | MCA929F | F | 100 Tests |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse IgG2a Negative Control:FITC | ||||||
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Human Seroblock | BUF070A | F | 50 Test | Log in | |||
| List Price | Your Price | ||||||
| Log in | |||||||
| Description | Human Seroblock | ||||||
| Human Seroblock | BUF070B | F | 200 Test | Log in | |||
| List Price | Your Price | ||||||
| Log in | |||||||
| Description | Human Seroblock | ||||||
Source Reference
-
Hogg, N. et al. (1984) Monoclonal antibodies specific for human monocytes, granulocytes and endothelium.
Immunology. 53 (4): 753-67.
References for CD14 antibody
-
Linch, D.C. et al. (1984) Monoclonal antibodies differentiating between monocytic and nonmonocytic variants of AML.
Blood. 63 (3): 566-73. -
Hogg, N. & Horton, M.A. (1987) Myeloid antigens: new and previously defined clusters
in Leucocyte Typing III White Cell differentiation antigens. Edited by McMichael, A.J.,et al. -
Jonker, M. et al. (1989) Reactivity of mAb specific for human CD markers with Rhesus monkey leucocytes.
Leucocyte Typing IV. Oxford University Press p 1058-63. -
Hsu, T.L. et al. (2002) Modulation of dendritic cell differentiation and maturation by decoy receptor 3.
J Immunol. 168: 4846-53. -
Karlsson, H. et al. (2002) Innate immune responses of human neonatal cells to bacteria from the normal gastrointestinal flora.
Infect Immun. 70: 6688-96. -
Kämmerer, U. et al. (2003) Unique appearance of proliferating antigen-presenting cells expressing DC-SIGN (CD209) in the decidua of early human pregnancy.
Am J Pathol. 162: 887-96. -
Köller, M. et al. (2004) Phenotypic and functional deficiencies of monocyte-derived dendritic cells in systemic lupus erythematosus (SLE) patients.
Int Immunol. 16: 1595-604. -
Goddard, S. et al. (2004) Interleukin-10 secretion differentiates dendritic cells from human liver and skin.
Am J Pathol. 164: 511-9.
View The Latest Product References
-
Chang, Y.C. et al. (2004) Modulation of macrophage differentiation and activation by decoy receptor 3.
J Leukoc Biol. 75: 486-94. -
Lin, C.W. et al. (2005) CD94 1A transcripts characterize lymphoblastic lymphoma/leukemia of immature natural killer cell origin with distinct clinical features.
Blood. 106: 3567-74. -
Angel, C.E. et al. (2006) Cutting edge: CD1a+ antigen-presenting cells in human dermis respond rapidly to CCR7 ligands.
J Immunol. 176 (10): 5730-4. -
Fischer, U. and Koellner, B. (2007) Cross-reactivity of human leukocyte differentiation antigen monoclonal antibodies on carp and rainbow trout cells.
Vet Immunol Immunopathol. 119: 142-55. -
Bournazos, S. et al. (2008) Monocyte functional responsiveness after PSGL-1-mediated platelet adhesion is dependent on platelet activation status.
Arterioscler Thromb Vasc Biol. 28: 1491-8. -
Iking-Konert, C. et al. (2008) T lymphocytes in patients with primary vasculitis: expansion of CD8+ T cells with the propensity to activate polymorphonuclear neutrophils.
Rheumatology (Oxford). 47: 609-16. -
Angel, C.E. et al. (2009) Distinctive localization of antigen-presenting cells in human lymph nodes.
Blood. 113: 1257-67. -
Brook, F.A. et al. (2010) Derivation and characterisation of the human embryonic stem cell line, OxF1.
In Vitro Cell Dev Biol Anim. 46: 173-7. -
Hovden, A.O. et al. (2011) Maturation of monocyte derived dendritic cells with OK432 boosts IL-12p70 secretion and conveys strong T-cell responses.
BMC Immunol. 12:2. -
Din JN et al. (2013) Effect of ω-3 fatty acid supplementation on endothelial function, endogenous fibrinolysis and platelet activation in male cigarette smokers.
Heart. 99 (3): 168-74. -
Bromberek, J.L. et al. (2016) Breed Distribution and Clinical Characteristics of B Cell Chronic Lymphocytic Leukemia in Dogs.
J Vet Intern Med. 30 (1): 215-22. -
Spiller, K.L. et al. (2016) Differential gene expression in human, murine, and cell line-derived macrophages upon polarization.
Exp Cell Res. 347 (1): 1-13. -
Kannegieter, N.M. et al. (2018) Analysis of NFATc1 amplification in T cells for pharmacodynamic monitoring of tacrolimus in kidney transplant recipients.
PLoS One. 13 (7): e0201113. -
Wu, T.C. et al. (2018) IL1 Receptor Antagonist Controls Transcriptional Signature of Inflammation in Patients with Metastatic Breast Cancer.
Cancer Res. 78 (18): 5243-58. -
Hoang, P.T. et al. (2018) Subtype Diversification and Synaptic Specificity of Stem Cell-Derived Spinal Interneurons.
Neuron. 100 (1): 135-149.e7. -
Matsusaka, K. et al. (2022) Distinct roles in phagocytosis of the early and late increases of cell surface calreticulin induced by oxaliplatin
Biochem Biophys Rep. 29: 101222.
- RRID
- AB_321314
- UniProt
- P08571
- Entrez Gene
- CD14
- GO Terms
- GO:0005886 plasma membrane
- GO:0001530 lipopolysaccharide binding
- GO:0001847 opsonin receptor activity
- GO:0006915 apoptosis
- GO:0006909 phagocytosis
- GO:0006954 inflammatory response
- GO:0008063 Toll signaling pathway
- GO:0031225 anchored to membrane
- GO:0016019 peptidoglycan receptor activity
- View More GO Terms
- GO:0032760 positive regulation of tumor necrosis factor production
- GO:0045087 innate immune response
- GO:0070891 lipoteichoic acid binding
- GO:0071222 cellular response to lipopolysaccharide
- GO:0071223 cellular response to lipoteichoic acid
MCA596F
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Human ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up