CD34 antibody | 1H6


Mouse anti Dog CD34:FITC
- Product Type
- Monoclonal Antibody
- Clone
- 1H6
- Isotype
- IgG1
- Specificity
- CD34
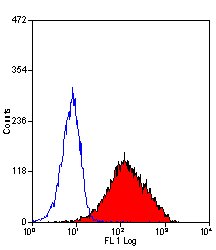
| Mouse anti dog CD34 antibody, clone 1H6 recognizes the canine homologue of CD34, a glycosylated type 1 transmembrane protein of approximately 110 kDa (McSweeney et al. 1998) expressed on the cell suface of endothelial cells and haematopoietic stem cells. Mouse anti dog CD34 antibody, clone 1H6 is a key marker of canine hematopoietic progenitor cells and is reported for use in CD34+ enrichment assays, (Goerner et al. 2001) and (Horn et al. 2004). |
- Target Species
- Dog
- Product Form
- Purified IgG conjugated to Fluorescein Isothiocyanate Isomer 1 (FITC) - liquid
- Preparation
- Purified IgG prepared by affinity chromatography on Protein A from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
0.09% Sodium Azide 1% Bovine Serum Albumin - Immunogen
- Canine CD34 fusion protein.
- Approx. Protein Concentrations
- IgG concentration 0.1 mg/ml
- Fusion Partners
- Spleen cells from immunized BALB/c mice were fused with cells of the mouse NS-1/FOX-NY myeloma cell line.
- Max Ex/Em
-
Fluorophore Excitation Max (nm) Emission Max (nm) FITC 490 525 - Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended. This product is photosensitive and should be protected from light.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Flow Cytometry | Neat | 1/10 |
- Flow Cytometry
- Use 10ul of the suggested working dilution to label 1x106 cells in 100ul.
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Mouse IgG1 Negative Control:FITC | MCA928F | F | 100 Tests |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse IgG1 Negative Control:FITC | ||||||
Source Reference
-
McSweeney, P.A. et al. (1998) Characterization of monoclonal antibodies that recognize canine CD34.
Blood. 91 (6): 1977-86.
References for CD34 antibody
-
Goerner, M. et al. (1999) The use of granulocyte colony-stimulating factor during retroviral transduction on fibronectin fragment CH-296 enhances gene transfer into hematopoietic repopulating cells in dogs.
Blood. 94 (7): 2287-92. -
Bhattacharya, V. et al. (2000) Enhanced endothelialization and microvessel formation in polyester grafts seeded with CD34(+) bone marrow cells.
Blood. 95 (2): 581-5. -
Goerner, M. et al. (2001) Sustained multilineage gene persistence and expression in dogs transplanted with CD34(+) marrow cells transduced by RD114-pseudotype oncoretrovirus vectors.
Blood. 98 (7): 2065-70. -
Georges, G. et al. (2001) Engraftment of DLA-haploidentical marrow with ex vivo expanded, retrovirally transduced cytotoxic T lymphocytes.
Blood. 98:3447-55. -
Horn, P.A. et al. (2004) Efficient lentiviral gene transfer to canine repopulating cells using an overnight transduction protocol.
Blood. 103 (10): 3710-6. -
Avallone, G. et al. (2007) The spectrum of canine cutaneous perivascular wall tumors: morphologic, phenotypic and clinical characterization.
Vet Pathol. 44 (5): 607-20. -
Palmieri, C. et al. (2013) Use of electron microscopy to classify canine perivascular wall tumors.
Vet Pathol. 50 (2): 226-33. -
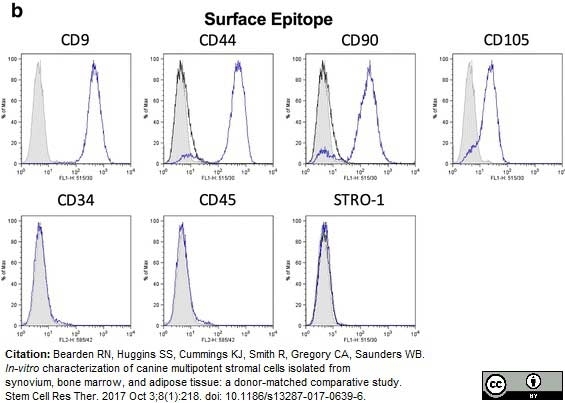
Bearden, R.N. et al. (2017) In-vitro characterization of canine multipotent stromal cells isolated from synovium, bone marrow, and adipose tissue: a donor-matched comparative study.
Stem Cell Res Ther. 8 (1): 218.
View The Latest Product References
-
Trindade, A.B. et al. (2017) Mesenchymal-like stem cells in canine ovary show high differentiation potential.
Cell Prolif. Oct 08 [Epub ahead of print]. -
Lee, S.H. et al. (2016) Impact of local injection of brain-derived neurotrophic factor-expressing mesenchymal stromal cells (MSCs) combined with intravenous MSC delivery in a canine model of chronic spinal cord injury.
Cytotherapy. Oct 28 [Epub ahead of print]. -
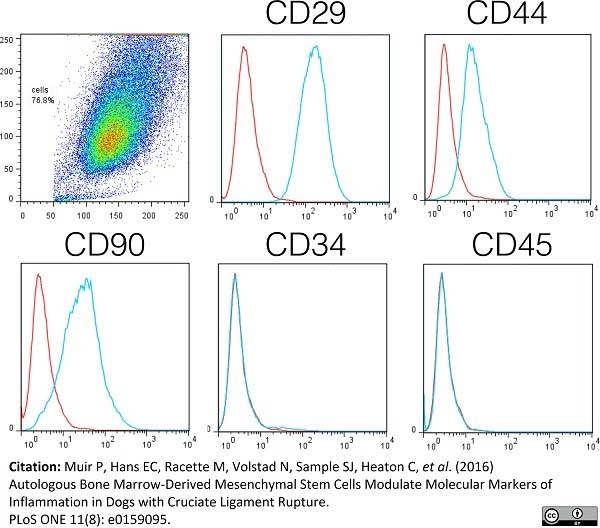
Muir, P. et al. (2016) Autologous Bone Marrow-Derived Mesenchymal Stem Cells Modulate Molecular Markers of Inflammation in Dogs with Cruciate Ligament Rupture.
PLoS One. 11 (8): e0159095. -
Rajawat, Y.S. et al. (2021) In Vivo Gene Therapy for Canine SCID-X1 Using Cocal-Pseudotyped Lentiviral Vector.
Hum Gene Ther. 32 (1-2): 113-27. -
Grudzien, M. et al. (2021) A newly established canine NK-type cell line and its cytotoxic properties.
Vet Comp Oncol. 19 (3): 567-77. -
Tongu, E.A.O. et al. (2021) Allogenic mesenchymal stem cell-conditioned medium does not affect sperm parameters and mitigates early endometrial inflammatory responses in mares.
Theriogenology. 169: 1-8. -
Jaensch, S. et al. (2022) Clinicopathologic and immunophenotypic features in dogs with presumptive large granular lymphocyte leukaemia
Australian Veterinary Journal. [Epub ahead of print]. -
Salari Sedigh, H. et al. (2023) In vitro investigation of canine periodontal ligament-derived mesenchymal stem cells: A possibility of promising tool for periodontal regeneration.
J Oral Biol Craniofac Res. 13 (3): 403-11. -
Papa, P.M. et al. (2023) Intratesticular transplantation of allogenic mesenchymal stem cells mitigates testicular destruction after induced heat stress in Miniature-horse stallions.
J Equine Vet Sci. 132: 104961. -
Rezaei, M. et al. (2019) Transplantation of Bone Marrow-Derived Mesenchymal Stem Cells, Platelet-Rich Plasma, and Fibrin Glue for Periodontal Regeneration.
Int J Periodontics Restorative Dent. 39 (1): e32-e45. -
Yang, V.K. et al. (2021) Intravenous administration of allogeneic Wharton jelly-derived mesenchymal stem cells for treatment of dogs with congestive heart failure secondary to myxomatous mitral valve disease.
Am J Vet Res. 82 (6): 487-93. -
Crain, S.K. et al. (2019) Extracellular Vesicles from Wharton's Jelly Mesenchymal Stem Cells Suppress CD4 Expressing T Cells Through Transforming Growth Factor Beta and Adenosine Signaling in a Canine Model.
Stem Cells Dev. 28 (3): 212-26. -
Sheng, R. et al. (2023) Prognostic significance of CD25 expression in dogs with a noninvasive diagnosis of B-cell lymphoma treated with CHOP chemotherapy.
Vet Comp Oncol. 21 (1): 28-35. -
Millanta, F. et al. (2020) Cytologic grading of canine and feline spindle-cell sarcomas of soft tissues and its correlation with histologic grading.
Top Companion Anim Med. 41: 100458.
Further Reading
-
McSweeney, P. et al. (1996) Canine CD34: cloning of the cDNA and evaluation of an antiserum to recombinant protein.
Blood. 88:1992-2003.
- RRID
- AB_2275714
- UniProt
- Q28270
- Entrez Gene
- CD34
- GO Terms
- GO:0016021 integral to membrane
- GO:0030246 carbohydrate binding
- GO:0016337 cell-cell adhesion
- GO:0050900 leukocyte migration
MCA2411F
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Dog ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up