MHC Class II DR antibody | CC108







Mouse anti Bovine MHC Class II DR
- Product Type
- Monoclonal Antibody
- Clone
- CC108
- Isotype
- IgG1
- Specificity
- MHC Class II DR
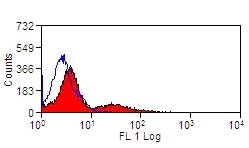
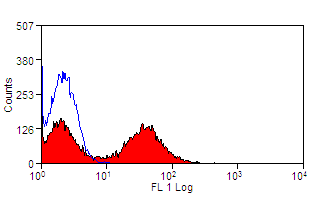
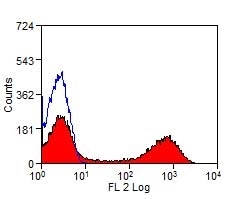
| Mouse anti Bovine MHC class II DR antibody, clone CC108 recognizes Bovine MHC Class II DR. MHC Class II molecules are constitutively expressed on antigen presenting cells such as dendritic cells, B lymphocytes, monocytes, macrophages, activated T lymphocytes and may be induced on a range of other cell types by interferon gamma. The major histocompatibility complex (MHC) is a cluster of genes some of which are important in the immune response to infections. In cattle, this complex is referred to as the bovine leukocyte antigen (BoLA) region. There are 2 major types of MHC class IIa molecules encoded by the BoLA which are DR and DQ each composed of an alpha and beta chain. |
- Target Species
- Bovine
- Product Form
- Purified IgG - liquid
- Preparation
- Purified IgG prepared by affinity chromatography on Protein G from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- 0.09% Sodium Azide (NaN3)
- Carrier Free
- Yes
- Approx. Protein Concentrations
- IgG concentration 1.0mg/ml
- Fusion Partners
- Spleen cells from immunized BALB/c mice were fused with cells of the Mouse NS1 myeloma cell line.
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Flow Cytometry | 1/25 | 1/200 |
- Flow Cytometry
- Use 10ul of the suggested working dilution to label 1x106 cells in 100ul.
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Rabbit F(ab')2 anti Mouse IgG:RPE | STAR12A | F | 1 ml |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Rabbit F(ab')2 anti Mouse IgG:RPE | ||||||
| Rabbit F(ab')2 anti Mouse IgG:FITC | STAR9B | F | 1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Rabbit F(ab')2 anti Mouse IgG:FITC | ||||||
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Mouse IgG1 Negative Control | MCA928 | F | 100 Tests |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse IgG1 Negative Control | ||||||
References for MHC Class II DR antibody
-
Stephens, S.A. & Howard, C.J. (2002) Infection and transformation of dendritic cells from bovine afferent lymph by Theileria annulata.
Parasitology. 124 (Pt 5): 485-93. -
Yamakawa, Y. et al. (2008) Identification and functional characterization of a bovine orthologue to DC-SIGN.
J Leukoc Biol. 83 (6): 1396-403. -
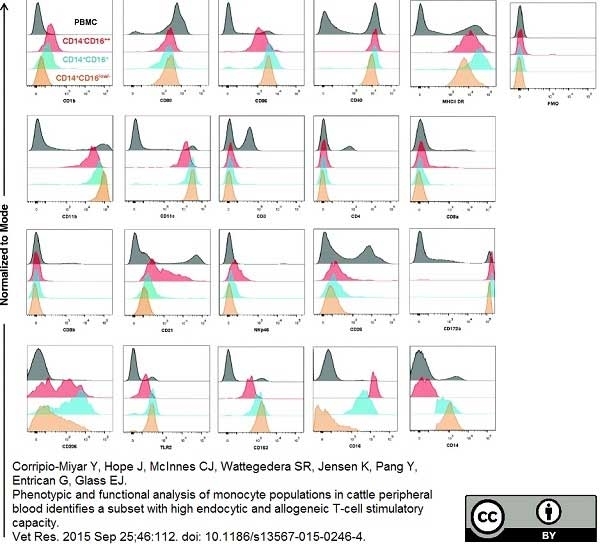
Corripio-Miyar, Y. et al. (2015) Phenotypic and functional analysis of monocyte populations in cattle peripheral blood identifies a subset with high endocytic and allogeneic T-cell stimulatory capacity.
Vet Res. 46: 112. -
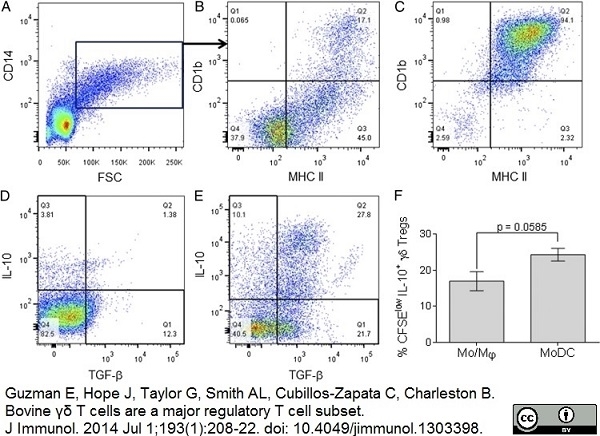
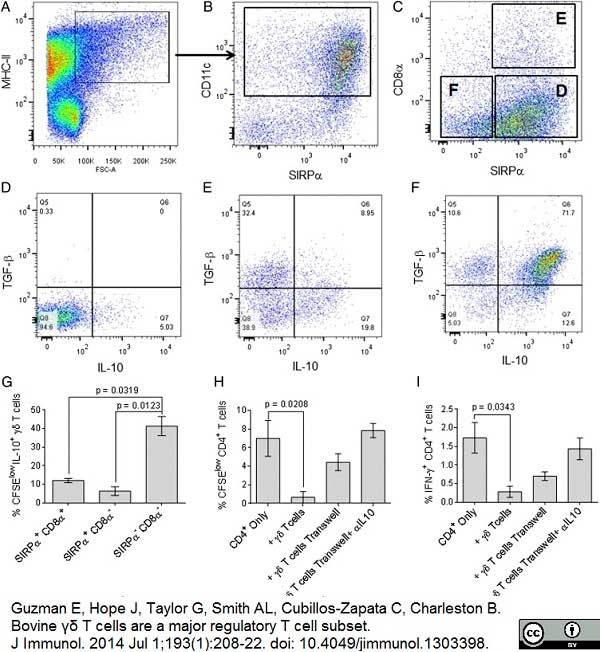
Guzman, E. et al. (2014) Bovine γδ T cells are a major regulatory T cell subset.
J Immunol. 193 (1): 208-22. -
Childerstone, A.J. et al. (1999) Demonstration of bovine CD8+ T-cell responses to foot-and-mouth disease virus.
J Gen Virol. 80 ( Pt 3): 663-9. -
Sopp, P. et al. (1994) Detection of bovine viral diarrhoea virus p80 protein in subpopulations of bovine leukocytes.
J Gen Virol. 75 ( Pt 5): 1189-94. -
Bembridge, G.P. et al. (1995) CD45RO expression on bovine T cells: relation to biological function.
Immunology. 86 (4): 537-44. -
Gibson, A.J. et al. (2016) Differential macrophage function in Brown Swiss and Holstein Friesian cattle.
Vet Immunol Immunopathol. 181: 15-23.
View The Latest Product References
-
Corripio-Miyar, Y. et al. (2017) 1,25-Dihydroxyvitamin D3 modulates the phenotype and function of Monocyte derived dendritic cells in cattle.
BMC Vet Res. 13 (1): 390. -
Risalde, M.A. et al. (2020) BVDV permissiveness and lack of expression of co-stimulatory molecules on PBMCs from calves pre-infected with BVDV.
Comp Immunol Microbiol Infect Dis. 68: 101388. -
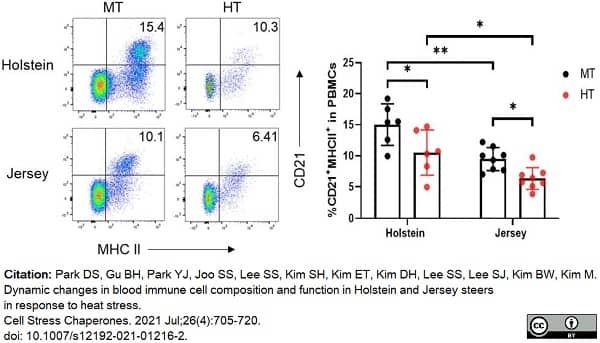
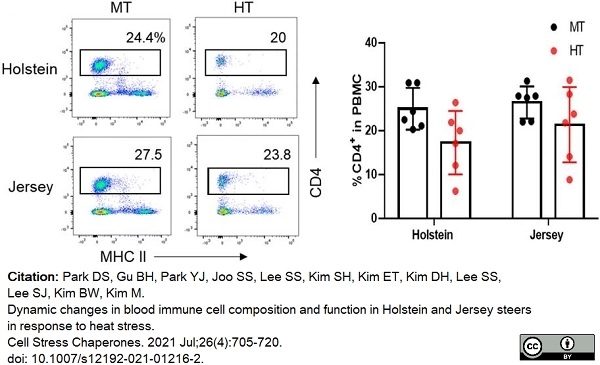
Park, D.S. et al. (2021) Dynamic changes in blood immune cell composition and function in Holstein and Jersey steers in response to heat stress.
Cell Stress Chaperones. 26 (4): 705-20.
- RRID
- AB_10843456
- UniProt
- Q30309
- P79464
MCA5656
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Bovine ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up