IFN Gamma antibody | CC302













Mouse anti Bovine Interferon Gamma:Biotin
- Product Type
- Monoclonal Antibody
- Clone
- CC302
- Isotype
- IgG1
- Specificity
- IFN Gamma
| Mouse anti Bovine IFN-γ antibody, clone CC302 recognizes bovine interferon-gamma, a 143 amino acid cytokine with potent activating, antiviral and anti proliferative properties, produced as a pro-peptide with an additional 23 amino acid N-terminal signal peptide sequence having a molecular weight of ~20 kDa. IFNγ is predominantly secreted by activated T lymphocytes in response to specific mitogens as a result of infection (Rhodes et al. 2000). Mouse anti bovine γ interferon antibody, clone CC302 has been demonstrated to be reactive to a number of mammalian species including human, sheep, dog, pig, goat and mink (Pedersen et al. 2002). Mouse anti Bovine IFNγ antibody, clone CC302 has been used successfully for the evaluation of γ interferon levels in the sera of calves naturally infected with M. avium. subsp paratuberculosis (Appana et al. 2013) as a detection reagent using an ELISA. |

Our IFN Gamma (CC302) Antibody has been referenced in >83 publications* *Based on June 2020 data from CiteAb's antibody search engine. |
- Target Species
- Bovine
- Species Cross-Reactivity
-
Target Species Cross Reactivity Human Pig Dog Horse Sheep Mustelid Expected from Sequence Goat Dolphin Ferret Mink Fin Whale Rabbit - N.B. Antibody reactivity and working conditions may vary between species.
- Product Form
- Purified IgG conjugated to biotin - liquid
- Preparation
- Purified IgG prepared by affinity chromatography on Protein G from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- 0.09% sodium azide (NaN3)
- Approx. Protein Concentrations
- IgG concentration 0.5 mg/ml
- Fusion Partners
- Spleen cells from immunized BALB/c mice were fused with cells of the mouse SP2/0 myeloma cell line.
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| ELISA | 5ug/ml |
- Flow Cytometry
- Use 10μl of the suggested working dilution to label 1x106 cells in 100μl.
- ELISA
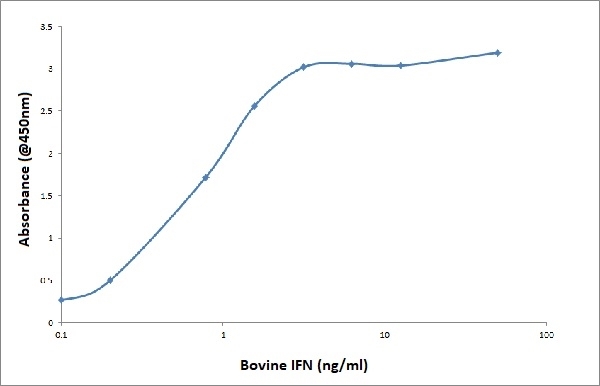
- Biotinylated mouse anti bovine IFNγ, clone CC302, may be used as the detection reagent in a sandwich ELISA with purified mouse anti bovine IFNγ, clone CC330, as the capture reagent and recombinant bovine IFNγ as the standard.
References for IFN Gamma antibody
-
Hasvold, H.J. et al. (2002) In vitro responses to purified protein derivate of caprine T lymphocytes following vaccination with live strains of Mycobacterium avium subsp paratuberculosis.
Vet Immunol Immunopathol. 90 (1-2): 79-89. -
Mwangi, W. et al. (2002) DNA-encoded fetal liver tyrosine kinase 3 ligand and granulocyte macrophage-colony-stimulating factor increase dendritic cell recruitment to the inoculation site and enhance antigen-specific CD4+ T cell responses induced by DNA vaccination of outbred animals.
J Immunol. 169 (7): 3837-46. -
Pedersen, L.G. et al. (2002) Identification of monoclonal antibodies that cross-react with cytokines from different animal species.
Vet Immunol Immunopathol. 88 (3-4): 111-22. -
Aasted, B. et al. (2002) Cytokine profiles in peripheral blood mononuclear cells and lymph node cells from piglets infected in utero with porcine reproductive and respiratory syndrome virus.
Clin Diagn Lab Immunol. 9 (6): 1229-34. -
Cameron, C.M. et al. (2005) Myxoma virus M141R expresses a viral CD200 (vOX-2) that is responsible for down-regulation of macrophage and T-cell activation in vivo.
J Virol. 79: 6052-67. -
Reber, A.J. et al. (2006) Evaluation of multiple immune parameters after vaccination with modified live or killed bovine viral diarrhea virus vaccines.
Comp Immunol Microbiol Infect Dis. 29 (1): 61-77. -
Katepalli, M.P. et al. (2008) The effect of age and telomere length on immune function in the horse.
Dev Comp Immunol. 32 (12): 1409-15. -
Johnson, W.C. et al. (2008) Bovine WC1(-) gammadeltaT cells incubated with IL-15 express the natural cytotoxicity receptor CD335 (NKp46) and produce IFN-gamma in response to exogenous IL-12 and IL-18.
Dev Comp Immunol. 32 (8): 1002-10.
View The Latest Product References
-
Rutigliano, J.A. et al. (2008) Screening monoclonal antibodies for cross-reactivity in the ferret model of influenza infection.
J Immunol Methods. 336: 71-7. -
Papadogiannakis, E.I. et al. (2009) Determination of intracellular cytokines IFN-γ and IL-4 in canine T lymphocytes by flow cytometry following whole-blood culture.
Can J Vet Res. 73: 137-43. -
Jensen, T.H. et al. (2009) Early life DNA vaccination with the H gene of Canine distemper virus induces robust protection against distemper.
Vaccine. 27: 5178-83. -
Nielsen, L. et al. (2009) Lymphotropism and host responses during acute wild-type canine distemper virus infections in a highly susceptible natural host.
J Gen Virol. 90: 2157-65. -
Martel, C.J. & Aasted, B. (2009) Characterization of antibodies against ferret immunoglobulins, cytokines and CD markers.
Vet Immunol Immunopathol. 132:109-15. -
Lybeck, K.R. et al. (2009) Neutralization of interleukin-10 from CD14(+) monocytes enhances gamma interferon production in peripheral blood mononuclear cells from Mycobacterium avium subsp. paratuberculosis-infected goats.
Clin Vaccine Immunol. 16 (7): 1003-11. -
Elhmouzi-Younes, J. et al. (2009) Bovine neonate natural killer cells are fully functional and highly responsive to interleukin-15 and to NKp46 receptor stimulation.
Vet Res. 40: 54. -
Gillan, S. et al. (2010) Identification of immune parameters to differentiate disease states among sheep infected with Mycobacterium avium subsp. paratuberculosis.
Clin Vaccine Immunol. 17: 108-17. -
Wagner, B. et al. (2010) Interferon-γ, interleukin-4 and interleukin-10 production by T helper cells reveals intact Th1 and regulatory TR1 cell activation and a delay of the Th2 cell response in equine neonates and foals.
Vet Res. 41: 47. -
Jaber, J.R. et al. (2010) Cross-reactivity of anti-human, anti-porcine and anti-bovine cytokine antibodies with cetacean tissues.
J Comp Pathol. 143: 45-51. -
Totté, P. et al. (2010) CD62L defines a subset of pathogen-specific bovine CD4 with central memory cell characteristics.
Dev Comp Immunol. 34 (2): 177-82. -
Contreras, V. et al. (2010) Existence of CD8α-like dendritic cells with a conserved functional specialization and a common molecular signature in distant mammalian species.
J Immunol. 185: 3313-25. -
de Mestre, A. et al. (2010) Split immunological tolerance to trophoblast.
Int J Dev Biol. 54: 445-55. -
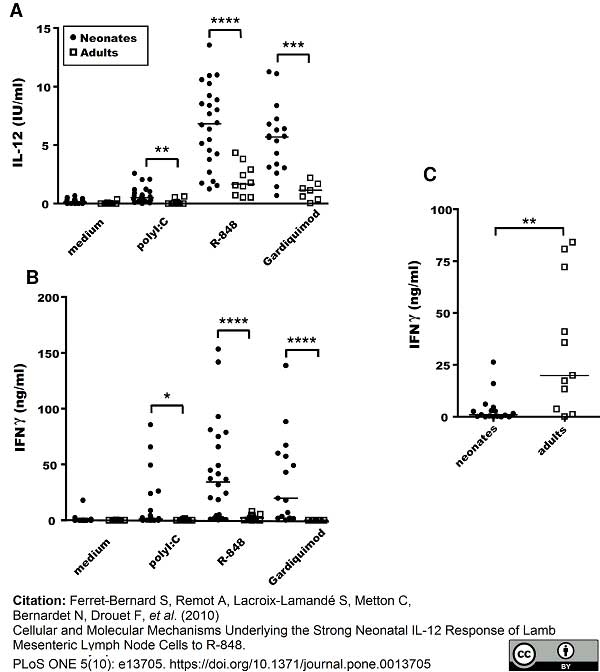
Ferret-Bernard, S. et al. (2010) Cellular and molecular mechanisms underlying the strong neonatal IL-12 response of lamb mesenteric lymph node cells to R-848.
PLoS One. 5: e13705. -
Dewals, B.G., et al.l (2011) Malignant catarrhal fever induced by Alcelaphine herpesvirus 1 is characterized by an expansion of activated CD3+CD8+CD4- T cells expressing a cytotoxic phenotype in both lymphoid and non-lymphoid tissues
Vet Res. 42(1): 95. -
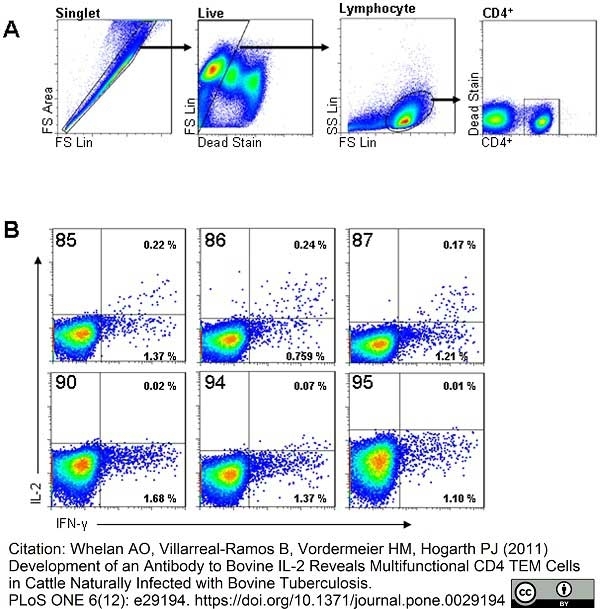
Whelan, A.O. et al. (2011) Development of an Antibody to Bovine IL-2 Reveals Multifunctional CD4 T(EM) Cells in Cattle Naturally Infected with Bovine Tuberculosis.
PLoS One. 6: e29194. -
Ferret-Bernard, S. et al. (2011) Mesenteric lymph node cells from neonates present a prominent IL-12 response to CpG oligodeoxynucleotide via an IL-15 feedback loop of amplification.
Vet Res. 42:19. -
Fellman, C.L. et al. (2011) Cyclosporine A affects the in vitro expression of T cell activation-related molecules and cytokines in dogs.
Vet Immunol Immunopathol. 140: 175-80. -
Sow, F.B. et al. (2011) Respiratory syncytial virus is associated with an inflammatory response in lungs and architectural remodeling of lung-draining lymph nodes of newborn lambs.
Am J Physiol Lung Cell Mol Physiol. 300 (1): L12-24. -
Skyberg, J.A. et al. (2011) Murine and bovine γδ T cells enhance innate immunity against Brucella abortus infections.
PLoS One. 6:e21978. -
Pillet, S. et al. (2011) Cellular immune response in the presence of protective antibody levels correlates with protection against 1918 influenza in ferrets.
Vaccine. 29 (39): 6793-801. -
Summers, C. et al. (2012) The distribution of immune cells in the lungs of classical and atypical ovine pulmonary adenocarcinoma.
Vet Immunol Immunopathol. 146: 1-7. -
Maślanka T et al. (2012) The presence of CD25 on bovine WC1+ γδ T cells is positively correlated with their production of IL-10 and TGF-β, but not IFN-γ.
Pol J Vet Sci. 15 (1): 11-20. -
Sun, L. et al. (2012) The role of proliferation in the regulation of interferon gamma (IFNγ) expression in foals.
Dev Comp Immunol. 36 (3): 534-9. -
Hansen, S. et al. (2013) Age-related changes in intracellular expression of IFN-γ and TNF-α in equine lymphocytes measured in bronchoalveolar lavage and peripheral blood.
Dev Comp Immunol. 39 (3): 228-33. -
Duncombe, L. et al. (2013) Investigating the Use of Protein Saver Cards for Storage and Subsequent Detection of Bovine Anti-Brucella abortus Smooth Lipopolysaccharide Antibodies and Gamma Interferon.
Clin Vaccine Immunol. 20: 1669-74. -
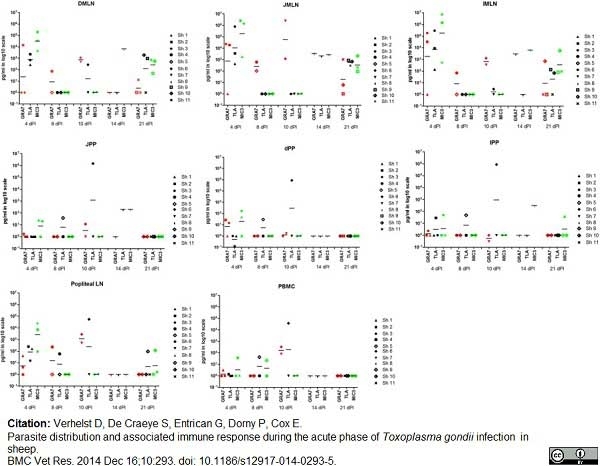
Verhelst, D. et al. (2014) Parasite distribution and associated immune response during the acute phase of Toxoplasma gondii infection in sheep.
BMC Vet Res. 2014 Dec 16;10(1):293. -
Hedges, J.F. et al. (2015) Amphotericin B stimulates γδ T and NK cells, and enhances protection from Salmonella infection.
Innate Immun. 21 (6): 598-608. -
Boshra H et al. (2015) A lumpy skin disease virus deficient of an IL-10 gene homologue provides protective immunity against virulent capripoxvirus challenge in sheep and goats.
Antiviral Res. 123: 39-49. -
Moreira, M.L. et al. (2015) Cross-reactivity of commercially available anti-human monoclonal antibodies with canine cytokines: establishment of a reliable panel to detect the functional profile of peripheral blood lymphocytes by intracytoplasmic staining.
Acta Vet Scand. 57: 51. -
Taylor, G. et al. (2015) Efficacy of a virus-vectored vaccine against human and bovine respiratory syncytial virus infections.
Sci Transl Med. 7 (300): 300ra127. -
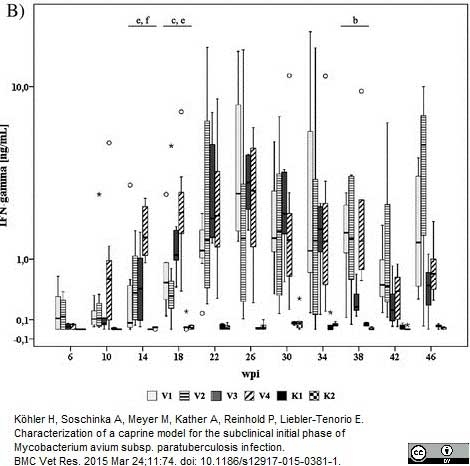
Köhler. H, et al. (2015) Characterization of a caprine model for the subclinical initial phase of Mycobacterium avium subsp. paratuberculosis infection
BMC Veterinary Research. 11 (1): 74. -
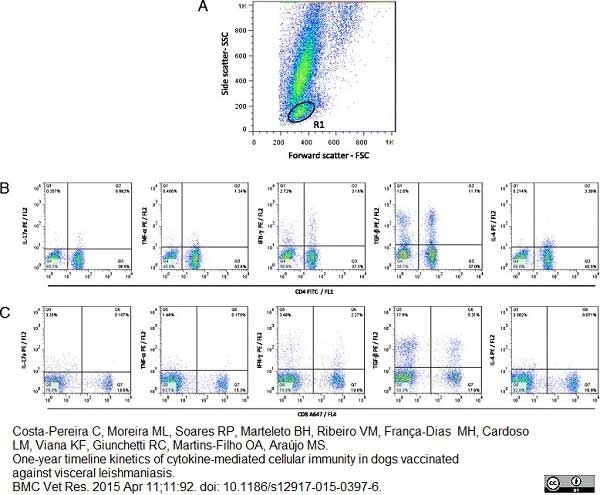
Costa-Pereira, C. et al. (2015) One-year timeline kinetics of cytokine-mediated cellular immunity in dogs vaccinated against visceral leishmaniasis.
BMC Vet Res. 11 (1): 92. -
El-Naggar, M.M. et al. (2015) Development of an improved ESAT-6 and CFP-10 peptide-based cytokine flow cytometric assay for bovine tuberculosis.
Comp Immunol Microbiol Infect Dis. 42: 1-7. -
Maggioli, M.F. et al. (2016) Increased TNF-α/IFN-γ/IL-2 and Decreased TNF-α/IFN-γ Production by Central Memory T Cells Are Associated with Protective Responses against Bovine Tuberculosis Following BCG Vaccination.
Front Immunol. 7: 421. -
Rodríguez-Gómez IM et al. (2016) Expression of T-bet, Eomesodermin and GATA-3 in porcine αβ T cells.
Dev Comp Immunol. 60: 115-26. -
Moreira, M.L. et al. (2016) Vaccination against canine leishmaniosis increases the phagocytic activity, nitric oxide production and expression of cell activation/migration molecules in neutrophils and monocytes.
Vet Parasitol. 220: 33-45. -
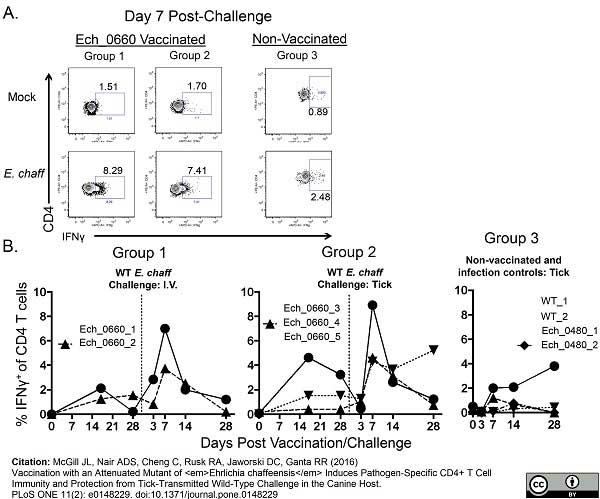
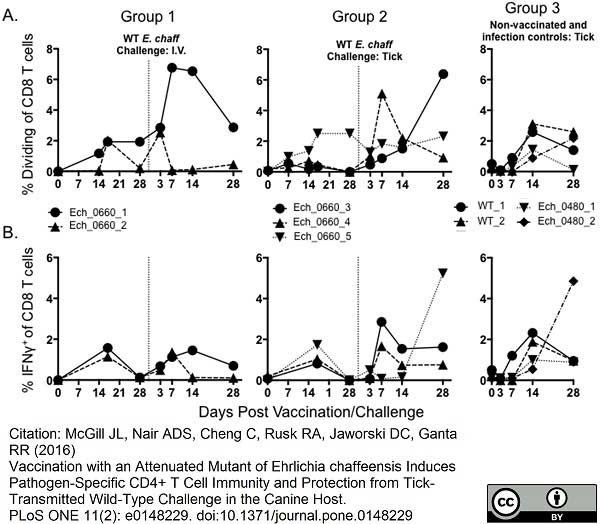
McGill, J.L. et al. (2016) Vaccination with an Attenuated Mutant of Ehrlichia chaffeensis Induces Pathogen-Specific CD4+ T Cell Immunity and Protection from Tick-Transmitted Wild-Type Challenge in the Canine Host.
PLoS One. 11 (2): e0148229. -
Vida, B. et al. (2016) Immunologic progression of canine leishmaniosis following vertical transmission in United States dogs.
Vet Immunol Immunopathol. 169: 34-8. -
Risalde, M.Á. et al. (2017) Development and evaluation of an interferon gamma assay for the diagnosis of tuberculosis in red deer experimentally infected with Mycobacterium bovis..
BMC Vet Res. 13 (1): 341. -
Cassady-Cain, R.L. et al. (2017) Inhibition of Antigen-Specific and Nonspecific Stimulation of Bovine T and B Cells by Lymphostatin from Attaching and Effacing Escherichia coli.
Infect Immun. 85 (2): e00845-16. -
Wattegedera, S.R. et al. (2017) Enhancing the toolbox to study IL-17A in cattle and sheep.
Vet Res. 48 (1): 20. -
Rodrigues, V. et al. (2017) Development of a bead-based multiplexed assay for simultaneous quantification of five bovine cytokines by flow cytometry.
Cytometry A. 91 (9): 901-7. -
Freer, H. et al. (2017) A monoclonal antibody for detection of intracellular and secreted interleukin-2 in horses.
Vet Immunol Immunopathol. 191: 30-5. -
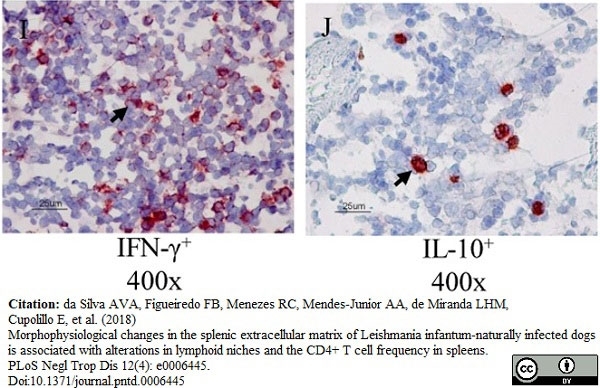
DaSilva, A.V.A. et al. (2018) Morphophysiological changes in the splenic extracellular matrix of Leishmania infantum-naturally infected dogs is associated with alterations in lymphoid niches and the CD4+ T cell frequency in spleens.
PLoS Negl Trop Dis. 12 (4): e0006445. -
Higgins, J.L. et al. (2018) Cell mediated immune response in goats after experimental challenge with the virulent Brucella melitensis strain 16M and the reduced virulence strain Rev. 1.
Vet Immunol Immunopathol. 202: 74-84. -
Roos, E.O. et al. (2018) IP-10: A potential biomarker for detection of Mycobacterium bovis infection in warthogs (Phacochoerus africanus).
Vet Immunol Immunopathol. 201: 43-8. -
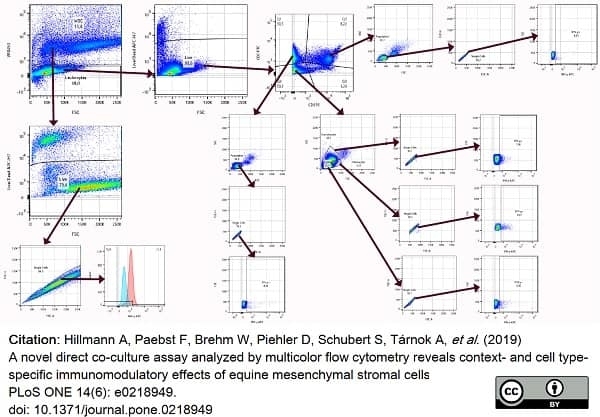
Hillmann, A. et al. (2019) A novel direct co-culture assay analyzed by multicolor flow cytometry reveals context- and cell type-specific immunomodulatory effects of equine mesenchymal stromal cells.
PLoS One. 14 (6): e0218949. -
Witonsky, S. et al. (2019) Can levamisole upregulate the equine cell-mediated macrophage (M1) dendritic cell (DC1) T-helper 1 (CD4 Th1) T-cytotoxic (CD8) immune response in vitro.?
J Vet Intern Med. 33 (2): 889-96. -
Fedorka, C.E. et al. (2019) Alteration of the mare's immune system by the synthetic progestin, altrenogest.
Am J Reprod Immunol. 82 (2): e13145. -
Aguiar-Soares, R.D.O. et al. (2020) Phase I and II Clinical Trial Comparing the LBSap, Leishmune(®), and Leish-Tec(®) Vaccines against Canine Visceral Leishmaniasis.
Vaccines (Basel). 8 (4): 690. -
Arrieta-Villegas, C. et al. (2020) Immunogenicity and Protection against Mycobacterium caprae Challenge in Goats Vaccinated with BCG and Revaccinated after One Year.
Vaccines (Basel). 8 (4): 751. -
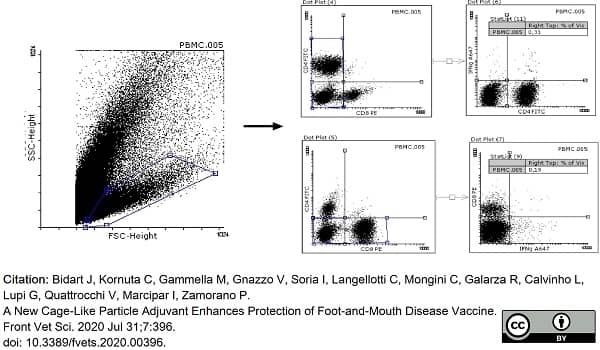
Bidart, J. et al. (2020) A New Cage-Like Particle Adjuvant Enhances Protection of Foot-and-Mouth Disease Vaccine.
Front Vet Sci. 7: 396. -
Lacasta, A. et al. (2021) Synergistic Effect of Two Nanotechnologies Enhances the Protective Capacity of the Theileria parva Sporozoite p67C Antigen in Cattle.
J Immunol. 206 (4): 686-99. -
Damani-Yokota, P. et al. (2021) Transcriptional programming and gene regulation in WC1+ γδ T cell subpopulations.
Mol Immunol. 142: 50-62. -
Villa-Mancera, A. et al. (2021) Phage display-based vaccine with cathepsin L and excretory-secretory products mimotopes of Fasciola hepatica. induces protective cellular and humoral immune responses in sheep.
Vet Parasitol. 289: 109340. -
Santillo, A. et al. (2022) Feeding tannins to dairy cows in different seasons improves the oxidative status of blood plasma and the antioxidant capacity of cheese.
J Dairy Sci. 105 (11): 8609-20. -
Ciliberti, M.G. et al. (2022) Green extraction of bioactive compounds from wine lees and their bio-responses on immune modulation using in vitro sheep model.
J Dairy Sci. 105 (5): 4335-53. -
Matralis, D.T. et al. (2023) Intracellular IFN-γ and IL-4 levels of CD4 + and CD8 + T cells in the peripheral blood of naturally infected (Leishmania infantum) symptomatic dogs before and following a 4-week treatment with miltefosine and allopurinol: a double-blinded, controlled and cross-sectional study.
Acta Vet Scand. 65 (1): 2. -
Bouroutzika, E. et al. (2023) Melatonin Administration to Pregnant Ewes for Coccidiosis Control in Their Offspring.
Animals (Basel). 13 (14): 2381. -
Ferreras-Colino, E. et al. (2023) Oral immunization with heat-inactivated Mycobacterium bovis reduces local parasite dissemination and hepatic granuloma development in mice infected with Leishmania amazonensis.
Res Vet Sci. 162: 104963. -
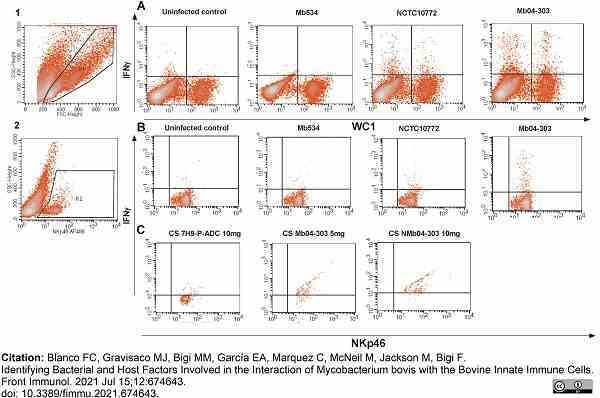
Blanco, F.C. et al. (2021) Identifying Bacterial and Host Factors Involved in the Interaction of Mycobacterium bovis with the Bovine Innate Immune Cells.
Front Immunol. 12: 674643. -
Archer, T.M. et al. (2018) In vivo effects of aspirin and cyclosporine on regulatory T cells and T-cell cytokine production in healthy dogs.
Vet Immunol Immunopathol. 197: 63-8. -
de Silva, K. et al. (2018) Defining resilience to mycobacterial disease: Characteristics of survivors of ovine paratuberculosis.
Vet Immunol Immunopathol. 195: 56-64. -
Faber, E. et al. (2024) Identification of T cell and linear B cell epitopes on African horse sickness virus serotype 4 proteins VP1-1, VP2, VP4, VP7 and NS3.
Vaccine. 42 (2): 136-45. -
Lebedev, M. et al. (2021) Myeloid-like γδ T cell subset in the immune response to an experimental Rift Valley fever vaccine in sheep.
Vet Immunol Immunopathol. 233: 110184. -
Svitek, N. et al. (2022) Systematic Determination of TCR-Antigen and Peptide-MHC Binding Kinetics among Field Variants of a Theileria parva Polymorphic CTL Epitope.
J Immunol. 208 (3): 549-561. -
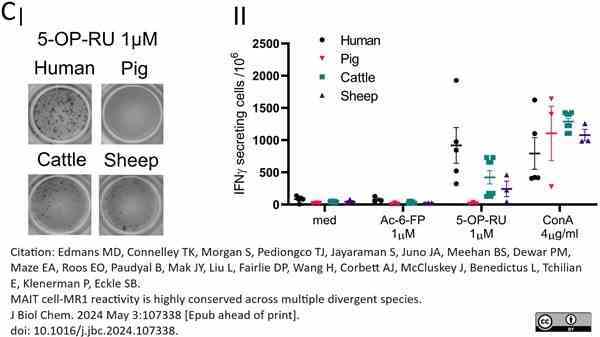
Edmans, M.D. et al. (2024) MAIT cell-MR1 reactivity is highly conserved across multiple divergent species.
J Biol Chem. 107338 [Epub ahead of print].
Further Reading
-
Rhodes, S. et al. (2000) Distinct response kinetics of gamma interferon and interleukin-4 in bovine tuberculosis.
Infect Immun. 68:5393-400.
- Synonyms
- Interferon Gamma
- RRID
- AB_324374
- UniProt
- P07353
- Entrez Gene
- IFNG
- GO Terms
- GO:0009615 response to virus
- GO:0005125 cytokine activity
- GO:0005133 interferon-gamma receptor binding
- GO:0005615 extracellular space
- GO:0006955 immune response
MCA1783B
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Bovine ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up