CD335 antibody | AKS1








Mouse anti Bovine CD335:RPE
- Product Type
- Monoclonal Antibody
- Clone
- AKS1
- Isotype
- IgG1
- Specificity
- CD335
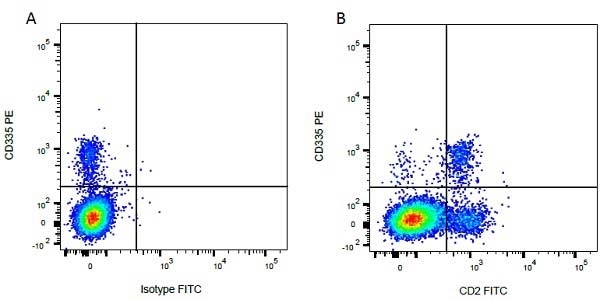
| Mouse anti Bovine CD335 antibody, clone AKS1 recognizes bovine CD335, also known as NKp46 and Natural cytotoxicity triggering receptor 1. CD335 is a type I transmembrane protein, with two extracellular C2-type immunoglobulin-like domains, which functions as an activating receptor. CD335 is expressed by human natural killer cells (Sivori et al. 1997). The bovine homologue is expressed on bovine NK cells (Storset et al. 2004) and no expression of CD335 has been detected on B cells, T cells, monocytes or granulocytes. Clone AKS1 is reported to activate lysis of FcgammaR-expressing cell line P815, by IL-2 activated NKp46+ cells (Storset et al. 2004). |
- Target Species
- Bovine
- Species Cross-Reactivity
-
Target Species Cross Reactivity American Bison - N.B. Antibody reactivity and working conditions may vary between species.
- Product Form
- Purified IgG conjugated to R. Phycoerythrin (RPE) - lyophilized
- Reconstitution
- Reconstitute with 1 ml distilled water
- Preparation
- Purified IgG prepared by affinity chromatography on Protein G from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- 0.09% sodium azide (NaN3)
1% bovine serum albumin
5% sucrose - Immunogen
- Fusion protein consisting of the extracellular region of bovine CD335.
- Fusion Partners
- Spleen cells from immunised Balb/c mice were fused with cells of the NS-0 myeloma cell line.
- Max Ex/Em
-
Fluorophore Excitation Max (nm) Emission Max (nm) RPE 488nm laser 496 578 - Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Following reconstitution store at +4°C.
DO NOT FREEZE.
This product should be stored undiluted. This product is photosensitive and should be protected from light. Should this product contain a precipitate we recommend microcentrifugation before use.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Flow Cytometry | Neat |
- Flow Cytometry
- Use 10μl of the suggested working dilution to label 106 cells in 100μl
How to Use the Spectraviewer
Watch the Tool Tutorial Video ▸- Start by selecting the application you are interested in, with the option to select an instrument from the drop down menu or create a customized instrument
- Select the fluorophores or fluorescent proteins you want to include in your panel to check compatibility
- Select the lasers and filters you wish to include
- Select combined or multi-laser view to visualize the spectra
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Mouse IgG1 Negative Control:RPE | MCA928PE | F | 100 Tests | Log in | |||
| List Price | Your Price | ||||||
| Log in | |||||||
| Description | Mouse IgG1 Negative Control:RPE | ||||||
References for CD335 antibody
-
Storset, A.K. et al. (2004) NKp46 defines a subset of bovine leukocytes with natural killer cell characteristics.
Eur J Immunol. 34 (3): 669-76. -
Kulberg, S. et al. (2004) Reference values for relative numbers of natural killer cells in cattle blood.
Dev Comp Immunol. 28 (9): 941-8. -
Van Rhijn, I. et al. (2007) Massive, sustained γ/δ T cell migration from the bovine skin in vivo.
J Leukoc Biol. 81: 968-73. -
Toka, F.N. et al. (2011) Rapid and Transient Activation of γ/δ T Cells to IFN-γ Production, NK Cell-Like Killing, and Antigen Processing during Acute Virus Infection.
J Immunol. 186: 4853-61. -
Bastos, R.G. et al. (2008) Bovine NK cells acquire cytotoxic activity and produce IFN-γ after stimulation by Mycobacterium bovis BCG- or Babesia bovis-exposed splenic dendritic cells.
Vet Immunol Immunopathol. 124: 302-12. -
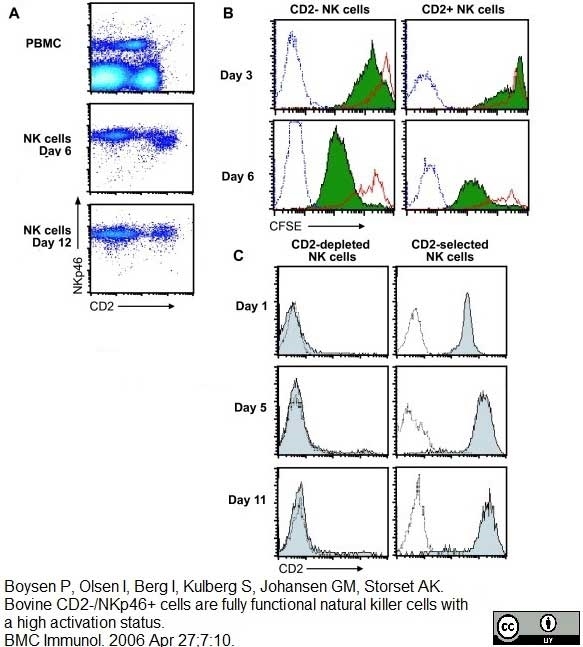
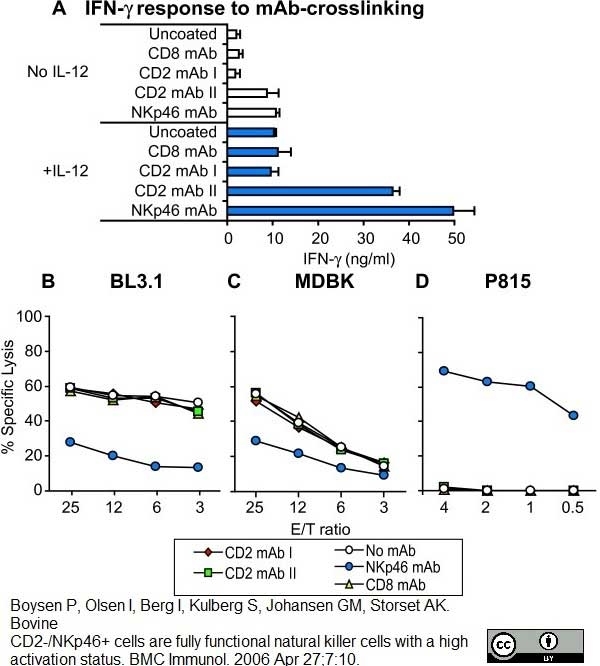
Boysen P et al. (2006) Bovine CD2-/NKp46+ cells are fully functional natural killer cells with a high activation status.
BMC Immunol. 7: 10. -
Elhmouzi-Younes, J. et al. (2009) Bovine neonate natural killer cells are fully functional and highly responsive to interleukin-15 and to NKp46 receptor stimulation.
Vet Res. 40: 54. -
Elhmouzi-Younes, J. et al. (2010) Ovine CD16+/CD14- blood lymphocytes present all the major characteristics of natural killer cells.
Vet Res. 41:4.
View The Latest Product References
-
Hoek, A. et al. (2009) Subpopulations of bovine WC1(+) γ/δ T cells rather than CD4(+)CD25(high) Foxp3(+) T cells act as immune regulatory cells ex vivo.
Vet Res. 40:6. -
Zuerner, R.L. et al. (2011) A Leptospira borgpetersenii Serovar Hardjo Vaccine Induces a Th1 Response, Activates NK Cells, and Reduces Renal Colonization.
Clin Vaccine Immunol. 18: 684-91. -
Nelson, D.D. et al. (2010) CD8(+)/perforin(+)/WC1(- ) γ/δT cells, not CD8(+) α/β T cells, infiltrate vasculitis lesions of American bison (Bison bison) with experimental sheep-associated malignant catarrhal fever.
Vet Immunol Immunopathol. 136: 284-91. -
Banos, G. et al. (2013) Identification of immune traits correlated with dairy cow health, reproduction and productivity.
PLoS One. 8: e65766. -
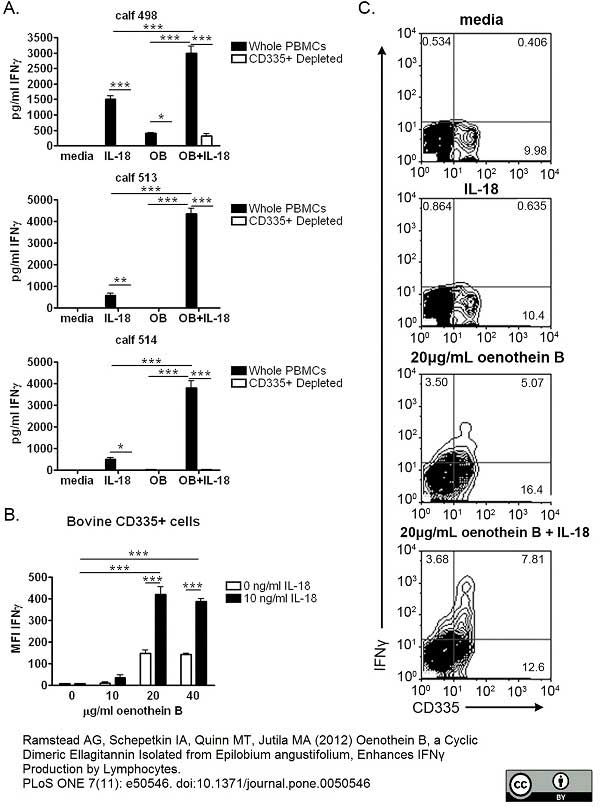
Ramstead, A.G. et al. (2015) Aging influences the response of T cells to stimulation by the ellagitannin, oenothein B.
Int Immunopharmacol. 26 (2): 367-77. -
Boysen, P. et al. (2008) Natural killer cells in lymph nodes of healthy calves express CD16 and show both cytotoxic and cytokine-producing properties.
Dev Comp Immunol. 32 (7): 773-83. -
González-Cano, P. et al. (2014) Two functionally distinct myeloid dendritic cell subpopulations are present in bovine blood.
Dev Comp Immunol. 44 (2): 378-88. -
Di Palma, S. et al. (2012) Comparative spatiotemporal analysis of the intrathecal immune response in natural listeric rhombencephalitis of cattle and small ruminants.
Comp Immunol Microbiol Infect Dis. 35 (5): 429-41. -
Pirson, C. et al. (2015) Highly purified mycobacterial phosphatidylinositol mannosides drive cell-mediated responses and activate NKT cells in cattle.
Clin Vaccine Immunol. 22 (2): 178-84. -
Schneider, D.A. et al. (2011) Dynamics of bovine spleen cell populations during the acute response to Babesia bovis infection: an immunohistological study.
Parasite Immunol. 33 (1): 34-44. -
Van Meulder F et al. (2015) Analysis of the protective immune response following intramuscular vaccination of calves against the intestinal parasite Cooperia oncophora.
Int J Parasitol. 45 (9-10): 637-46. -
Patch, J.R. et al. (2014) Infection with foot-and-mouth disease virus (FMDV) induces a natural killer (NK) cell response in cattle that is lacking following vaccination.
Comp Immunol Microbiol Infect Dis. 37 (4): 249-57. -
Hedges, J.F. et al. (2015) Amphotericin B stimulates γδ T and NK cells, and enhances protection from Salmonella infection.
Innate Immun. 21 (6): 598-608. -
Allan, A.J. et al. (2015) Cattle NK Cell Heterogeneity and the Influence of MHC Class I.
J Immunol. 195 (5): 2199-206. -
Maślanka, T. et al. (2016) Prostaglandin E2 exerts the proapoptotic and antiproliferative effects on bovine NK cells
Res Vet Sci. 107: 80-7. -
Hecker YP et al. (2015) Cell mediated immune responses in the placenta following challenge of vaccinated pregnant heifers with Neospora caninum.
Vet Parasitol. 214 (3-4): 247-54. -
Ohira K et al. (2016) Bovine leukemia virus reduces anti-viral cytokine activities and NK cytotoxicity by inducing TGF-β secretion from regulatory T cells.
Immun Inflamm Dis. 4 (1): 52-63. -
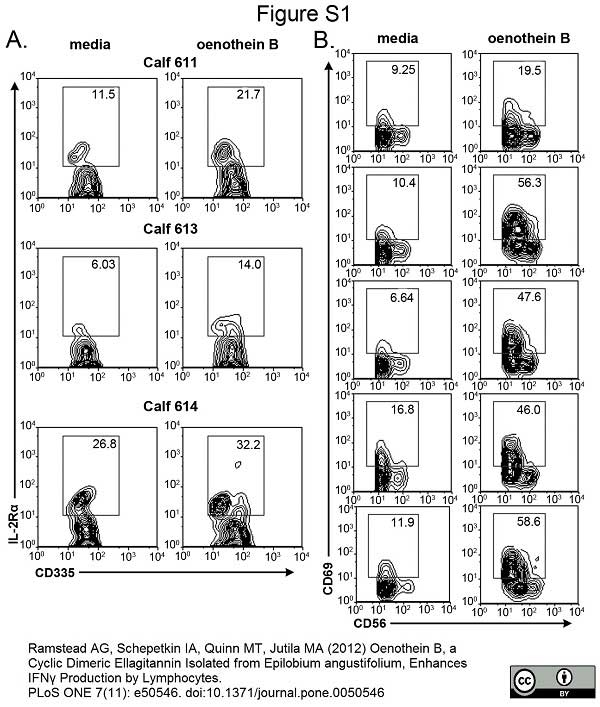
Johnson, W.C. et al. (2008) Bovine WC1(-) γδT cells incubated with IL-15 express the natural cytotoxicity receptor CD335 (NKp46) and produce IFN-γ in response to exogenous IL-12 and IL-18.
Dev Comp Immunol. 32 (8): 1002-10. -
Sipka, A. et al. (2016) Bovine natural killer cells are present in Escherichia coli infected mammary gland tissue and show antimicrobial activity in vitro.
Comp Immunol, Microbiol Infect Dis. 48: 54-60. -
Hamilton, C.A. et al. (2016) Interactions between natural killer cells and dendritic cells favour T helper1-type responses to BCG in calves.
Vet Res. 47 (1): 85. -
Krueger, L.A. et al. (2016) Gamma delta T cells are early responders to Mycobacterium avium ssp. paratuberculosis in colostrum-replete Holstein calves.
J Dairy Sci. 99 (11): 9040-50. -
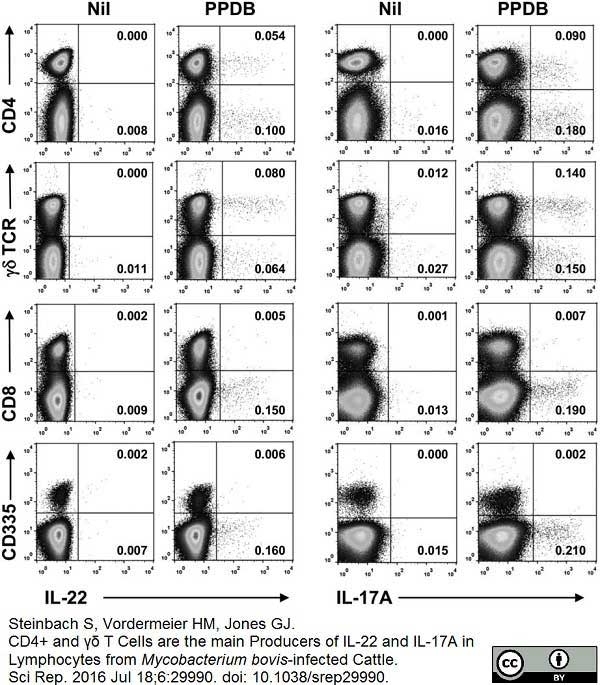
Steinbach, S. et al. (2016) CD4+ and γδ T Cells are the main Producers of IL-22 and IL-17A in Lymphocytes from Mycobacterium bovis-infected Cattle.
Sci Rep. 6: 29990. -
Rutigliano, H.M. et al. (2016) Trophoblast Major Histocompatibility Complex Class I Expression Is Associated with Immune-Mediated Rejection of Bovine Fetuses Produced by Cloning.
Biol Reprod. 95 (2): 39. -
González-Hernández A et al. (2016) Host protective ASP-based vaccine against the parasitic nematode Ostertagia ostertagi triggers NK cell activation and mixed IgG1-IgG2 response.
Sci Rep. 6: 29496. -
Khosa, S. et al. (2020) Bovine Adenovirus-3 Tropism for Bovine Leukocyte Sub-Populations.
Viruses. 12 (12): 1431. -
Denholm, S.J. et al. (2017) Estimating genetic and phenotypic parameters of cellular immune-associated traits in dairy cows.
J Dairy Sci. 100 (4): 2850-2862. -
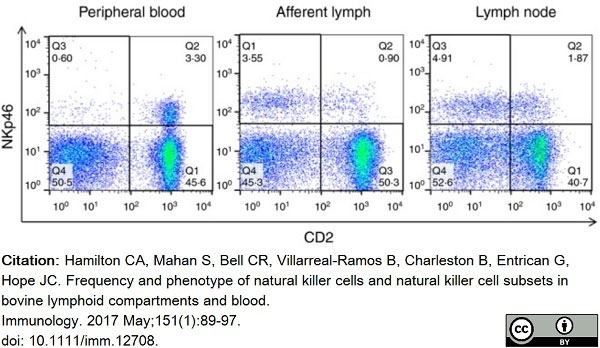
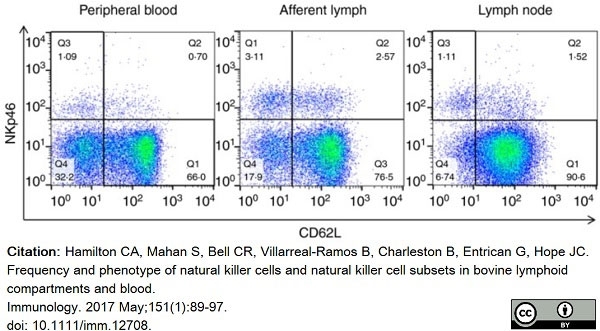
Hamilton, C.A. et al. (2017) Frequency and phenotype of natural killer cells and natural killer cell subsets in bovine lymphoid compartments and blood.
Immunology. 151 (1): 89-97. -
Bassi, P.B. et al. (2018) Parasitological and immunological evaluation of cattle experimentally infected with Trypanosoma vivax.
Exp Parasitol. 185: 98-106. -
de Araújo, F.F. et al. (2019) Distinct immune response profile during Rhipicephalus. (Boophilus.) microplus. infestations of guzerat dairy herd according to the maternal lineage ancestry (mitochondrial DNA).
Vet Parasitol. 273: 36-44. -
Nakajima, N. et al. (2019) Effects of direct exposure to cold weather under grazing in winter on the physiological, immunological, and behavioral conditions of Japanese Black beef cattle in central Japan.
Anim Sci J. 90 (8): 1033-1041. -
Okino, C.H. et al. (2020) A polymorphic CD4 epitope related to increased susceptibility to Babesia bovis. in Canchim calves.
Vet Immunol Immunopathol. 230: 110132. -
de Araújo, F.F. et al. (2019) Distinct immune response profile during Rhipicephalus (Boophilus) microplus. infestations of guzerat dairy herd according to the maternal lineage ancestry (mitochondrial DNA).
Vet Parasitol. 273: 36-44. -
Colombatti Olivieri, M.A. et al. (2021) Evaluation of a virulent strain of Mycobacterium avium subsp. paratuberculosis used as a heat-killed vaccine.
Vaccine. 39 (51): 7401-7412. -
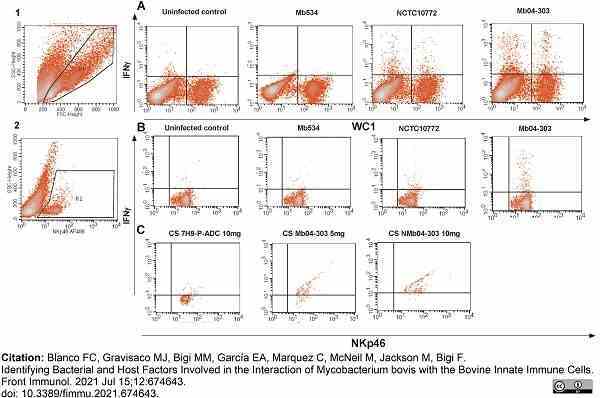
Blanco, F.C. et al. (2021) Identifying Bacterial and Host Factors Involved in the Interaction of Mycobacterium bovis with the Bovine Innate Immune Cells.
Front Immunol. 12: 674643.
Further Reading
-
Sivori, S. et al. (1997) p46, a novel natural killer cell-specific surface molecule that mediates cell activation.
J Exp Med. 186 (7): 1129-36. -
Storset, A.K. et al. (2003) Natural killer cell receptors in cattle: a bovine killer cell immunoglobulin-like receptor multigene family contains members with divergent signaling motifs.
Eur J Immunol. 33 (4): 980-90.
- Synonyms
- NKp46
- RRID
- AB_2149299
- UniProt
- Q863H2
- Entrez Gene
- NCR1
- GO Terms
- GO:0005886 plasma membrane
- GO:0016021 integral to membrane
- GO:0004872 receptor activity
MCA2365PE
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Bovine ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up