Understanding Resolution and Signal Separation in Flow Cytometry
Author: Mike Blundell | Reviewer: Chloe Fenton
What Is Resolution in Flow Cytometry?
Resolution in flow cytometry refers to the ability to distinguish a positive cell population from a negative population, determined by instrument configuration, fluorophore selection, and background noise such as spillover or nonspecific staining.
Understanding Resolution in Multicolor Flow Cytometry
Multicolor flow cytometry is the terminology used when analyzing multiple fluorescent parameters in one sample, may it be surface markers, intracellular markers, DNA, or all combined. In addition to ensuring the right controls (discussed in Chapter 4), optimizing your experimental procedure (discussed in Chapter 5), and careful sample preparation, there are a few other considerations that should be taken into account to ensure meaningful results can be obtained.
Definition of Resolution
Resolution can be described as the potential of an instrument, together with a combination of fluorophores, to separate a positive population from a negative population.
How Fluorophore Interactions Affect Resolution
Each additional fluorophore you add to your flow cytometry panel has the potential to influence another fluorophore. The result of this can be unwanted fluorescence in additional channels, which has to be compensated for (discussed in Chapter 2), and a loss in resolution.
Factors That Reduce Resolution
High levels of noise caused by nonspecific staining, high background staining, cell autofluorescence, and spillover can all contribute to a reduction in sensitivity and resolution.
Optimizing Resolution in Panel Design
To ensure optimal resolution, there are a few simple rules that can be followed that will help form the basics of all panel design. While following these best practice rules may not result in a perfect panel first time every time, it will ensure you have a solid starting point that should result in fewer errors due to incompatible reagents.
Example of Cell Population Resolution
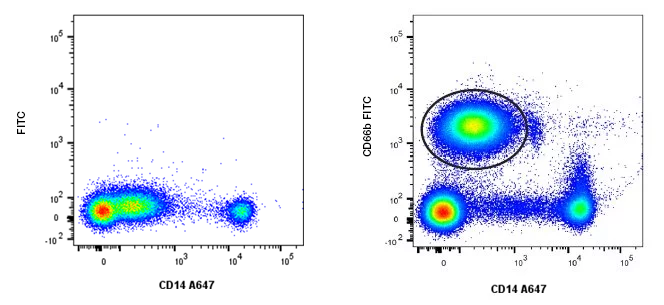
Fig. 29. Cell population resolution. Using an additional marker CD66b FITC (MCA216F), the granulocytes (circled in black) that express low levels of CD14 A647 (MCA1568A647) can be resolved from lymphocytes, which are negative for CD14 and CD66b.
Instrument Configuration and Its Impact
Instrument configuration is important to understand before you start to build your panel. You simply cannot use a fluorophore your instrument is not configured for, regardless of whether it is the theoretical best fit. Instrument configuration is the setup of the lasers, optics, and filters that are contained within the cytometer. This can vary significantly between cytometers.
The S3e Cell Sorter from Bio-Rad has 2 lasers and has 4 fluorescence detectors, whereas the ZE5 Cell Analyzer has 5 lasers and the potential to simultaneously detect 27 fluorophores.
Frequently Asked Questions
What reduces flow cytometry resolution?
Resolution is reduced by factors such as high background staining, autofluorescence, nonspecific binding, and fluorophore spillover between channels.
How can resolution be improved in flow cytometry?
Resolution can be improved by optimizing fluorophore selection, reducing spillover, minimizing background staining, and ensuring the instrument configuration matches the panel design.
Why is resolution important in multicolor flow cytometry?
Good resolution ensures that distinct cell populations can be clearly separated, which improves the accuracy and reliability of experimental data.
Flow Cytometry Basics Guide Download
Get your own copy to peruse at your leisure
Download our updated Flow Cytometry Basics Guide to have practical advice, best practice examples, and a basic overview of all the important flow cytometry principles in one handy location.
Resources
Bio-Rad is committed to helping you succeed in Flow Cytometry by sharing knowledge and best practices from our experts. Below are some extremely useful resources to enhance your journey of discovery and support your success.
