Flow Cytometry Optics and Detection
Author: Mike Blundell | Reviewer: Chloe Fenton
Light Interaction After Hydrodynamic Focusing
After hydrodynamic focusing, each particle passes through one or more focused laser beams. Light scattering or fluorescence emission from the particle labeled with a fluorophore provides information about the particle’s properties.
Lasers, which are the most commonly used light sources in flow cytometry, produce a single wavelength of light (a laser beam) at a specific frequency. They are available at different wavelengths ranging from deep ultraviolet (UV) to infrared (IR), and have a variable range of power levels (photon output/time typically specified in mW).
Forward Scatter (FSC)
Light that is scattered in the forward direction, after interacting with a particle and typically up to 20º offset from the laser beam’s axis, is collected by a photomultiplier tube (PMT) or photodiode, and is known as the forward scatter (FSC) channel.
This angle can, however, vary depending on your instrument, leading to variation of FSC signals between different machines. This FSC measurement can give an estimation of particle size because larger particles refract more light than smaller particles. However, this depends on several factors such as the sample, laser wavelength, collection angle, refractive index of the sample, and sheath fluid.
The detection of small particles provides a good example. When the particles are smaller than the wavelength of the illumination source, such as a 200 nm exosome illuminated using a 488 nm laser, the light is not necessarily scattered in a forward direction. Therefore, forward scatter may not be a good estimate of size.
Side Scatter (SSC)
Light measured at a 90º angle to the excitation line is called side scatter (SSC). The SSC can provide information about the relative complexity (for example, granularity and internal structures) of a cell or particle; however, as with forward scatter, this can depend on various factors.
Both FSC and SSC are unique for every particle and a combination of the two may be used to roughly differentiate cell types in a heterogeneous population such as lymphocytes, monocytes, and granulocytes in peripheral blood.
Limitations of Scatter Measurements Alone
However, FSC and SSC characteristics vary based on the sample type and quality of sample preparation, so fluorescent labeling is generally required to obtain more detailed information.
Fluorescence Detection
Fluorescence measurements taken at different wavelengths can provide quantitative and qualitative data about fluorophore-labeled cell surface receptors or intracellular molecules, such as DNA and cytokines.
Most flow cytometers use separate channels and detectors to detect emitted light, the number of which varies according to the instrument and manufacturer.
Detectors Used in Flow Cytometry
Detectors are either PMTs or photodiodes, specifically avalanche photodiodes (APD). PMTs are the most commonly used detectors, but APDs are becoming more popular due to the cost and having improved sensitivity when detecting longer wavelengths.
Optical Filters in Conventional Flow Cytometry
In a conventional flow cytometer, such as the Bio-Rad ZE5 Cell Analyzer, detection specificity is controlled by optical filters that block certain wavelengths while transmitting (passing) others. There are three major filter types. Long pass filters allow light through above a cutoff wavelength, short pass filters permit light below a certain wavelength, and band pass filters transmit light within a specified narrow range of wavelengths (termed a bandwidth).
These dichroic filters can block light by phased reflection, allowing certain light to pass through and interfering with other wavelengths (Figure 2).
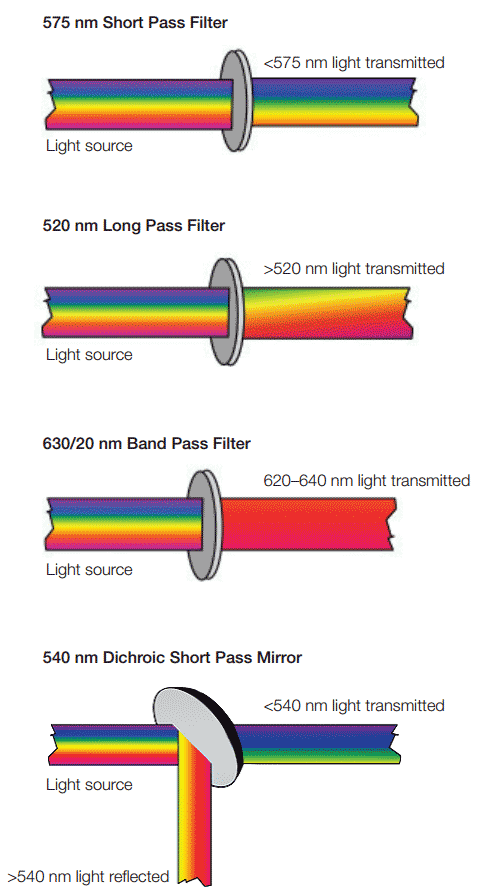
Fig. 2. Different types of optical filters
Dichroic Filters and Light Path Control
A dichroic filter is also a mirror when placed at an angle to the oncoming light. This type of filter can perform two functions. Firstly, it allows specific wavelengths to pass in the forward direction. Secondly, it can reflect light at a 90º angle. This allows the light path to be passed through a series of filters. The precise choice and order of the filters can be arranged so that multiple signals can be detected simultaneously (Figure 3).
ZE5 Cell Analyzer Configuration
Visit the Flow Cytometry Applications page to learn how the flexible configuration of the ZE5 Cell Analyzer can help you design complex experiments with high speed and sensitivity.

Fig. 3. Schematic overview of a typical flow cytometer setup. FL, fluorescence; PMT, photomultiplier tube; SSC, side scatter; FSC, forward scatter; blue arrow, light path.
Full Spectrum Flow Cytometry
Full spectrum cytometry (often called spectral cytometry) does not use filters to partition the emitted light from each laser. Instead, all the emitted light is captured across an array of detectors (16–32 per laser). As the name suggests, this allows the entire spectral profile of a fluorescent dye from multiple lasers to be captured. Multiple spectral profiles can then be unmixed from each other to identify the proportion of signal coming from each fluorescent dye.
Flow Cytometry Basics Guide Download
Get your own copy to peruse at your leisure
Download our updated Flow Cytometry Basics Guide to have practical advice, best practice examples, and a basic overview of all the important flow cytometry principles in one handy location.
Resources
Bio-Rad is committed to helping you succeed in Flow Cytometry by sharing knowledge and best practices from our experts. Below are some extremely useful resources to enhance your journey of discovery and support your success.
