Nerve Growth Factor 2.5S


Purified Mouse Nerve Growth Factor 2.5S
- Product Type
- Purified Protein
- Specificity
- Nerve Growth Factor 2.5S
| Purified Mouse Nerve Growth Factor 2.5S is prepared form mouse submaxillary glands (Bocchini and Angeletti 1969) and has an apparent molecular mass of ~30 kDa. Nerve growth factor has a variety of effects on the growth and development of sensory and sympathetic neurons. In the peripheral nervous system, NGF is required for the development and maintenance of sympathetic nerve cells that use catecholamine neurotransmitters. Purified Mouse Nerve Growth Factor 2.5S has been used to demonstrate the importance of NGF in regulation of neuronal function through the up-regulation of the transcription factor NFAT (Nuclear Factor of Activated T-cells) via activation of the PI3K/Akt pathway (Kim et al. 2014). |
- Target Species
- Mouse
- Species Cross-Reactivity
-
Target Species Cross Reactivity Rat - N.B. Antibody reactivity and working conditions may vary between species.
- Product Form
- Purified natural murine nerve growth factor - lyophilized
- Reconstitution
- Reconstitute with 1.0 ml distilled water. Care should be taken during reconstitution as the protein may appear as a film at the bottom of the vial. Bio-Rad recommend that the vial is gently mixed after reconstitution.
- Preparation
- Murine nerve growth factor is prepared from the submaxillary glands of mice by sephadex and subsequent cellulose chromatography (Bocchini and Angeletti 1969).
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- None present
- Purity
- >98% by SDS PAGE
- Approx. Protein Concentrations
- 1.0mg/ml
- Protein Molecular Weight
- Approximately 30 kDa
- Regulatory
- For research purposes only
- Guarantee
- 3 months from date of reconstitution
This product should be stored undiluted.
Storage in frost-free freezers is not recommended. Avoid repeated freezing and thawing as this may denature the protein. Should this product contain a precipitate we recommend microcentrifugation before use.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Functional Assays |
References for Nerve Growth Factor 2.5S
-
Rohn, T.A. et al. (2011) A Virus-Like Particle-Based Anti-Nerve Growth Factor Vaccine Reduces Inflammatory Hyperalgesia: Potential Long-Term Therapy for Chronic Pain.
J Immunol. 186: 1769-80. -
Laursen, L.S. et al. (2011) Translation of myelin basic protein mRNA in oligodendrocytes is regulated by integrin activation and hnRNP-K.
J Cell Biol. 192: 797-811. -
Colbert, R.A. et al. (1994) Vasoactive intestinal peptide stimulates neuropeptide Y gene expression and causes neurite extension in PC12 cells through independent mechanisms.
J Neurosci. 14: 7141-7. -
Smith-Thomas, L.C. et al. (1995) Increased axon regeneration in astrocytes grown in the presence of proteoglycan synthesis inhibitors.
J Cell Sci. 108: 1307-15. -
Barrie, A.P. et al. (1997) Pituitary adenylyl cyclase-activating peptide stimulates extracellular signal-regulated kinase 1 or 2 (ERK1/2) activity in a Ras-independent, mitogen-activated protein Kinase/ERK kinase 1 or 2-dependent manner in PC12 cells.
J Biol Chem. 272: 19666-71. -
Liu, N. et al. (2005) Enhancement of Schwann cell myelin formation by K252a in the Trembler-J mouse dorsal root ganglion explant culture.
J Neurosci Res. 79: 310-7. -
Eibl, J.K. et al. (2010) Multipotent neurotrophin antagonist targets brain-derived neurotrophic factor and nerve growth factor.
J Pharmacol Exp Ther. 332: 446-54. -
Vogelezang, M. et al. (2007) Neurite outgrowth on a fibronectin isoform expressed during peripheral nerve regeneration is mediated by the interaction of paxillin with alpha4beta1 integrins.
BMC Neurosci. 8: 44.
View The Latest Product References
-
Seiberlich, V. et al. (2015) Downregulation of the microtubule associated protein Tau impairs process outgrowth and myelin basic protein mRNA transport in oligodendrocytes.
Glia. 63 (9): 1621-35. -
von Büdingen, H.C. et al. (2015) The myelin oligodendrocyte glycoprotein directly binds nerve growth factor to modulate central axon circuitry.
J Cell Biol. 210 (6): 891-8. -
Miyamoto, Y. et al. (2015) Involvement of the Tyro3 receptor and its intracellular partner Fyn signaling in Schwann cell myelination.
Mol Biol Cell. 26 (19): 3489-503. -
Huang, J.K. et al. (2011) Retinoid X receptor gamma signaling accelerates CNS remyelination.
Nat Neurosci. 14 (1): 45-53. -
Tsai, M.S. et al. (2018) Nerve growth factor upregulates sirtuin 1 expression in cholestasis: a potential therapeutic target.
Exp Mol Med. 50 (1): e426. -
Lager, A.M. et al. (2018) Rapid functional genetics of the oligodendrocyte lineage using pluripotent stem cells.
Nat Commun. 9 (1): 3708. -
Kim, S.M. (2022) NGF activates NFAT via the MEK1/2 pathway in PC12 cells
All Life. 15 (1): 183-190. -
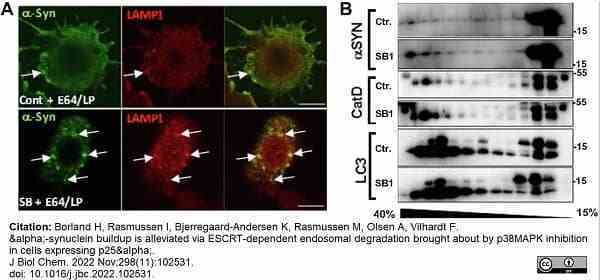
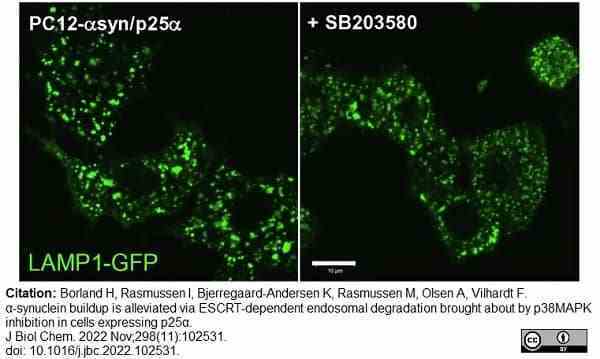
Borland, H. et al. (2022) α-synuclein buildup is alleviated via ESCRT-dependent endosomal degradation brought about by p38MAPK inhibition in cells expressing p25α.
J Biol Chem. 298 (11): 102531. -
Holloway, R.K. et al. (2023) Localized microglia dysregulation impairs central nervous system myelination in development.
Acta Neuropathol Commun. 11 (1): 49. -
Bekku, Y. et al. (2024) Glia trigger endocytic clearance of axonal proteins to promote rodent myelination.
Dev Cell. S1534-5807(24)00028-5. -
Mahú, I. et al. (2020) Brain-Sparing Sympathofacilitators Mitigate Obesity without Adverse Cardiovascular Effects.
Cell Metab. 31 (6): 1120-1135.e7.
Further Reading
-
Bocchini V & Angeletti PU (1969) The nerve growth factor: purification as a 30,000-molecular-weight protein.
Proc Natl Acad Sci U S A. 64 (2): 787-94.
- Synonyms
- NGF BETA
- UniProt
- P01139
- Entrez Gene
- Ngf
- GO Terms
- GO:0005057 receptor signaling protein activity
- GO:0005788 endoplasmic reticulum lumen
- GO:0007422 peripheral nervous system development
- GO:0008083 growth factor activity
- GO:0019233 sensory perception of pain
- GO:0043524 negative regulation of neuron apoptosis
- GO:0045773 positive regulation of axon extension
- GO:0046928 regulation of neurotransmitter secretion
PMP04Z
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Mouse ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up