TGN38 antibody

Sheep anti Rat TGN38
- Product Type
- Polyclonal Antibody
- Isotype
- Polyclonal IgG
- Specificity
- TGN38
| Sheep anti Rat TGN38 antibody recognizes rat TGN38, a 357 amino acid single pass trans membrane glycoprotein found primarily in the trans-golgi network, and acts as an excellent marker for this cellular organelle (Humphrey et al. 1993). TGN38 is likely to have a role in intracellular transport (McNamara et al. 2004) and plays a role in morphological maintenance (Girotti and Banting 1996). It is the homologue of human TGN46 and macaque TGN47 (Ponnambalam et al. 1996). |
- Target Species
- Rat
- Species Cross-Reactivity
-
Target Species Cross Reactivity Mouse - N.B. Antibody reactivity and working conditions may vary between species.
- Product Form
- Purified IgG - liquid
- Antiserum Preparation
- Antisera to rat TGN38 were raised by repeated immunisation of sheep with highly purified antigen. Purified IgG prepared by affinity chromatography.
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- <0.1% Sodium Azide (NaN3)
0.5% Bovine Serum Albumin
25% Glycerol - Immunogen
- Recombinant fusion protein corresponding to extracellular domain of TGN38.
- Approx. Protein Concentrations
- IgG concentration 0.1 mg/ml
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Immunofluorescence | |||
| Immunohistology - Frozen | 0.1ug/ml | 1ug/ml | |
| Western Blotting | 0.1ug/ml | 1ug/ml |
- Immunohistology
- Fixation with methanol or methanol/acetone recommended.
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Rabbit anti Sheep IgG (H/L):Biotin | 5184-2304 | C E F WB | 1.5 ml |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Rabbit anti Sheep IgG (H/L):Biotin | ||||||
References for TGN38 antibody
-
Vo, Y.P. et al. (2004) Recycling of the dense-core vesicle membrane protein phogrin in Min6 beta-cells.
Biochem Biophys Res Commun. 324: 1004-10. -
Prabhu, Y. et al. (2014) Defective Transport of the Obesity Mutant PC1/3 N222D Contributes to Loss of Function.
Endocrinology. 155: 2391-401. -
Ni-Komatsu, L. et al. (2008) Identification of quinolines that inhibit melanogenesis by altering tyrosinase family trafficking.
Mol Pharmacol. 74:1576-86. -
Mathews, P.M. et al. (2002) Alzheimer's disease-related overexpression of the cation-dependent mannose 6-phosphate receptor increases Abeta secretion: role for altered lysosomal hydrolase distribution in beta-amyloidogenesis.
J Biol Chem. 277: 5299-307. -
Phillips, S.E. et al. (2006) Specific and nonspecific membrane-binding determinants cooperate in targeting phosphatidylinositol transfer protein beta-isoform to the mammalian trans-Golgi network.
Mol Biol Cell. 17: 2498-512. -
Waugh, M.G. et al. (2011) Detergent-free isolation and characterization of cholesterol-rich membrane domains from trans-Golgi network vesicles.
J Lipid Res. 52: 582-9. -
Farah,. C.A. et al. (2006) Tau interacts with Golgi membranes and mediates their association with microtubules.
Cell Motil Cytoskeleton. 63: 710-24. -
Hesse, D. et al. (2010) Altered GLUT4 trafficking in adipocytes in the absence of the GTPase Arfrp1.
Biochem Biophys Res Commun. 394: 896-903.
View The Latest Product References
-
Miranda-Saksena, M. et al. (2002) In rat dorsal root ganglion neurons, herpes simplex virus type 1 tegument forms in the cytoplasm of the cell body.
J Virol. 76: 9934-51. -
Procino, G. et al. (2006) Adipocytes support cAMP-dependent translocation of aquaporin-2 from intracellular sites distinct from the insulin-responsive GLUT4 storage compartment.
Am J Physiol Renal Physiol. 290: F985-94. -
Han, L. et al. (2008) A large form of secretogranin III functions as a sorting receptor for chromogranin A aggregates in PC12 cells.
Mol Endocrinol. 22: 1935-49. -
Probst, O.C. et al. (2006) The 46-kDa mannose 6-phosphate receptor does not depend on endosomal acidification for delivery of hydrolases to lysosomes.
J Cell Sci. 119 (Pt 23): 4935-43. -
Wasmeier, C. et al. (2006) Rab38 and Rab32 control post-Golgi trafficking of melanogenic enzymes.
J Cell Biol. 175: 271-81. -
Hou, J.C. et al. (2006) A specific dileucine motif is required for the GGA-dependent entry of newly synthesized insulin-responsive aminopeptidase into the insulin-responsive compartment.
J Biol Chem. 281: 33457-66. -
Fortin, M.E. et al. (2006) Modulation of GJA1 turnover and intercellular communication by proinflammatory cytokines in the anterior pituitary folliculostellate cell line TtT/GF.
Biol Reprod. 74: 2-12. -
Acquatella-Tran Van Ba, I. et al. (2011) Reg-1α is a new neuronal secreted factor that stimulates neurite outgrowth via the Exostosin tumor-like 3 (EXTL3) receptor.
J Biol Chem. 287: 4726-39. -
Watanabe, T. et al. (2012) A unique ball-shaped Golgi apparatus in the rat pituitary gonadotrope: its functional implications in relation to the arrangement of the microtubule network.
J Histochem Cytochem. 60: 588-602. -
Yamamoto, H. et al. (2016) Posttranslational processing of FGF23 in osteocytes during the osteoblast to osteocyte transition.
Bone. 84: 120-30. -
Koga, D. et al. (2015) Correlative Light and Scanning Electron Microscopy for Observing the Three-Dimensional Ultrastructure of Membranous Cell Organelles in Relation to Their Molecular Components.
J Histochem Cytochem. 63 (12): 968-79. -
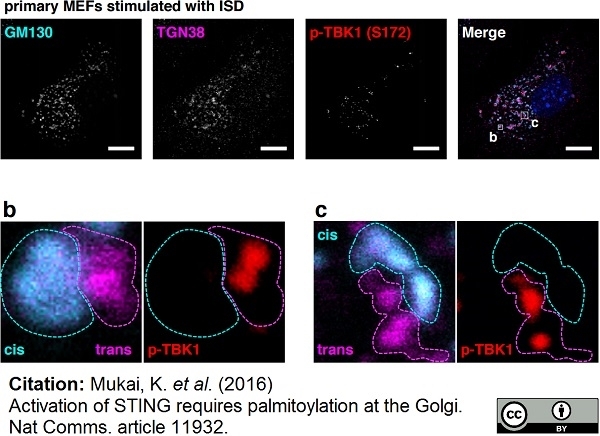
Mukai, K. et al. (2016) Activation of STING requires palmitoylation at the Golgi.
Nat Commun. 7: 11932. -
Koga, D. et al. (2017) Changes in the three-dimensional ultrastructure of membranous organelles in male rat pituitary gonadotropes after castration.
Biomed Res. 38 (1): 1-18. -
Kusumi, S. et al. (2018) Combination of a cryosectioning method and section scanning electron microscopy for immuno-scanning electron microscopy.
Biomed Res. 39 (1): 21-25. -
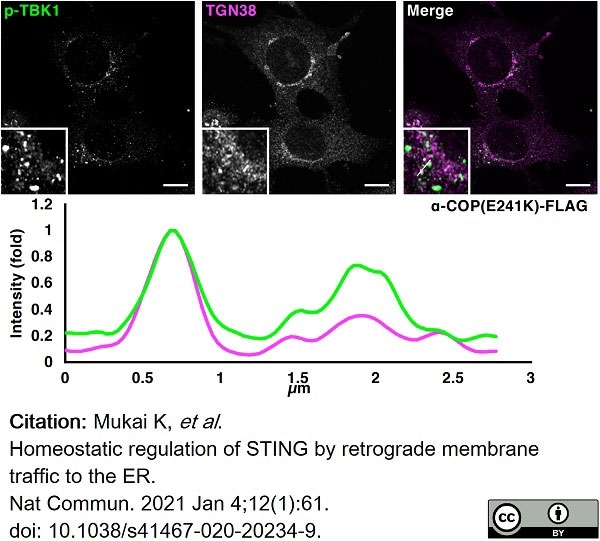
Mukai, K. et al. (2021) Homeostatic regulation of STING by retrograde membrane traffic to the ER.
Nat Commun. 12 (1): 61. -
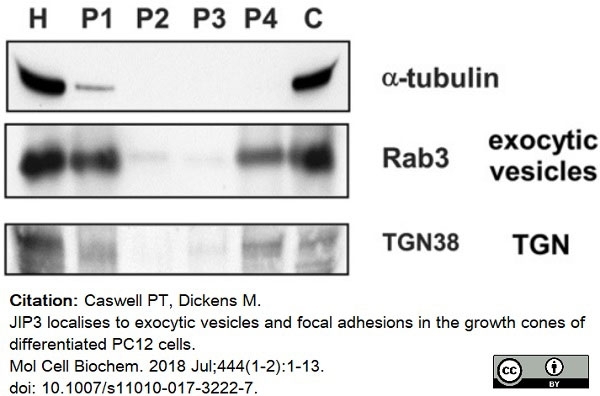
Caswell, P.T. & Dickens, M. (2018) JIP3 localises to exocytic vesicles and focal adhesions in the growth cones of differentiated PC12 cells.
Mol Cell Biochem. 444 (1-2): 1-13. -
Ogawa, E. et al. (2018) The binding of TBK1 to STING requires exocytic membrane traffic from the ER.
Biochem Biophys Res Commun. 503 (1): 138-145. -
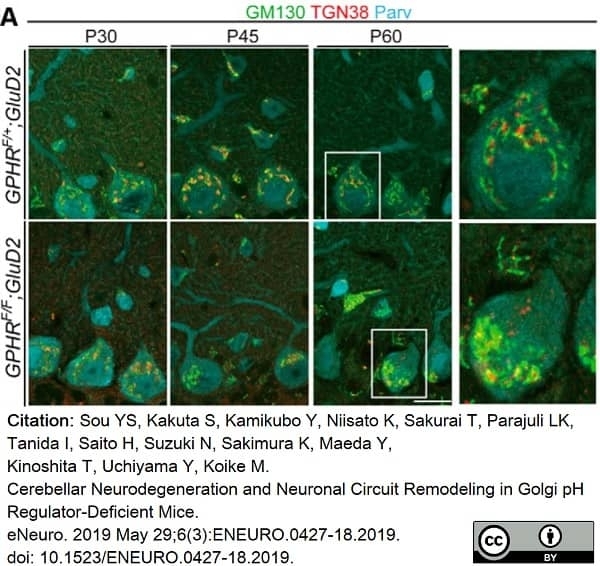
Sou, Y.S. et al. (2019) Cerebellar Neurodegeneration and Neuronal Circuit Remodeling in Golgi pH Regulator-Deficient Mice.
eNeuro. 6(3):ENEURO.0427-18.2019. -
Koga, D. et al. (2021) Applications of Scanning Electron Microscopy Using Secondary and Backscattered Electron Signals in Neural Structure.
Front Neuroanat. 15: 759804. -
Mukai, K. et al. (2021) Homeostatic regulation of STING by retrograde membrane traffic to the ER.
Nat Commun. 12 (1): 61.
Further Reading
-
Luzio, J.P. et al. (1990) Identification, sequencing and expression of an integral membrane protein of the trans-Golgi network (TGN38).
Biochem J. 270: 97-102.
- RRID
- AB_2203272
- UniProt
- P19814
- Entrez Gene
- Tgoln1
- GO Terms
- GO:0005515 protein binding
- GO:0016021 integral to membrane
- GO:0005768 endosome
- GO:0005794 Golgi apparatus
- GO:0006895 Golgi to endosome transport
AHP499G
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Rat ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up