von Willebrand Factor antibody
Sheep anti Human von Willebrand Factor
- Product Type
- Polyclonal Antibody
- Isotype
- Polyclonal IgG
- Specificity
- von Willebrand Factor
Filter by Application:
IF F WB IC Reset| Sheep anti Human von Willebrand Factor antibody recognizes Human von Willebrand factor, a glycoprotein synthesized in endothelial cells and megakaryocytes and circulating in the blood as a noncovalent complex by association with factor VIII. |
- Target Species
- Human
- Species Cross-Reactivity
-
Target Species Cross Reactivity Rat Pig - N.B. Antibody reactivity and working conditions may vary between species.
- Product Form
- Purified IgG - liquid
- Preparation
- Purified IgG was prepared from serum by ion exchange chromatography
- Antiserum Preparation
- Antisera to von Willebrand factor were raised by repeated immunisation of sheep with highly purified antigen. Purified IgG was prepared from serum by ion exchange chromatography
- Buffer Solution
- Glycine buffered saline
- Preservative Stabilisers
- <0.1% Sodium Azide (NaN3)
0.1% EACA
0.01% Benzamidine
1 mM EDTA - Immunogen
- Purified human von Willebrand factor prepared from citrated human plasma
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| ELISA | |||
| Flow Cytometry | |||
| Immunodiffusion | Neat | ||
| Immunofluorescence | |||
| Immunohistology - Frozen 1 | |||
| Immunohistology - Paraffin |
- 1The epitope recognised by this antibody is reported to be sensitive to formaldehyde fixation and tissue processing. Bio-Rad recommends the use of acetone fixation for frozen sections.
- Histology Positive Control Tissue
- Tonsil
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Rabbit anti Sheep IgG (H/L):Biotin | 5184-2304 | C E F WB | 1.5 ml |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Rabbit anti Sheep IgG (H/L):Biotin | ||||||
References for von Willebrand Factor antibody
-
Tait, A.S. et al. (2001) Phenotype changes resulting in high-affinity binding of von Willebrand factor to recombinant glycoprotein Ib-IX: analysis of the platelet-type von Willebrand disease mutations.
Blood. 98: 1812-8. -
Kriehuber, E. et al. (2001) Isolation and characterization of dermal lymphatic and blood endothelial cells reveal stable and functionally specialized cell lineages.
J Exp Med. 194: 797-808. -
Lee, L. et al. (2002) Identification of synovium-specific homing peptides by in vivo phage display selection.
Arthritis Rheum. 46: 2109-20. -
Blades, M.C. et al. (2002) Stromal cell-derived factor 1 (CXCL12) induces human cell migration into human lymph nodes transplanted into SCID mice.
J Immunol. 168 (9): 4308-17. -
Blagoveshchenskaya, A.D. et al. (2002) Selective and signal-dependent recruitment of membrane proteins to secretory granules formed by heterologously expressed von Willebrand factor.
Mol Biol Cell. 13:1582-93. -
Friis,T. et al. (2003) A quantitative ELISA-based co-culture angiogenesis and cell proliferation assay.
APMIS. 111: 658-68. -
Butthep, P. et al. (2004) Endothelial injury and altered hemodynamics in thalassemia.
Clin Hemorheol Microcirc. 31: 287-93. -
Holmén, C. et al. (2005) Heterogeneity of human nasal vascular and sinusoidal endothelial cells from the inferior turbinate.
Am J Respir Cell Mol Biol. 32: 18-27.
View The Latest Product References
-
Zhu, B. et al. (2005) Cyclic GMP-specific phosphodiesterase 5 regulates growth and apoptosis in pulmonary endothelial cells.
Am J Physiol Lung Cell Mol Physiol. 289: L196-206. -
Glinert, I.S. et al. (2008) Photodynamic ablation of a selected rat embryo: a model for the treatment of extrauterine pregnancy.
Hum Reprod. 23: 1491-8. -
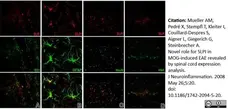
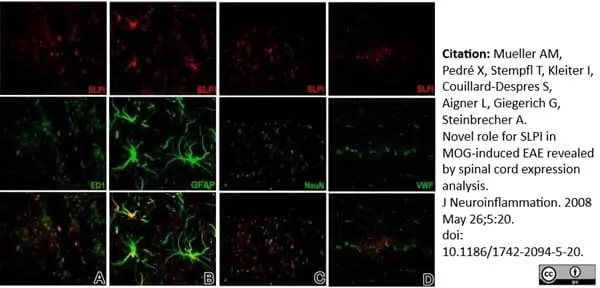
Mueller, A.M. et al. (2008) Novel role for SLPI in MOG-induced EAE revealed by spinal cord expression analysis.
J Neuroinflammation. 5: 20. -
Renault, M.A. et al. (2009) The Hedgehog transcription factor Gli3 modulates angiogenesis.
Circ Res. 105: 818-26. -
Babich, V. et al. (2009) Differential effect of extracellular acidosis on the release and dispersal of soluble and membrane proteins secreted from the Weibel-Palade body.
J Biol Chem. 284: 12459-68. -
Kiskin, N.I. et al. (2010) Protein mobilities and P-selectin storage in Weibel-Palade bodies.
J Cell Sci. 123: 2964-75. -
Chowdhury, F. et al. (2010) Interactions between endothelial cells and epithelial cells in a combined cell model of airway mucosa: effects on tight junction permeability.
Exp Lung Res. 36: 1-11. -
Knipe, L. et al. (2010) A revised model for the secretion of tPA and cytokines from cultured endothelial cells.
Blood. 116: 2183-91. -
Johnson, L.A. & Jackson, D.G. (2010) Inflammation-induced secretion of CCL21 in lymphatic endothelium is a key regulator of integrin-mediated dendritic cell transmigration.
Int Immunol. 22 (10): 839-49. -
Johnson, L.A. and Jackson, D.G. (2010) Inflammation-induced secretion of CCL21 in lymphatic endothelium is a key regulator of integrin-mediated dendritic cell transmigration.
Int Immunol. 22: 839-49. -
Johnson, L.A. & Jackson, D.G. (2013) The chemokine CX3CL1 promotes trafficking of dendritic cells through inflamed lymphatics.
J Cell Sci. 126 (Pt 22): 5259-70. -
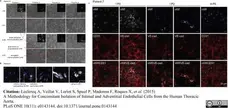
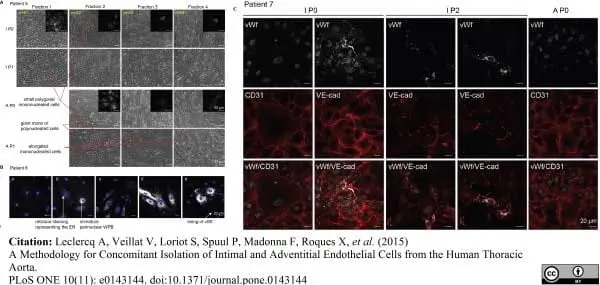
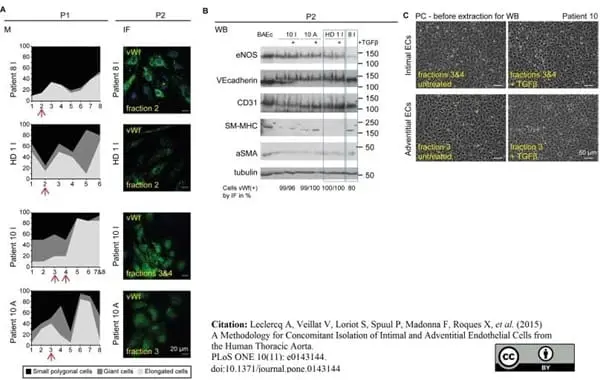
Leclercq, A. et al. (2015) A Methodology for Concomitant Isolation of Intimal and Adventitial Endothelial Cells from the Human Thoracic Aorta.
PLoS One. 10 (11): e0143144. -
Cheung, K. et al. (2015) CD31 signals confer immune privilege to the vascular endothelium.
Proc Natl Acad Sci U S A. 112 (43): E5815-24. -
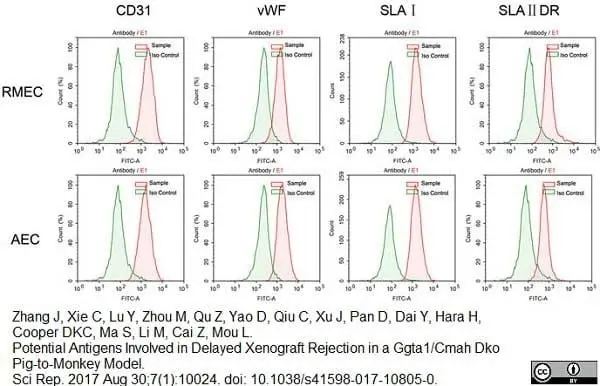
Zhang, J. et al. (2017) Potential Antigens Involved in Delayed Xenograft Rejection in a Ggta1/Cmah Dko Pig-to-Monkey Model.
Sci Rep. 7 (1): 10024. -
Rivera, J.R. & Czuprynski, C.J. (2019) Procoagulant activity of bovine neutrophils incubated with conditioned media or extracellular vesicles from Histophilus somni. stimulated bovine brain endothelial cells.
Vet Immunol Immunopathol. 211: 49-57. -
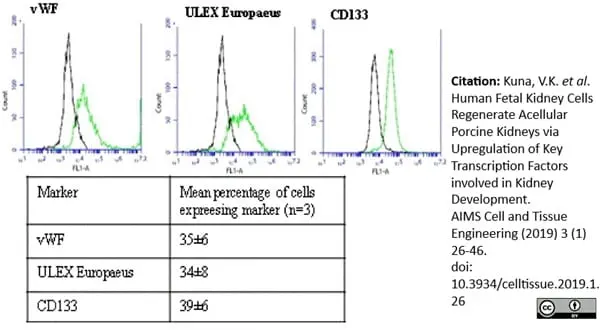
Kuna, V.K. et al. (2019) Human fetal kidney cells regenerate acellular porcine kidneys via upregulation of key transcription factors involved in kidney development.
AIMS Cell and Tissue Engineering. 3 (1): 26-46. -
Boeckelmann, D. et al. (2020) Panel Sequencing Identified a Novel Splice Site Mutation in Hermansky-Pudlak Syndrome Type 1 Patients
Arch Clin Med Case Rep. 4 (6): 1172-81 -
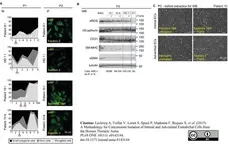
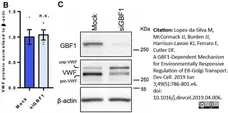
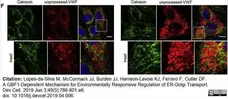
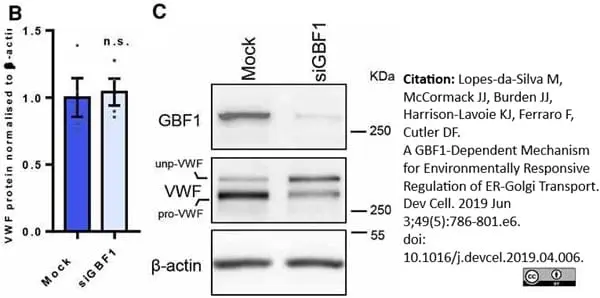
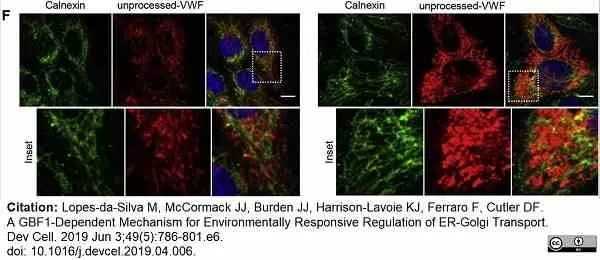
Lopes-da-Silva, M. et al. (2019) A GBF1-Dependent Mechanism for Environmentally Responsive Regulation of ER-Golgi Transport.
Dev Cell. 49 (5): 786-801.e6. -
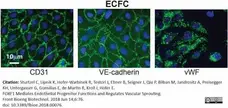
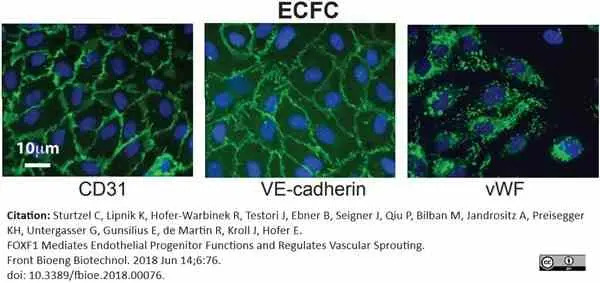
Sturtzel, C. et al. (2018) FOXF1 Mediates Endothelial Progenitor Functions and Regulates Vascular Sprouting.
Front Bioeng Biotechnol. 6: 76.
- Synonyms
- Factor VIII Related Antigen
- RRID
- AB_322241
- UniProt
- P04275
- Entrez Gene
- VWF
- GO Terms
- GO:0001948 glycoprotein binding
- GO:0002020 protease binding
- GO:0002576 platelet degranulation
- GO:0051087 chaperone binding
- GO:0005783 endoplasmic reticulum
- GO:0005178 integrin binding
- GO:0005518 collagen binding
- GO:0005578 proteinaceous extracellular matrix
- GO:0007597 blood coagulation, intrinsic pathway
- View More GO Terms
- GO:0019865 immunoglobulin binding
- GO:0030168 platelet activation
- GO:0031093 platelet alpha granule lumen
- GO:0031589 cell-substrate adhesion
- GO:0033093 Weibel-Palade body
- GO:0042803 protein homodimerization activity
- GO:0051260 protein homooligomerization
- GO:0047485 protein N-terminus binding
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Human ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up