CD45 antibody | 1.11.32





Mouse anti Sheep CD45:RPE
- Product Type
- Monoclonal Antibody
- Clone
- 1.11.32
- Isotype
- IgG1
- Specificity
- CD45
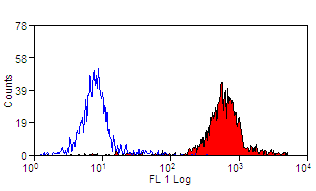
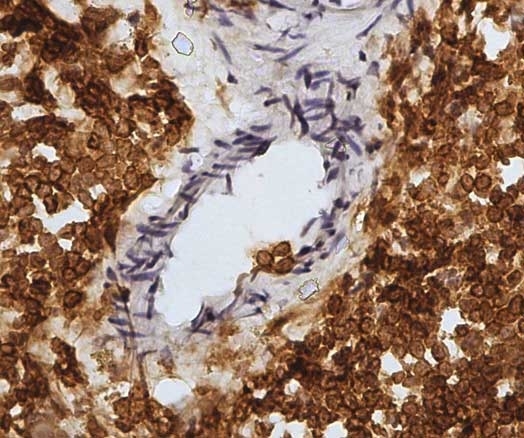
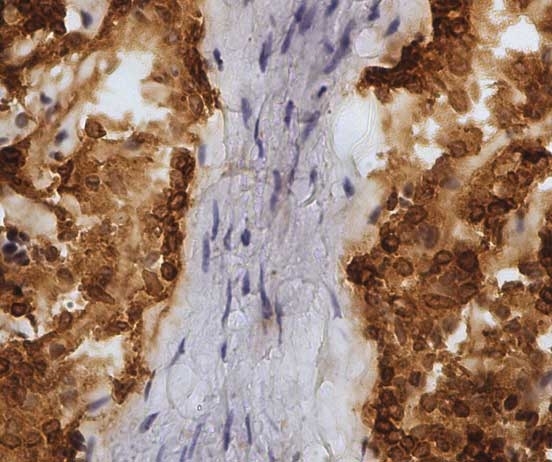
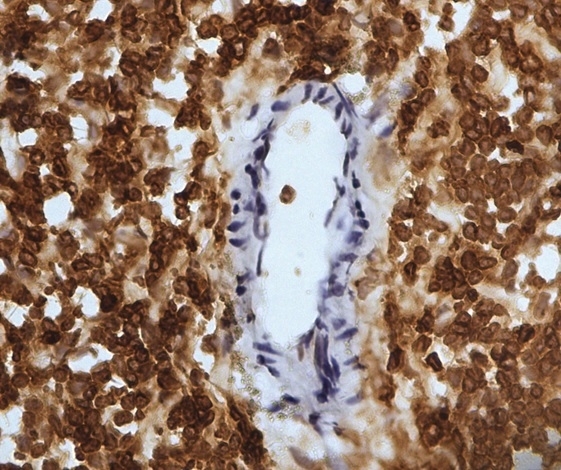
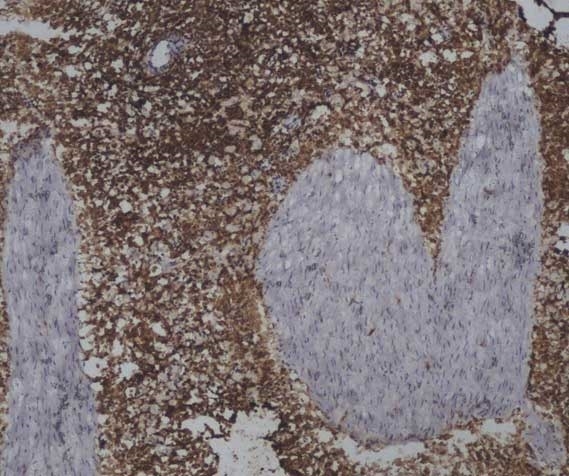
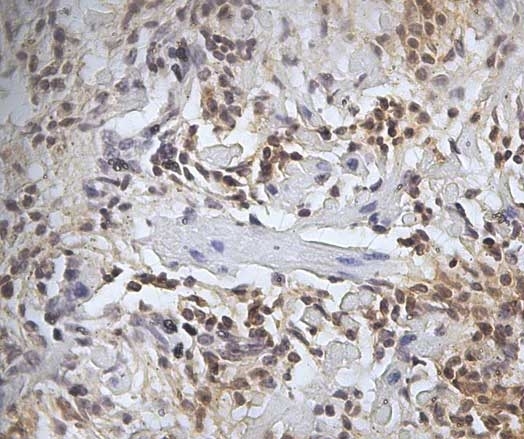
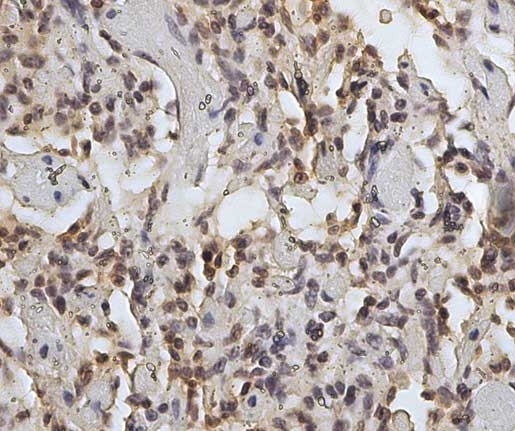
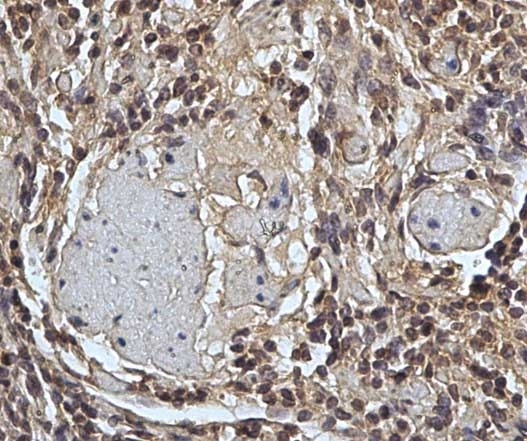
| Mouse anti Sheep CD45 antibody, clone 1.11.32 recognizes the ovine CD45 (Leucocyte common antigen), expressed on all ovine lymphocytes, macrophages and granulocytes. Mouse anti Sheep CD45 antibody, clone 1.11.32 immunoprecipitates CD45 molecules of 190 kDa, 210 kDa and 225 kDa from lymph node lysates. |
- Target Species
- Sheep
- Species Cross-Reactivity
-
Target Species Cross Reactivity Bovine Goat - N.B. Antibody reactivity and working conditions may vary between species.
- Product Form
- Purified IgG conjugated to R. Phycoerythrin (RPE) - lyophilized
- Reconstitution
- Reconstitute with 1 ml distilled water
- Preparation
- Purified IgG prepared by affinity chromatography on Protein A from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- 0.09% sodium azide (NaN3)
1% bovine serum albumin
5% sucrose - Immunogen
- Ovine efferent lymphatic duct lymphocytes.
- Fusion Partners
- Spleen cells from immunized BALB/c mice were fused with cells of the mouse NS-1 myeloma cell line.
- Max Ex/Em
-
Fluorophore Excitation Max (nm) Emission Max (nm) RPE 488nm laser 496 578 - Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
DO NOT FREEZE.
This product should be stored undiluted. This product is photosensitive and should be protected from light. Should this product contain a precipitate we recommend microcentrifugation before use.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Flow Cytometry | Neat |
- Flow Cytometry
- Use 10μl of the suggested working dilution to label 106 cells in 100μl
How to Use the Spectraviewer
Watch the Tool Tutorial Video ▸- Start by selecting the application you are interested in, with the option to select an instrument from the drop down menu or create a customized instrument
- Select the fluorophores or fluorescent proteins you want to include in your panel to check compatibility
- Select the lasers and filters you wish to include
- Select combined or multi-laser view to visualize the spectra
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Mouse IgG1 Negative Control:RPE | MCA928PE | F | 100 Tests | Log in | |||
| List Price | Your Price | ||||||
| Log in | |||||||
| Description | Mouse IgG1 Negative Control:RPE | ||||||
Source Reference
-
Maddox, J.F. et al. (1985) The sheep analogue of leucocyte common antigen (LCA).
Immunology. 55 (2): 347-53.
References for CD45 antibody
-
Mackay, C.R. et al. (1987) A monoclonal antibody to the p220 component of sheep LCA identifies B cells and a unique lymphocyte subset.
Cell Immunol. 110 (1): 46-55. -
Zannettino, A.C. et al. (2010) Comparative assessment of the osteoconductive properties of different biomaterials in vivo seeded with human or ovine mesenchymal stem/stromal cells.
Tissue Eng Part A. 16 (12): 3579-87. -
Mackay, C.R. et al. (1989) Gamma/delta T cells express a unique surface molecule appearing late during thymic development.
Eur J Immunol. 19 (8): 1477-83. -
Breugelmans, S. et al. (2011) Immunoassay of lymphocyte subsets in ovine palatine tonsils.
Acta Histochem. 113 (4): 416-22. -
Breugelmans, S. et al. (2011) Differences between the ovine tonsils based on an immunohistochemical quantification of the lymphocyte subpopulations.
Comp Immunol Microbiol Infect Dis. 34: 217-25. -
Herdrich, B.J. et al. (2010) Regenerative healing following foetal myocardial infarction.
Eur J Cardiothorac Surg. 38: 691-8. -
Reichert, J.C. et al. (2010) Ovine bone- and marrow-derived progenitor cells and their potential for scaffold-based bone tissue engineering applications in vitro and in vivo.
J Tissue Eng Regen Med. 4: 565-76. -
Galinsky, R. et al. (2011) Effect of intra-amniotic lipopolysaccharide on nephron number in preterm fetal sheep.
Am J Physiol Renal Physiol. 301 (2): F280-5.
View The Latest Product References
-
Kallapur, S.G. et al. (2011) Pulmonary and systemic inflammatory responses to intra-amniotic IL-1α in fetal sheep.
Am J Physiol Lung Cell Mol Physiol. 301 (3): L285-95. -
Stickler, P. et al. (2010) Cyclically stretching developing tissue in vivo enhances mechanical strength and organization of vascular grafts.
Acta Biomater. 6 (7): 2448-56. -
Berardinelli, P. et al. (2013) Role of amniotic fluid mesenchymal cells engineered on MgHA/collagen-based scaffold allotransplanted on an experimental animal study of sinus augmentation.
Clin Oral Investig. 17 (7): 1661-75. -
Geherin, S.A. et al. (2012) The skin, a novel niche for recirculating B cells.
J Immunol. 188: 6027-35. -
Jahroomishirazi, R. et al. (2014) Isolation and Characterization of CD271+ Stem Cells Derived from Sheep Dermal Skin.
Cells Tissues Organs. 200 (2): 141-52. -
Stephens, E.H. et al. (2016) Extracellular matrix remodeling in wound healing of critical size defects in the mitral valve leaflet.
Heart Vessels. 31 (7): 1186-95. -
Mokhtari, S. et al. (2016) Boosting Hematopoietic Engraftment after in Utero Transplantation through Vascular Niche Manipulation.
Stem Cell Reports. 6 (6): 957-69. -
Bischoff, J. et al. (2016) CD45 Expression in Mitral Valve Endothelial Cells After Myocardial Infarction.
Circ Res. 119 (11): 1215-25. -
Lydon, H. et al. (2018) Peripheral mononuclear blood cell apheresis in a preclinical ovine model.
BMC Vet Res. 14 (1): 47. -
Schwarz, E.R. et al. (2020) Experimental Infection of Mid-Gestation Pregnant Female and Intact Male Sheep with Zika Virus.
Viruses. 12 (3)Mar 07 [Epub ahead of print]. -
Barboni, B. et al. (2013) Synthetic bone substitute engineered with amniotic epithelial cells enhances bone regeneration after maxillary sinus augmentation.
PLoS One. 8 (5): e63256. -
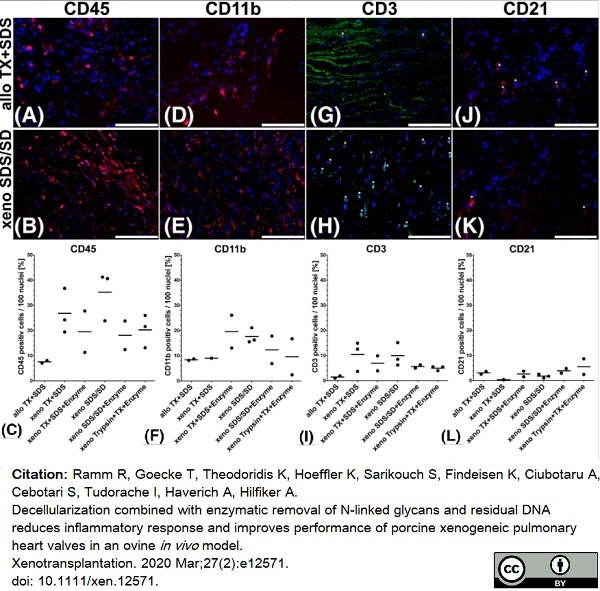
Ramm, R. et al. (2020) Decellularization combined with enzymatic removal of N-linked glycans and residual DNA reduces inflammatory response and improves performance of porcine xenogeneic pulmonary heart valves in an ovine in vivo model.
Xenotransplantation. 27 (2): e12571. -
López-Fernández, A. et al. (2020) Effect of Allogeneic Cell-Based Tissue-Engineered Treatments in a Sheep Osteonecrosis Model.
Tissue Eng Part A. 26 (17-18): 993-1004. -
Wooldridge, A.L. et al. (2019) Maternal allergic asthma during pregnancy alters fetal lung and immune development in sheep: potential mechanisms for programming asthma and allergy.
J Physiol. 597 (16): 4251-4262. -
Savy, V. et al. (2021) Effect of Embryo Aggregation on In Vitro Development of Adipose-Derived Mesenchymal Stem Cell-Derived Bovine Clones.
Cell Reprogram. 23 (5): 277-89. -
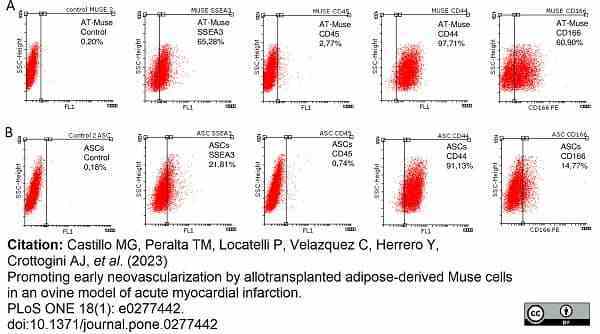
Castillo, M.G. et al. (2023) Promoting early neovascularization by allotransplanted adipose-derived Muse cells in an ovine model of acute myocardial infarction.
PLoS One. 18 (1): e0277442. -
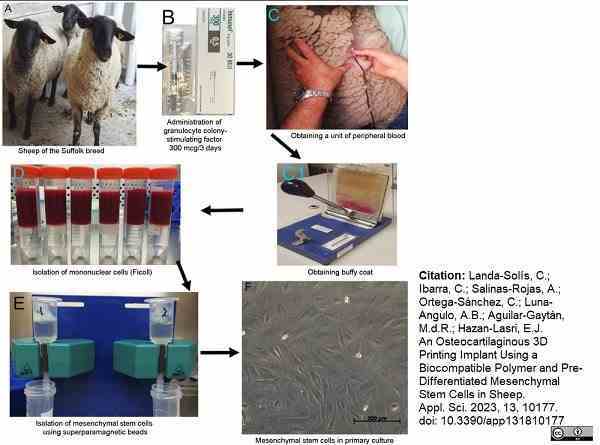
Landa-Solís, C. et al. (2023) An Osteocartilaginous 3D Printing Implant Using a Biocompatible Polymer and Pre-Differentiated Mesenchymal Stem Cells in Sheep
Applied Sciences. 13 (18): 10177. -
Abe, T. et al. (2021) Fetal sheep support the development of hematopoietic cells in vivo from human induced pluripotent stem cells.
Exp Hematol. 95: 46-57.e8. -
Landa-Solís, C. et al. (2023) An Osteocartilaginous 3D Printing Implant Using a Biocompatible Polymer and Pre-Differentiated Mesenchymal Stem Cells in Sheep
Applied Sciences. 13 (18): 10177.
- Synonyms
- LCA
- RRID
- AB_324751
MCA2220PE
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Sheep ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up