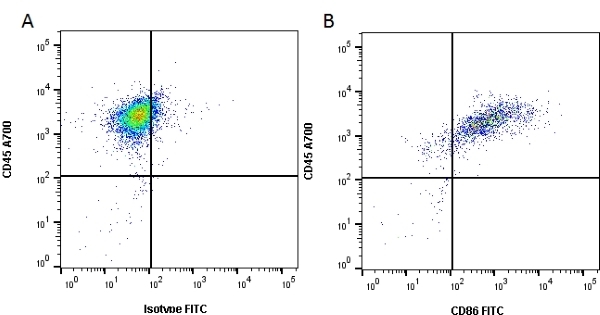
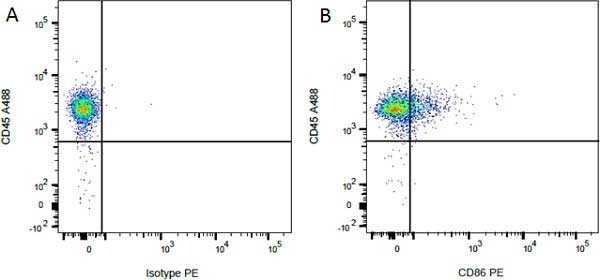
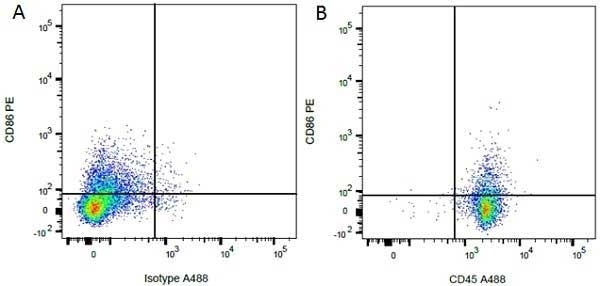
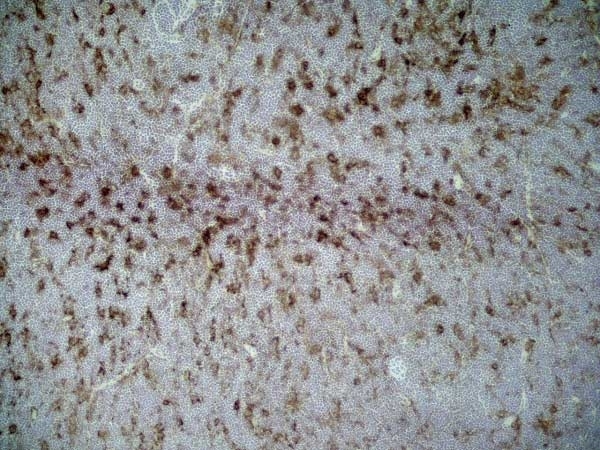
CD86 antibody | 24F








Mouse anti Rat CD86:RPE
- Product Type
- Monoclonal Antibody
- Clone
- 24F
- Isotype
- IgG1
- Specificity
- CD86
| Mouse anti Rat CD86 antibody, clone 24F recognizes rat CD86, otherwise known as B7-2, a type I transmembrane protein and member of the Ig superfamily, which acts as a ligand for both CD28 and CD152 (CTLA-4), and is primarily expressed on antigen presenting cells (APCs) including dendritic cells, and also on germinal centre B cells and macrophages. Like CD80, CD86 is an accessory molecule which functions in the CD28-CD80/CD86 co-stimulatory pathway, vital for T cell activation, crosstalk between T and B cells, and Th2-mediated Ig production. Mouse anti Rat CD86 antibody, clone 24F has been shown to block the co-stimulatory activity of rat CD86 (Maeda et al. 1997). |
- Target Species
- Rat
- Product Form
- Purified IgG conjugated to R. Phycoerythrin (RPE) - lyophilized
- Reconstitution
- Reconstitute with 1ml distilled water
- Preparation
- Purified IgG prepared by affinity chromatography on Protein G from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- 0.09% Sodium Azide (NaN3)
1% Bovine Serum Albumin
5% Sucrose - Immunogen
- HTLV-1 transformed Lewis-S1 cells.
- Fusion Partners
- Spleen cells from immunised Balb/c mice were fused with cells of the P3U1 mouse myeloma cell line.
- Max Ex/Em
-
Fluorophore Excitation Max (nm) Emission Max (nm) RPE 488nm laser 496 578 - Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
After reconstitution store at +4oC.
DO NOT FREEZE. This product should be stored undiluted. This product is photosensitive and should be protected from light. Should this product contain a precipitate we recommend microcentrifugation before use.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Flow Cytometry | Neat |
- Flow Cytometry
- Use 10ul of the suggested working dilution to label 1x106 cells in 100ul.
How to Use the Spectraviewer
Watch the Tool Tutorial Video ▸- Start by selecting the application you are interested in, with the option to select an instrument from the drop down menu or create a customized instrument
- Select the fluorophores or fluorescent proteins you want to include in your panel to check compatibility
- Select the lasers and filters you wish to include
- Select combined or multi-laser view to visualize the spectra
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Mouse IgG1 Negative Control:RPE | MCA1209PE | F | 100 Tests | Log in | |||
| List Price | Your Price | ||||||
| Log in | |||||||
| Description | Mouse IgG1 Negative Control:RPE | ||||||
Source Reference
-
Maeda, K. et al. (1997) Characterization of rat CD80 and CD86 by molecular cloning and mAb.
Int Immunol. 9 (7): 993-1000.
References for CD86 antibody
-
Damoiseaux, J.G. et al. (1998) Costimulatory molecules CD80 and CD86 in the rat; tissue distribution and expression by antigen-presenting cells.
J Leukoc Biol. 64 (6): 803-9. -
Kano, M. et al. (1998) A crucial role of host CD80 and CD86 in rat cardiac xenograft rejection in mice.
Transplantation. 65: 837-43. -
Sacedón, R. et al. (1999) Glucocorticoid-mediated regulation of thymic dendritic cell function.
Int Immunol. 11: 1217-24. -
Hanabuchi, S. et al. (2000) Development of human T-cell leukemia virus type 1-transformed tumors in rats following suppression of T-cell immunity by CD80 and CD86 blockade.
J Virol. 74: 428-35. -
Tamatani, T. et al. (2000) AILIM/ICOS: a novel lymphocyte adhesion molecule.
Int Immunol. 12: 51-5. -
Kawai, T. et al. (2000) T(h)1 transmigration anergy: a new concept of endothelial cell-T cell regulatory interaction.
Int Immunol. 12: 937-48. -
Macphee, I.A. et al. (2002) The Th2-response in mercuric chloride-induced autoimmunity requires continuing costimulation via CD28.
Clin Exp Immunol. 129: 405-10. -
Ghiringhelli, F. et al. (2005) Tumor cells convert immature myeloid dendritic cells into TGF-beta-secreting cells inducing CD4+CD25+ regulatory T cell proliferation.
J Exp Med. 202: 919-29.
View The Latest Product References
-
MacPhee, I.A. et al. (2006) Blockade of OX40-ligand after initial triggering of the T helper 2 response inhibits mercuric chloride-induced autoimmunity.
Immunology. 117: 402-8. -
Yrlid, U. et al. (2006) A distinct subset of intestinal dendritic cells responds selectively to oral TLR7/8 stimulation.
Eur J Immunol. 36: 2639-48. -
Dilek, N. et al. (2012) Control of transplant tolerance and intragraft regulatory T cell localization by myeloid-derived suppressor cells and CCL5.
J Immunol. 188: 4209-16. -
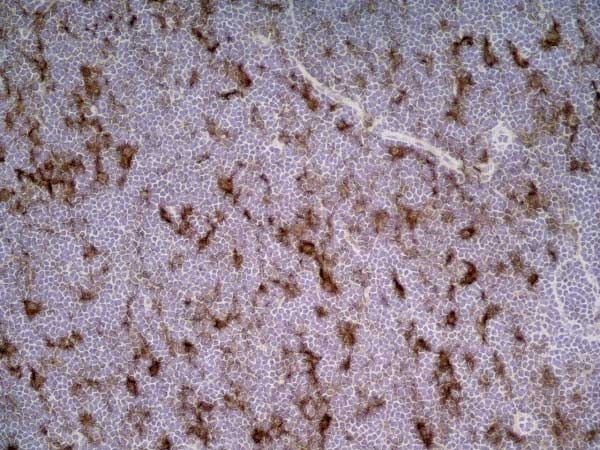
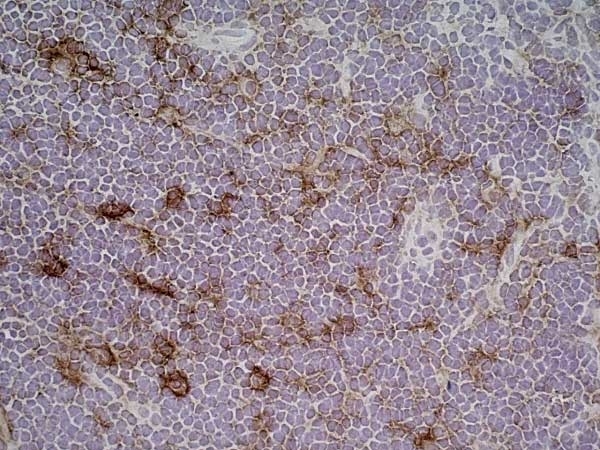
Matsumoto, S. et al. (2015) CD200+ and CD200- macrophages accumulated in ischemic lesions of rat brain: the two populations cannot be classified as either M1 or M2 macrophages.
J Neuroimmunol. 282: 7-20. -
Patil, P.S. et al. (2016) Fluorinated methacrylamide chitosan hydrogels enhance collagen synthesis in wound healing through increased oxygen availability.
Acta Biomater. 36: 164-74. -
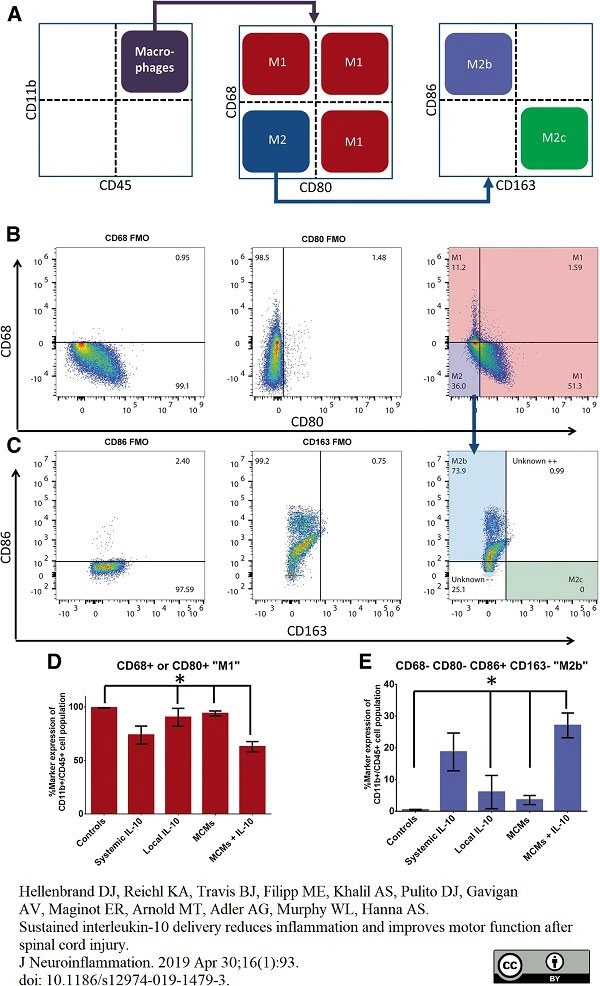
Hellenbrand, D.J. et al. (2019) Sustained interleukin-10 delivery reduces inflammation and improves motor function after spinal cord injury.
J Neuroinflammation. 16 (1): 93. -
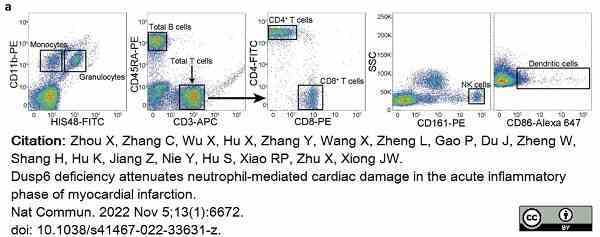
Zhou, X. et al. (2022) Dusp6 deficiency attenuates neutrophil-mediated cardiac damage in the acute inflammatory phase of myocardial infarction.
Nat Commun. 13 (1): 6672.
- Synonyms
- B7-2
- RRID
- AB_2075122
MCA2874PE
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Rat ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up