CD8 Alpha antibody | OX-8








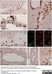
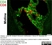
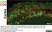




Mouse anti Rat CD8 Alpha:Alexa Fluor® 647
- Product Type
- Monoclonal Antibody
- Clone
- OX-8
- Isotype
- IgG1
- Specificity
- CD8 Alpha
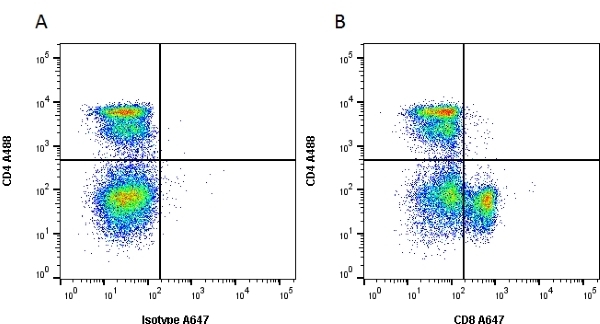
| Mouse anti Rat CD8α, clone MRC OX-8, recognizes the rat CD8 alpha cell surface antigen, expressed by a subset of T lymphocytes, most thymocytes and the majority of NK cells. Mouse anti Rat CD8α, clone MRC OX-8 is suitable for use in in vitro blocking assays (Popov et al.2001). Mouse anti Rat CD8α, clone MRC OX-8 reacts with paraffin-embedded material following PLP Fixation (periodate-lysine paraformaldehyde) (Whiteland et al. 1995). Mouse anti Rat CD8α, clone MRC OX-8 is routinely tested in flow cytometry on rat splenocytes. |
- Target Species
- Rat
- Product Form
- Purified IgG conjugated to Alexa Fluor® 647 - liquid
- Preparation
- Purified IgG prepared by affinity chromatography on Protein A from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
0.09% Sodium Azide 1% Bovine Serum Albumin - Immunogen
- Rat thymocyte membrane glycoproteins.
- Approx. Protein Concentrations
- IgG concentration 0.05 mg/ml
- Fusion Partners
- Spleen cells from immunized BALB/c mice were fused with cells of the mouse NS1 myeloma cell line.
- Max Ex/Em
-
Fluorophore Excitation Max (nm) Emission Max (nm) Alexa Fluor®647 650 665 - Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
- Acknowledgements
- This product is provided under an intellectual property licence from Life Technologies Corporation. The transfer of this product is contingent on the buyer using the purchase product solely in research, excluding contract research or any fee for service research, and the buyer must not sell or otherwise transfer this product or its components for (a) diagnostic, therapeutic or prophylactic purposes; (b) testing, analysis or screening services, or information in return for compensation on a per-test basis; (c) manufacturing or quality assurance or quality control, or (d) resale, whether or not resold for use in research. For information on purchasing a license to this product for purposes other than as described above, contact Life Technologies Corporation, 5791 Van Allen Way, Carlsbad CA 92008 USA or outlicensing@thermofisher.com
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended. This product is photosensitive and should be protected from light.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Flow Cytometry | Neat |
- Flow Cytometry
- Use 10ul of the suggested working dilution to label 106 cells in 100ul.
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Mouse IgG1 Negative Control:Alexa Fluor® 647 | MCA1209A647 | F | 100 Tests/1ml |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse IgG1 Negative Control:Alexa Fluor® 647 | ||||||
Source Reference
-
Brideau, R.J. et al. (1980) Two subsets of rat T lymphocytes defined with monoclonal antibodies.
Eur J Immunol. 10 (8): 609-15.
References for CD8 Alpha antibody
-
Thomas, M.L. & Green, J.R. (1983) Molecular nature of the W3/25 and MRC OX-8 marker antigens for rat T lymphocytes: comparisons with mouse and human antigens.
Eur J Immunol. 13 (10): 855-8. -
Bukovský A et al. (1984) Association of some cell surface antigens of lymphoid cells and cell surface differentiation antigens with early rat pregnancy.
Immunology. 52 (4): 631-40. -
Torres-Nagel, N. et al. (1992) Differential thymus dependence of rat CD8 isoform expression.
Eur J Immunol. 22 (11): 2841-8. -
Whiteland, J.L. et al. (1995) Immunohistochemical detection of T-cell subsets and other leukocytes in paraffin-embedded rat and mouse tissues with monoclonal antibodies.
J Histochem Cytochem. 43 (3): 313-20. -
Mitnacht, R. et al. (1998) Opposite CD4/CD8 lineage decisions of CD4+8+ mouse and rat thymocytes to equivalent triggering signals: correlation with thymic expression of a truncated CD8 alpha chain in mice but not rats.
J Immunol. 160 (2): 700-7. -
Popov, I. et al. (2001) The effect of an anti-HLA-B27 immune response on CTL recognition of Chlamydia.
J Immunol. 167 (6): 3375-82. -
Hashimoto, Y. et al. (2003) Two major histocompatibility complex class I-restricted epitopes of the Borna disease virus p10 protein identified by cytotoxic T lymphocytes induced by DNA-based immunization.
J Virol. 77: 6076-81. -
Bradl, M. et al. (2005) Complementary contribution of CD4 and CD8 T lymphocytes to T-cell infiltration of the intact and the degenerative spinal cord.
Am J Pathol. 166: 1441-50.
View The Latest Product References
-
Latta, M. et al. (2007) CXCR6 is expressed on T cells in both T helper type 1 (Th1) inflammation and allergen-induced Th2 lung inflammation but is only a weak mediator of chemotaxis.
Immunology. 121: 555-64 -
King, G.D. et al. (2008) Flt3L in combination with HSV1-TK-mediated gene therapy reverses brain tumor-induced behavioral deficits.
Mol Ther. 16: 682-90 -
Bode, U. et al. (2008) Dendritic cell subsets in lymph nodes are characterized by the specific draining area and influence the phenotype and fate of primed T cells.
Immunology. 123: 480-90. -
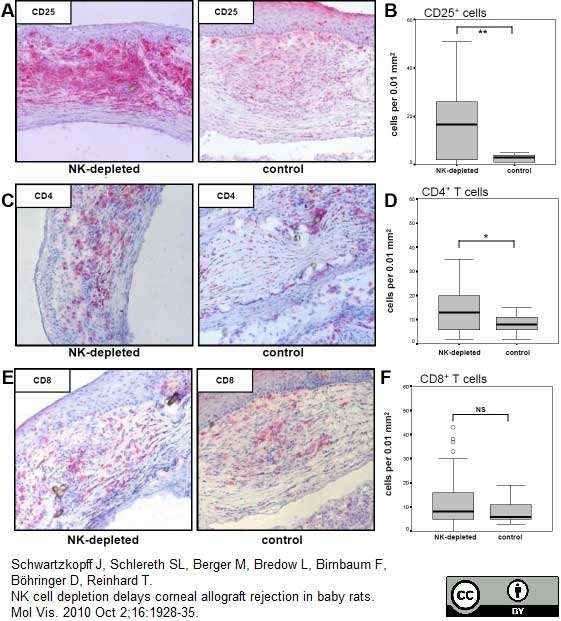
Schwartzkopff, J. et al. (2010) NK cell depletion delays corneal allograft rejection in baby rats.
Mol Vis. 16: 1928-35. -
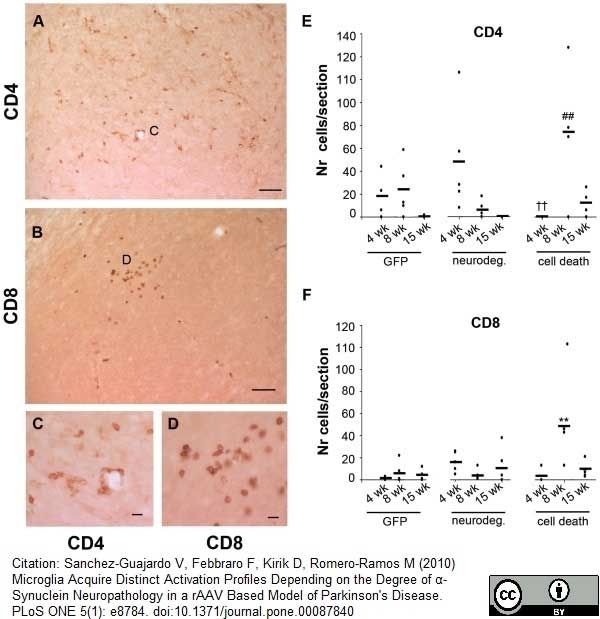
Sanchez-Guajardo, V. (2010) Microglia acquire distinct activation profiles depending on the degree of alpha-synuclein neuropathology in a rAAV based model of Parkinson's disease.
PLoS One. 5: e8784. -
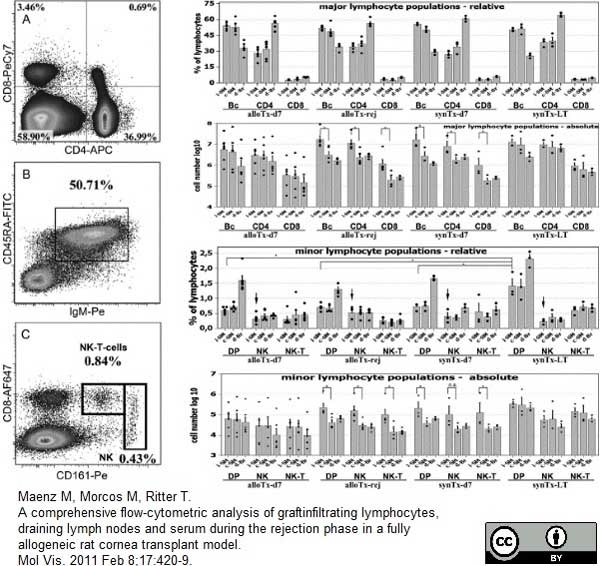
Maenz, M. et al. (2011) A comprehensive flow-cytometric analysis of graft infiltrating lymphocytes, draining lymph nodes and serum during the rejection phase in a fully allogeneic rat cornea transplant model.
Mol Vis. 2011 Feb 8;17:420-9. -
Arndt, T. et al. (2013) A variable CD3⁺ T-cell frequency in peripheral blood lymphocytes associated with type 1 diabetes mellitus development in the LEW.1AR1-iddm rat.
PLoS One. 8 (5): e64305. -
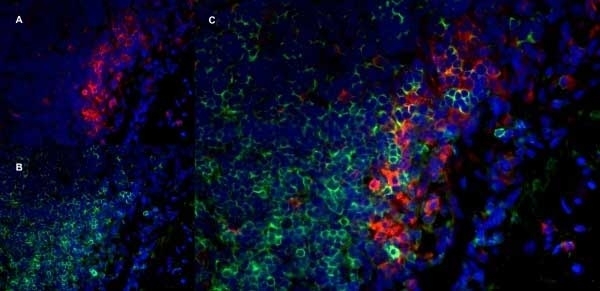
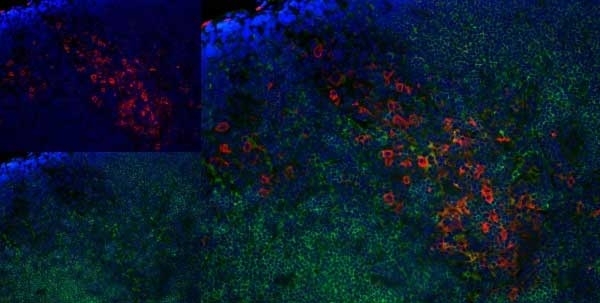
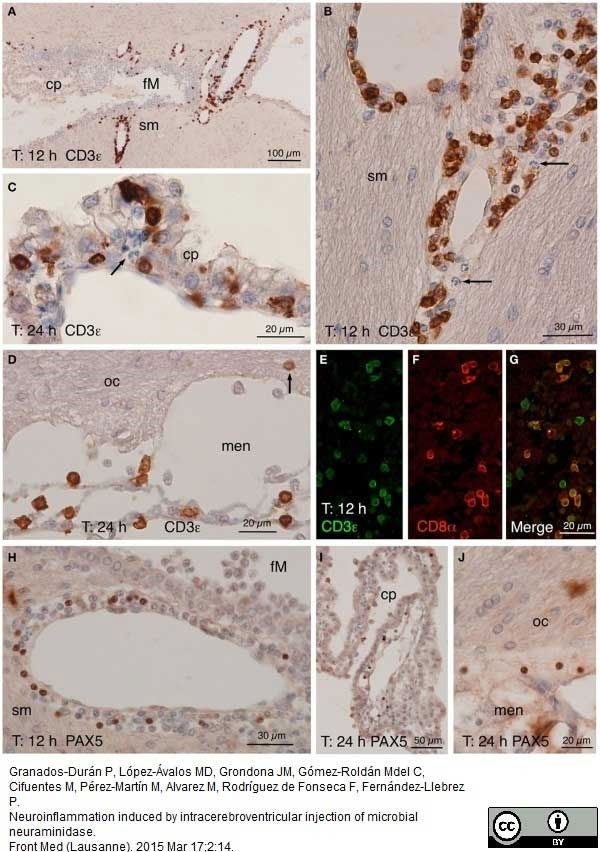
Granados-Durán P et al. (2015) Neuroinflammation induced by intracerebroventricular injection of microbial neuraminidase.
Front Med (Lausanne). 2: 14. -
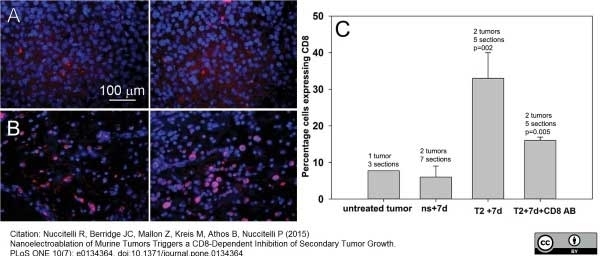
Nuccitelli R et al. (2015) Nanoelectroablation of Murine Tumors Triggers a CD8-Dependent Inhibition of Secondary Tumor Growth.
PLoS One. 10 (7): e0134364. -
Zhang, Z.M. et al. (2016) Lesional accumulation of CD8(+) cells in sciatic nerves of experimental autoimmune neuritis rats.
Neurol Sci. 37 (2): 199-203. -
Pamukcu, O. et al. (2016) Anti-inflammatory role of obestatin in autoimmune myocarditis.
Clin Exp Pharmacol Physiol. 43 (1): 47-55. -
Dabrowska, S. et al. (2019) Human bone marrow mesenchymal stem cell-derived extracellular vesicles attenuate neuroinflammation evoked by focal brain injury in rats.
J Neuroinflammation. 16 (1): 216. -
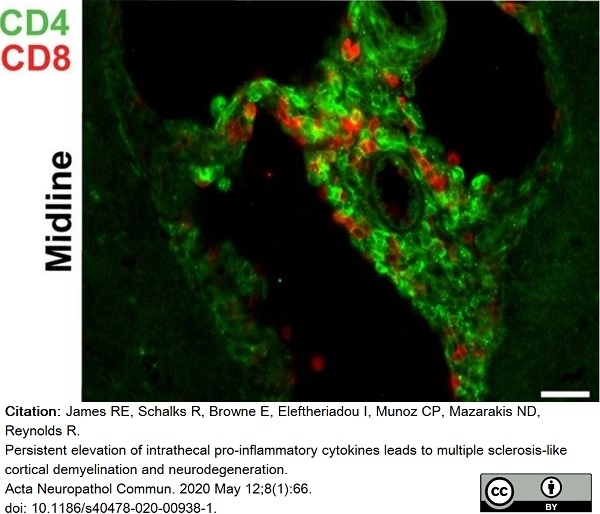
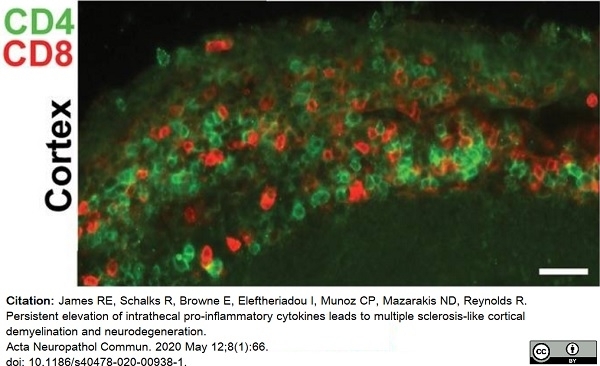
James, R.E. et al. (2020) Persistent elevation of intrathecal pro-inflammatory cytokines leads to multiple sclerosis-like cortical demyelination and neurodegeneration.
Acta Neuropathol Commun. 8 (1): 66. -
Matsuyama, S. et al. (2021) Properties of macrophages and lymphocytes appearing in rat renal fibrosis followed by repeated injection of cisplatin.
J Vet Med Sci. 83 (9): 1435-42. -
Dabrowska, S. et al. (2021) Neuroinflammation evoked by brain injury in a rat model of lacunar infarct.
Exp Neurol. 336: 113531. -
Schmiedl, A. et al. (2021) Lung development and immune status under chronic LPS exposure in rat pups with and without CD26/DPP4 deficiency.
Cell Tissue Res. 386 (3): 617-36. -
Zakerkish, F. et al. (2021) Differential effects of the immunosuppressive calcineurin inhibitors cyclosporine-A and tacrolimus on ovulation in a murine model.
Hum Reprod Open. 2021 (2): hoab012. -
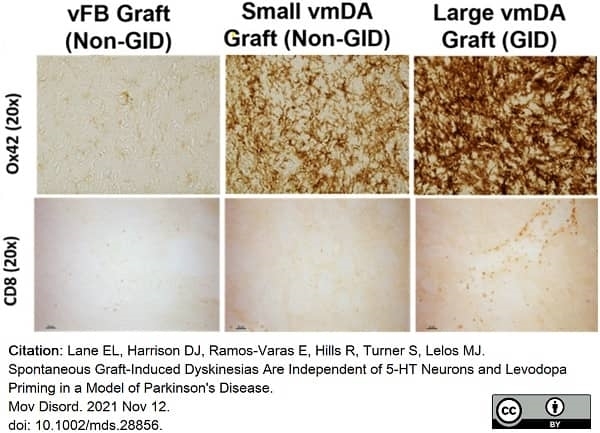
Lane, E.L. et al. (2022) Spontaneous Graft-Induced Dyskinesias Are Independent of 5-HT Neurons and Levodopa Priming in a Model of Parkinson's Disease.
Mov Disord. 37 (3): 613-9. -
Silva, B.A. et al. (2022) Understanding the role of the blood brain barrier and peripheral inflammation on behavior and pathology on ongoing confined cortical lesions.
Mult Scler Relat Disord. 57: 103346. -
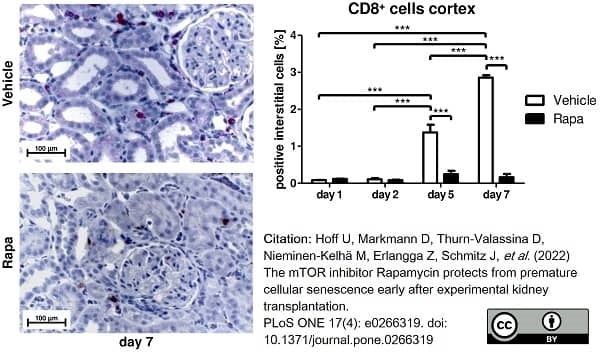
Hoff, U. et al. (2022) The mTOR inhibitor Rapamycin protects from premature cellular senescence early after experimental kidney transplantation.
PLoS One. 17 (4): e0266319. -
Gad, R.A. et al. (2022) Mitigating effects of Passiflora incarnata. on oxidative stress and neuroinflammation in case of pilocarpine-Induced status epilepticus model
J King Saud Uni - Science. 34 (3): 101886. -
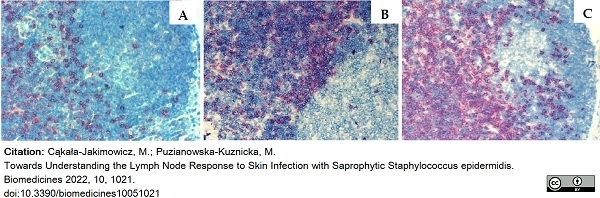
Cąkała-Jakimowicz, M. & Puzianowska-Kuznicka, M. (2022) Towards Understanding the Lymph Node Response to Skin Infection with Saprophytic Staphylococcus epidermidis.
Biomedicines. 10 (5): 1021. -
Du, K. et al. (2023) Pathogenesis of selective damage of granule cell layer in cerebellum of rats exposed to methylmercury
J Toxicol Sci 48 (7): 429-39. -
Santos Filho, L.E.D. et al. (2023) Dietary Soy Isoflavones Prevent Metabolic Disturbs Associated with a Deleterious Combination of Obesity and Menopause.
J Med Food. 26 (2): 104-13.
- RRID
- AB_324902
- UniProt
- P07725
- Entrez Gene
- Cd8a
- GO Terms
- GO:0016021 integral to membrane
- GO:0006954 inflammatory response
- GO:0019901 protein kinase binding
MCA48A647
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Rat ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up