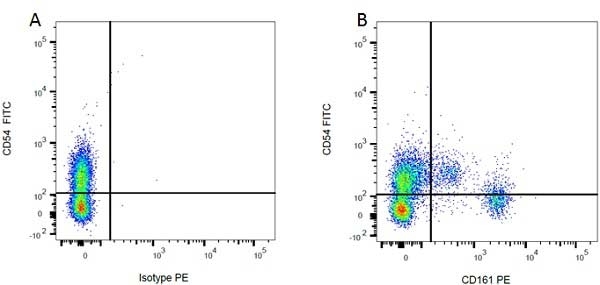
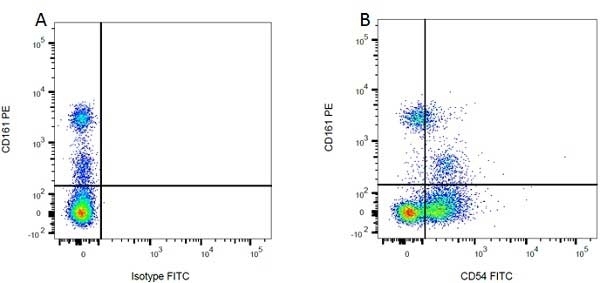
CD54 antibody | 1A29











Mouse anti Rat CD54:FITC
- Product Type
- Monoclonal Antibody
- Clone
- 1A29
- Isotype
- IgG1
- Specificity
- CD54
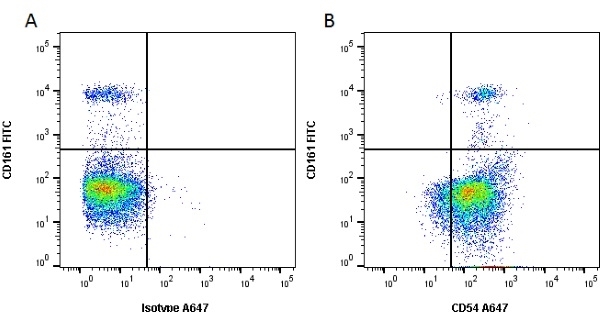
| Mouse anti Rat CD54 antibody, clone 1A29 recognizes the rat CD54 cell surface antigen, also known as intercellular adhesion molecule-1 (ICAM-1), a ~90 kDa adhesion molecule belonging to the immunoglobulin superfamily. CD54 is a cell surface ligand of the lymphocyte integrin, LFA-1 and plays an important role in various cell-cell interactions in the immune system. Cross-linking of ICAM-1 using clone 1A29 induces calcium signaling (Etienne et al. 1998). Mouse anti Rat CD54 antibody, clone 1A29 inhibits homotypic aggregation of cells including PHA blasts (Tamatani & Miyasaka 1990). |
- Target Species
- Rat
- Product Form
- Purified IgG conjugated to Fluorescein Isothiocyanate Isomer 1 (FITC) - liquid
- Preparation
- Purified IgG prepared by affinity chromatography on Protein A from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
0.09% Sodium Azide 1% Bovine Serum Albumin - Immunogen
- Rat Ax cells (a HEV derived cell line).
- Approx. Protein Concentrations
- IgG concentration 0.1 mg/ml
- Fusion Partners
- Spleen cells from immunised BALB/c mice were fused with cells from the PAI mouse myeloma cell line.
- Max Ex/Em
-
Fluorophore Excitation Max (nm) Emission Max (nm) FITC 490 525 - Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended. This product is photosensitive and should be protected from light.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Flow Cytometry | Neat |
- Flow Cytometry
- Use 10ul of the suggested working dilution to label 106 cells in 100ul.
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Mouse IgG1 Negative Control:FITC | MCA1209F | F | 0.1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse IgG1 Negative Control:FITC | ||||||
References for CD54 antibody
-
Etienne, S. et al. (1998) ICAM-1 signaling pathways associated with Rho activation in microvascular brain endothelial cells.
J Immunol. 161 (10): 5755-61. -
Kawai, T. et al. (1999) Selective diapedesis of Th1 cells induced by endothelial cell RANTES.
J Immunol. 163: 3269-78. -
Adamson, P. et al. (1999) Lymphocyte migration through brain endothelial cell monolayers involves signaling through endothelial ICAM-1 via a rho-dependent pathway.
J Immunol. 162: 2964-73. -
Sato, N. et al. (2000) Roles of ICAM-1 for abnormal leukocyte recruitment in the microcirculation of bleomycin-induced fibrotic lung injury.
Am J Respir Crit Care Med. 161: 1681-8. -
Etienne-Manneville, S. et al. (2000) ICAM-1-coupled cytoskeletal rearrangements and transendothelial lymphocyte migration involve intracellular calcium signaling in brain endothelial cell lines.
J Immunol. 165 (6): 3375-83. -
Arsenović-Ranin, N. et al. (2000) A monoclonal antibody to the rat Crry/p65 antigen, a complement regulatory membrane protein, stimulates adhesion and proliferation of thymocytes.
Immunology. 100: 334-44. -
Beck-Schimmer, B. et al. (2001) Hypoxia mediates increased neutrophil and macrophage adhesiveness to alveolar epithelial cells.
Am J Respir Cell Mol Biol. 25: 780-7. -
McKechnie, N. M. et al. (2002) Antigenic mimicry: Onchocerca volvulus antigen-specific T cells and ocular inflammation.
Invest Ophthalmol Vis Sci. 43:411-8.
View The Latest Product References
-
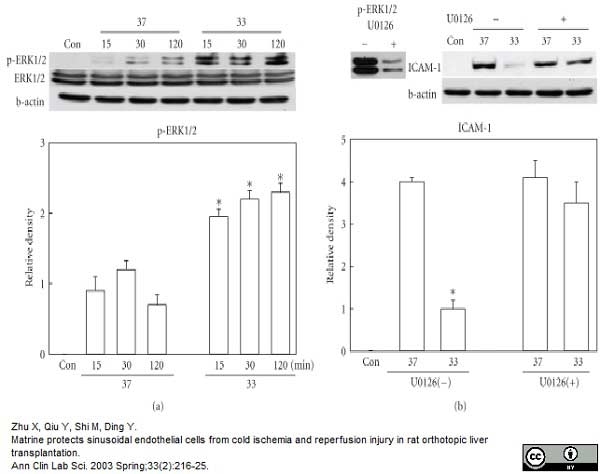
Zhu, X. et al. (2003) Matrine protects sinusoidal endothelial cells from cold ischemia and reperfusion injury in rat orthotopic liver transplantation.
Ann Clin Lab Sci. 33: 216-25. -
Deng, H. et al. (2003) Mild hypothermia inhibits inflammation after experimental stroke and brain inflammation.
Stroke. 34: 2495-501. -
Ikezumi, Y. et al. (2004) Macrophage-mediated renal injury is dependent on signaling via the JNK pathway.
J Am Soc Nephrol. 15: 1775-84. -
Westermann, J. et al. (2005) Naive, effector, and memory T lymphocytes efficiently scan dendritic cells in vivo: contact frequency in T cell zones of secondary lymphoid organs does not depend on LFA-1 expression and facilitates survival of effector T cells.
J Immunol. 174: 2517-24. -
Westermann, D. et al. (2007) Cardioprotective and anti-inflammatory effects of interleukin converting enzyme inhibition in experimental diabetic cardiomyopathy.
Diabetes. 56: 1834-41. -
Couty, J.P. (2007) PECAM-1 engagement counteracts ICAM-1-induced signaling in brain vascular endothelial cells.
J Neurochem. 103: 793-801. -
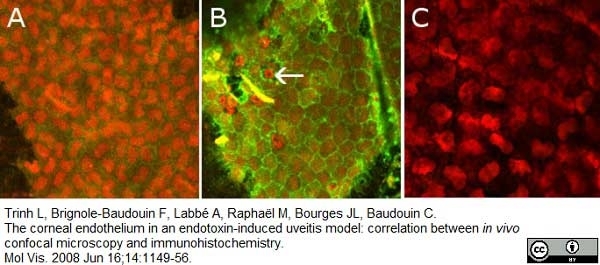
Trinh, L. et al. (2008) The corneal endothelium in an endotoxin-induced uveitis model: correlation between in vivo confocal microscopy and immunohistochemistry.
Mol Vis. 14: 1149-56. -
Kanellis, J. et al. (2010) JNK signalling in human and experimental renal ischaemia/reperfusion injury.
Nephrol Dial Transplant. 25: 2898-908. -
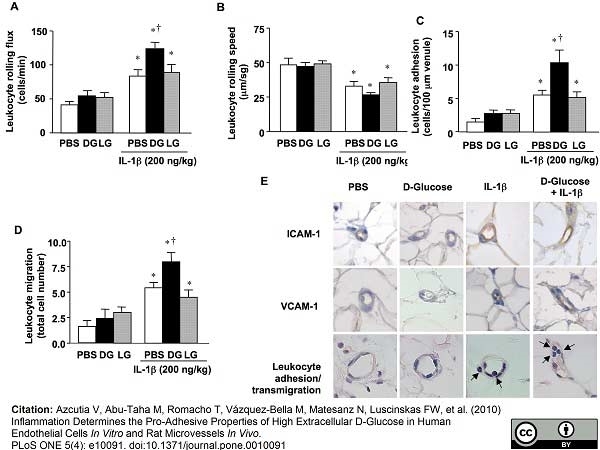
Azcutia V et al. (2010) Inflammation determines the pro-adhesive properties of high extracellular d-glucose in human endothelial cells in vitro and rat microvessels in vivo.
PLoS One. 5 (4): e10091. -
Choi, J.S. et al. (2011) Mild Hypothermia Attenuates Intercellular Adhesion Molecule-1 Induction via Activation of Extracellular Signal-Regulated Kinase-1/2 in a Focal Cerebral Ischemia Model.
Stroke Res Treat. 2011: 846716. -
Li, W. & Klein, S.L. (2012) Seoul virus-infected rat lung endothelial cells and alveolar macrophages differ in their ability to support virus replication and induce regulatory T cell phenotypes.
J Virol. 86 (21): 11845-55. -
Gates, D. et al. (2012) Apo J/clusterin expression and secretion: evidence for 15-deoxy-Δ(12,14)-PGJ(2)-dependent mechanism.
Biochim Biophys Acta. 1821 (2): 335-42. -
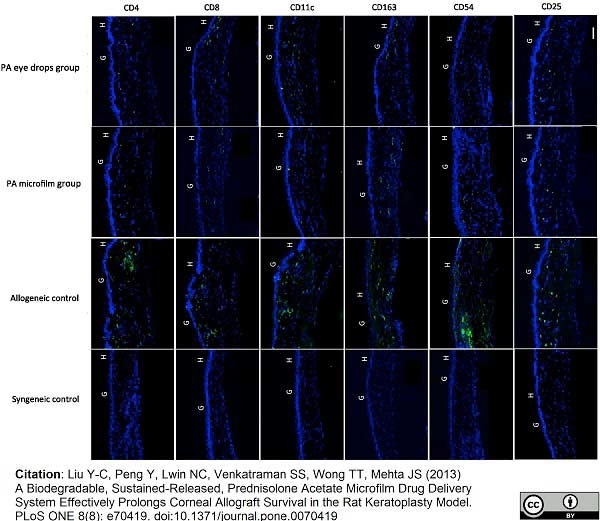
Liu, Y.C. et al. (2013) A biodegradable, sustained-released, prednisolone acetate microfilm drug delivery system effectively prolongs corneal allograft survival in the rat keratoplasty model.
PLoS One. 8 (8): e70419. -
Li, Z. et al. (2015) Three-dimensional graphene foams loaded with bone marrow derived mesenchymal stem cells promote skin wound healing with reduced scarring.
Mater Sci Eng C Mater Biol Appl. 57: 181-8. -
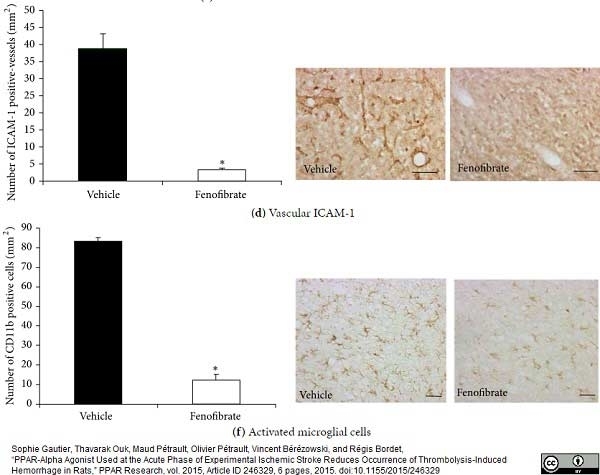
Gautier, S. et al. (2015) PPAR-Alpha Agonist Used at the Acute Phase of Experimental Ischemic Stroke Reduces Occurrence of Thrombolysis-Induced Hemorrhage in Rats.
PPAR Res. 2015: 246329. -
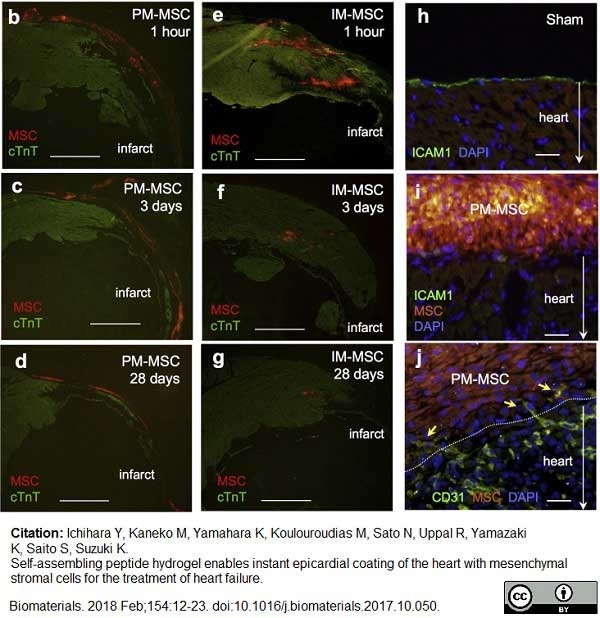
Ichihara, Y. et al. (2018) Self-assembling peptide hydrogel enables instant epicardial coating of the heart with mesenchymal stromal cells for the treatment of heart failure.
Biomaterials. 154: 12-23. -
Cąkała-Jakimowicz, M. & Puzianowska-Kuznicka, M. (2022) Towards Understanding the Lymph Node Response to Skin Infection with Saprophytic Staphylococcus epidermidis.
Biomedicines. 10 (5): 1021.
- Synonyms
- ICAM-1
- RRID
- AB_321790
- UniProt
- Q00238
- Entrez Gene
- Icam1
- GO Terms
- GO:0001541 ovarian follicle development
- GO:0001975 response to amphetamine
- GO:0016021 integral to membrane
- GO:0005178 integrin binding
- GO:0005615 extracellular space
- GO:0007569 cell aging
- GO:0008360 regulation of cell shape
- GO:0042493 response to drug
- GO:0010212 response to ionizing radiation
- View More GO Terms
- GO:0010477 response to sulfur dioxide
- GO:0014070 response to organic cyclic compound
- GO:0031669 cellular response to nutrient levels
- GO:0034698 response to gonadotropin stimulus
- GO:0043200 response to amino acid stimulus
- GO:0043498 cell surface binding
- GO:0046688 response to copper ion
- GO:0045429 positive regulation of nitric oxide biosynthetic process
- GO:0045471 response to ethanol
- GO:0045907 positive regulation of vasoconstriction
- GO:0050900 leukocyte migration
- GO:0051092 positive regulation of NF-kappaB transcription factor activity
- GO:0051926 negative regulation of calcium ion transport
- GO:0071222 cellular response to lipopolysaccharide
- GO:0071312 cellular response to alkaloid
- GO:0071333 cellular response to glucose stimulus
- GO:0071347 cellular response to interleukin-1
- GO:0071356 cellular response to tumor necrosis factor
- GO:0071456 cellular response to hypoxia
MCA773F
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Rat ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up