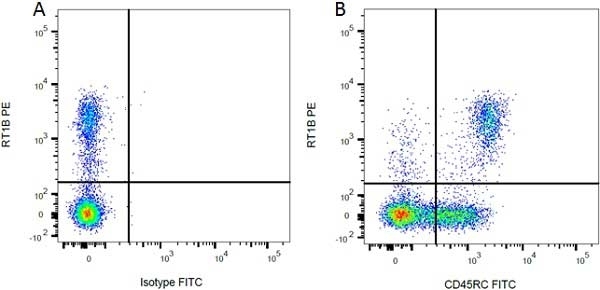
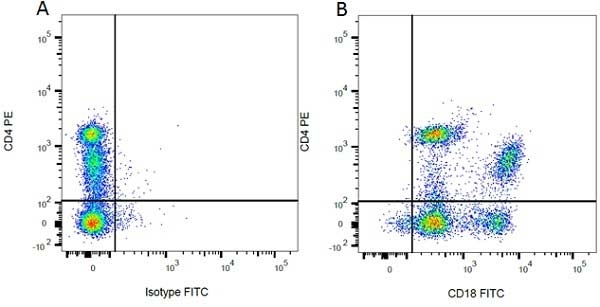
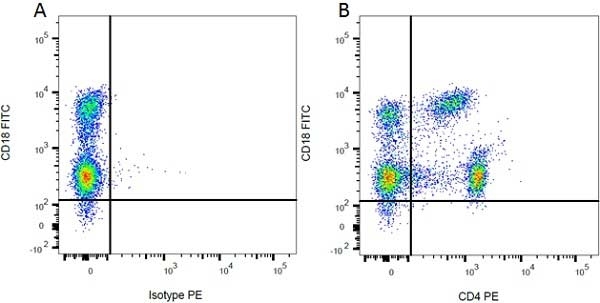
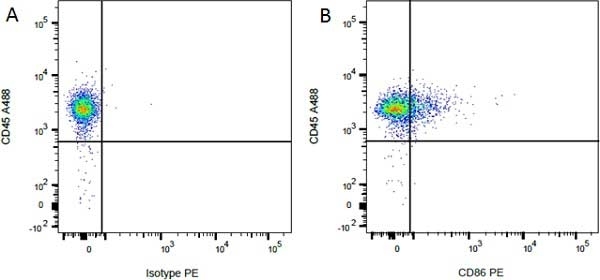
Mouse IgG1 Negative Control antibody
























Mouse IgG1 Negative Control:FITC
- Product Type
- Negative/Isotype Control
- Isotype
- IgG1
- Specificity
- Mouse IgG1 Negative Control
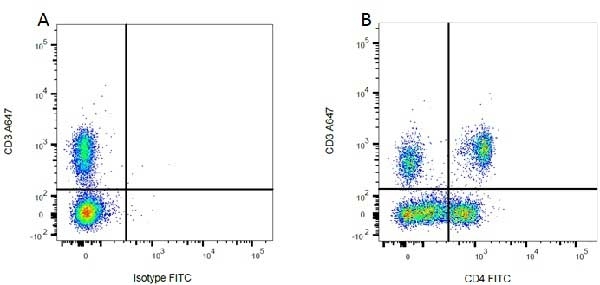
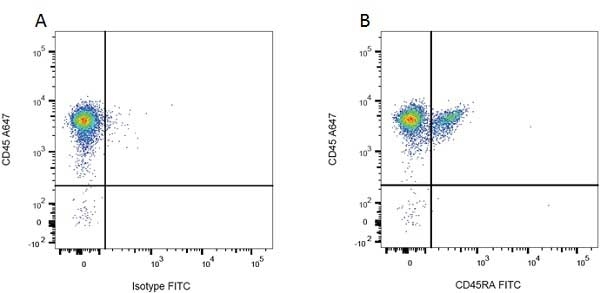
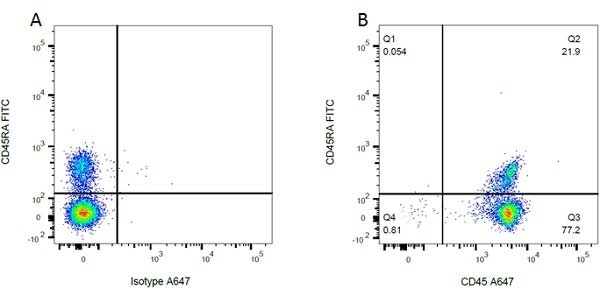
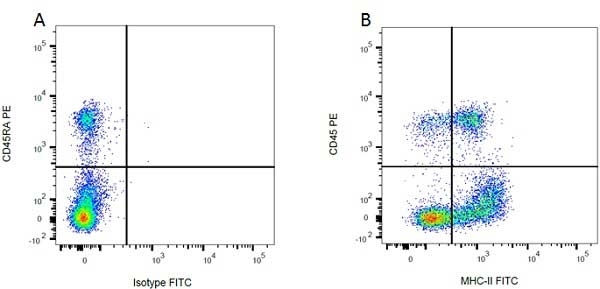
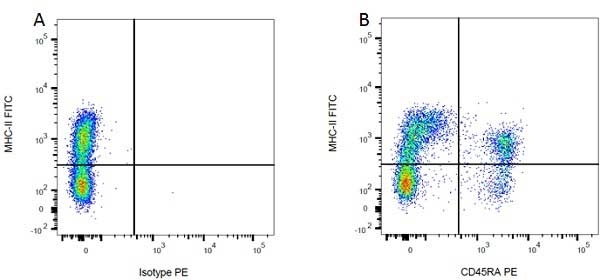
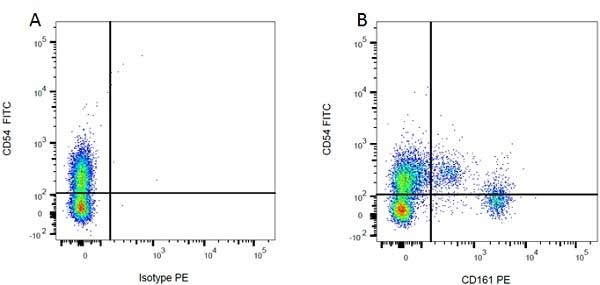
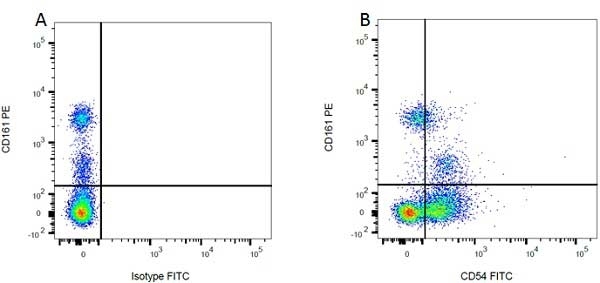
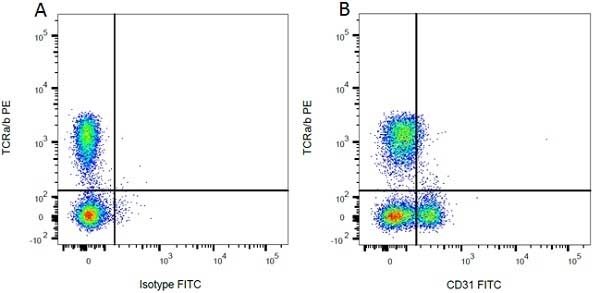
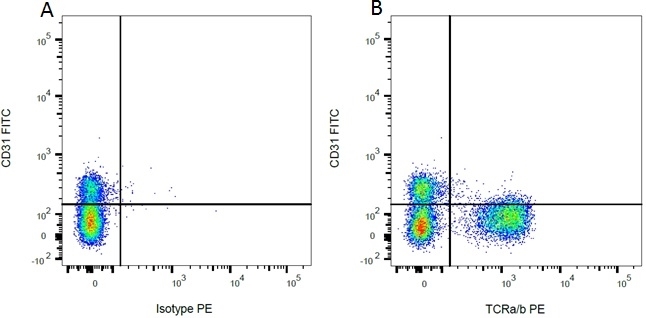
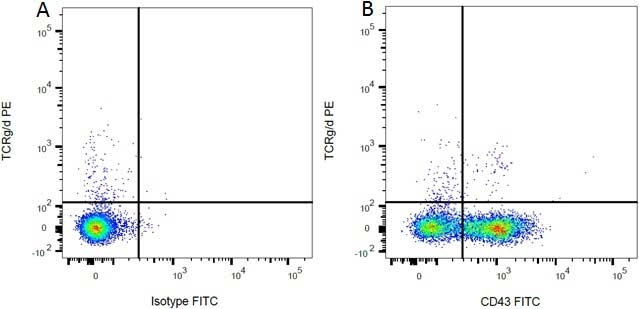
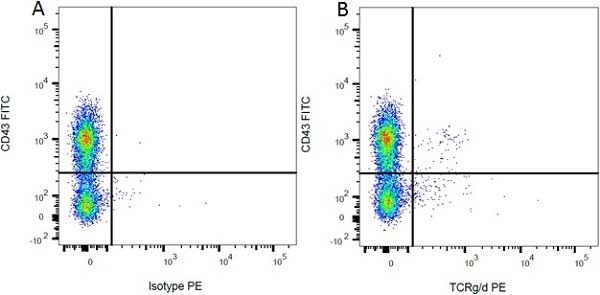
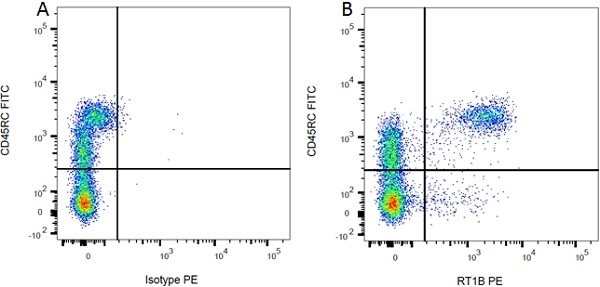
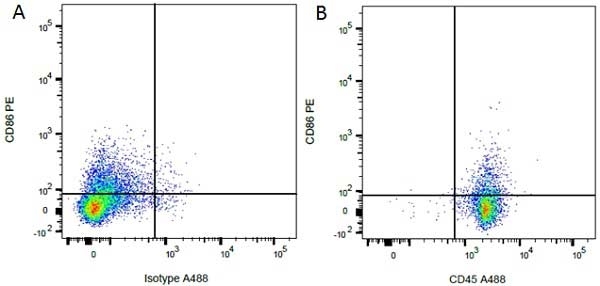
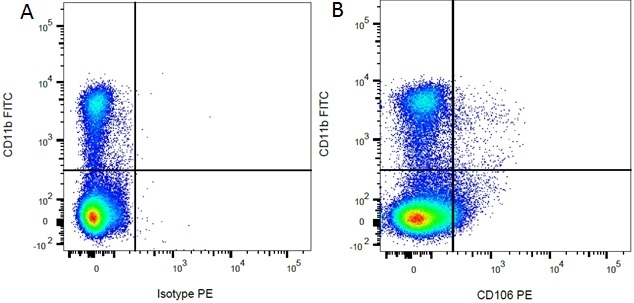
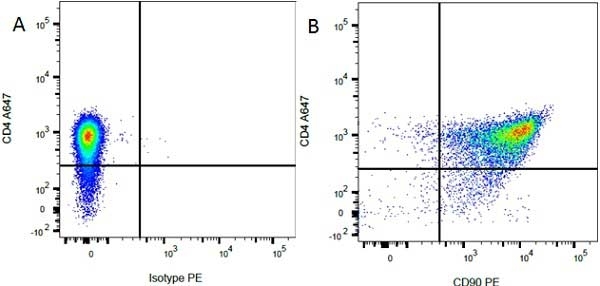
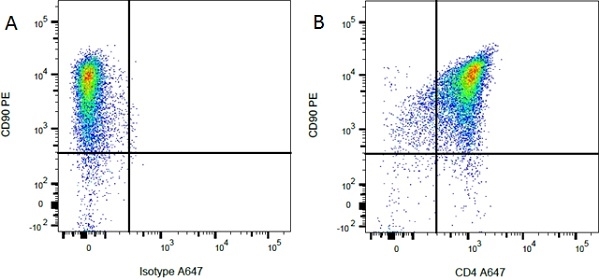
| Mouse IgG1 Negative Control antibody is suitable for use as a negative control to assess non-specific binding of mouse IgG1 antibodies to target cells. Mouse IgG1 Negative Control antibody has been tested and found to be negative on the following rat cell types, peripheral blood leucocytes, thymocytes, splenocytes and macrophages. Clone F8-11-13 recognizes the human CD45RA antigen, and therefore human leucocytes may be used as a positive control for this product. NOT SUITABLE FOR USE AS A NEGATIVE CONTROL ON HUMAN TISSUES |
- Target Species
- Negative Control
- Product Form
- Purified IgG conjugated to Fluorescein Isothiocyanate Isomer 1 (FITC) - liquid
- Preparation
- Purified IgG prepared by affinity chromatography on Protein A from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- 0.09% sodium azide (NaN3)
1% bovine serum albumin - Immunogen
- Human T lymphocytes.
- Approx. Protein Concentrations
- IgG concentration 0.1 mg/ml
- Fusion Partners
- Spleen cells from immunized BALB/c mice were fused with cells of the NS1 mouse myeloma cell line.
- Max Ex/Em
-
Fluorophore Excitation Max (nm) Emission Max (nm) FITC 490 525 - Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended. This product is photosensitive and should be protected from light.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Flow Cytometry | * |
- Flow Cytometry
- Use 10μl of the suggested working dilution to label 106 cells in 100μl
References for Mouse IgG1 Negative Control antibody
-
Weiss, D.J. et al. (2008) Bovine monocyte TLR2 receptors differentially regulate the intracellular fate of Mycobacterium avium subsp. paratuberculosis and Mycobacterium avium subsp. avium.
J Leukoc Biol. 83 (1): 48-55. -
Chen, W. et al. (2009) Expression of toll-like receptor 4 in uvea-resident tissue macrophages during endotoxin-induced uveitis.
Mol Vis. 15: 619-28. -
Safeukui I et al. (2015) Malaria induces anemia through CD8+ T cell-dependent parasite clearance and erythrocyte removal in the spleen.
MBio. 6 (1) pii: e02493-14. -
Aricha, R. et al. (2016) Suppression of experimental autoimmune myasthenia gravis by autologous T regulatory cells.
J Autoimmun. 67: 57-64. -
Wattegedera, S.R. et al. (2017) Enhancing the toolbox to study IL-17A in cattle and sheep.
Vet Res. 48 (1): 20. -
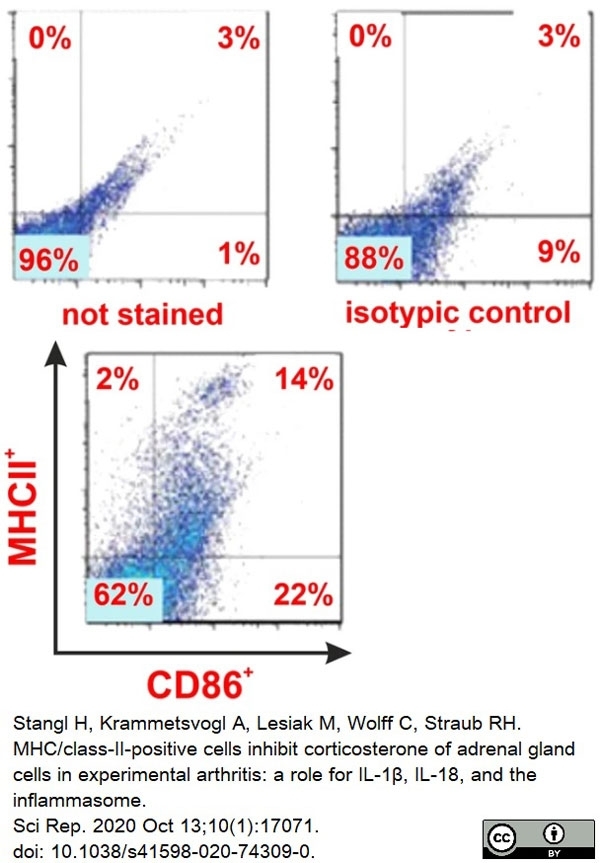
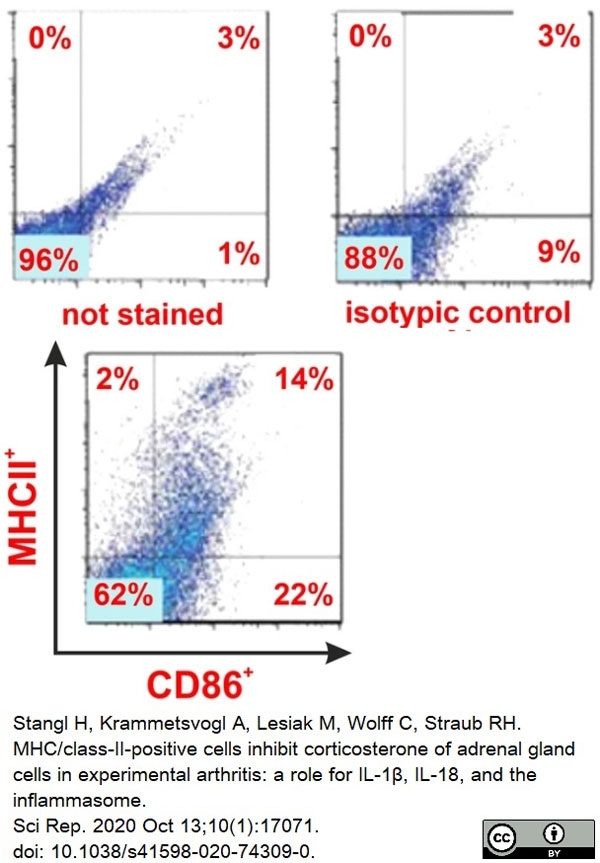
Stangl, H. et al. (2020) MHC/class-II-positive cells inhibit corticosterone of adrenal gland cells in experimental arthritis: a role for IL-1β, IL-18, and the inflammasome.
Sci Rep. 10 (1): 17071. -
Terpeluk, R.E. et al. (2024) Supplementation of Foals with a Saccharomyces cerevisiae Fermentation Product Alters the Early Response to Vaccination
Animals. 14 (6): 960.
- RRID
- AB_567337
MCA1209F
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Negative Control ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up