IgA antibody | K61 1B4



Mouse anti Pig IgA
- Product Type
- Monoclonal Antibody
- Clone
- K61 1B4
- Isotype
- IgG1
- Specificity
- IgA
| Mouse Anti Pig IgA antibody, clone K61 1B4 recognizes porcine immunoglobulin (Ig) alpha chain. No cross-reactivity is observed with either porcine IgM or IgG. It has been suggested that pigs possess two subclasses of IgA, referred to as either IgA1 and IgA2 similar to the human IgA subclasses. More recent research indicates that these are allotypic variants and described as IgAa and IgAb (Navarro et al. 2000). Research undertaken in connection with porcine IgA response to Foot and Mouth disease (Pacheco et al. 2010) has demonstrated that clone K61 1B4 recognises both IgAa and IgAb allotypes. IgA is the dominant immunoglobulin found in surface secretions where its role is in the protection of body surfaces. High levels of IgA may be detected in tracheal secretions, saliva, intestinal fluid and urogenital tract secretions. The primary function of IgA is to prevent adherence of bacteria and virus to epithelial surfaces. IgA may also act within epithelial cells where it has a role in interrupting viral replication. |
- Target Species
- Pig
- Product Form
- Purified IgG - liquid
- Preparation
- Purified IgG prepared by affinity chromatography on Protein A from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- 0.09% Sodium Azide (NaN3)
- Carrier Free
- Yes
- Immunogen
- Porcine IgA
- Approx. Protein Concentrations
- IgG concentration 1.0 mg/ml
- Fusion Partners
- Spleen cells from immunised mice were fused with cells of the mouse P3-X63-Ag8.653 myeloma cell line
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
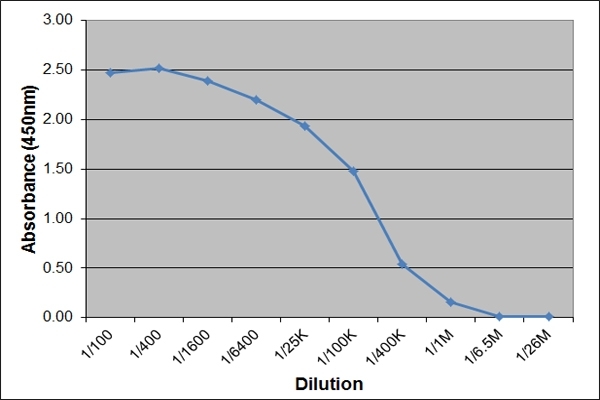
| ELISA | 1/50K | 1/500K | |
| Flow Cytometry | |||
| Immunohistology - Frozen |
- ELISA
- This product may be used as a detection reagent in ELISA applications
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Goat anti Mouse IgG (H/L):HRP (Multi Species Adsorbed) | STAR117P | C E WB | 0.5 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Goat anti Mouse IgG (H/L):HRP (Multi Species Adsorbed) | ||||||
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Mouse IgG1 Negative Control | MCA928 | F | 100 Tests |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse IgG1 Negative Control | ||||||
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Mouse anti Pig Ig Lambda Light Chain | MCA633GA | C E F P | 0.1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse anti Pig Ig Lambda Light Chain | ||||||
| Mouse anti Pig IgA Secretory Component | MCA634GA | E | 0.1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse anti Pig IgA Secretory Component | ||||||
| Mouse anti Pig IgG1 | MCA635GA | E | 0.1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse anti Pig IgG1 | ||||||
| Mouse anti Pig IgG2 | MCA636GA | E | 0.1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse anti Pig IgG2 | ||||||
| Mouse anti Pig IgM | MCA637GA | C E F | 0.1 mg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse anti Pig IgM | ||||||
References for IgA antibody
-
Leitão, A. et al. (2001) The non-haemadsorbing African swine fever virus isolate ASFV/NH/P68 provides a model for defining the protective anti-virus immune response.
J Gen Virol. 82 (Pt 3): 513-23. -
Bourges, D. et al. (2004) T and IgA B lymphocytes of the pharyngeal and palatine tonsils: differential expression of adhesion molecules and chemokines.
Scand J Immunol. 60 (4): 338-50. -
Nejsum, P. et al. (2009) Population dynamics of Trichuris suis in trickle-infected pigs.
Parasitology. 136 (6): 691-7. -
Pacheco, J.M. et al. (2010) IgA antibody response of swine to foot-and-mouth disease virus infection and vaccination.
Clin Vaccine Immunol. 17: 550-8. -
Inman CF et al. (2012) Neonatal colonisation expands a specific intestinal antigen-presenting cell subset prior to CD4 T-cell expansion, without altering T-cell repertoire.
PLoS One. 7 (3): e33707. -
Ewaschuk JB et al. (2012) Barley-derived β-glucans increases gut permeability, ex vivo epithelial cell binding toE. coli, and naive T-cell proportions in weanling pigs.
J Anim Sci. 90 (8): 2652-62. -
Kandasamy, S. et al. (2014) Lactobacilli and Bifidobacteria enhance mucosal B cell responses and differentially modulate systemic antibody responses to an oral human rotavirus vaccine in a neonatal gnotobiotic pig disease model.
Gut Microbes. 5 (5): 639-51. -
Kandasamy, S. et al. (2014) Prenatal vitamin A deficiency impairs adaptive immune responses to pentavalent rotavirus vaccine (RotaTeq®) in a neonatal gnotobiotic pig model.
Vaccine. 32 (7): 816-24.
View The Latest Product References
-
Kringel H et al. (2015) Serum antibody responses in pigs trickle-infected with Ascaris and Trichuris: Heritabilities and associations with parasitological findings.
Vet Parasitol. 211 (3-4): 306-11. -
Guzman-Bautista, E.R. et al. (2015) Tracheal and bronchial polymeric immunoglobulin secretory immune system (PISIS) development in a porcine model.
Dev Comp Immunol. 53 (2): 271-82. -
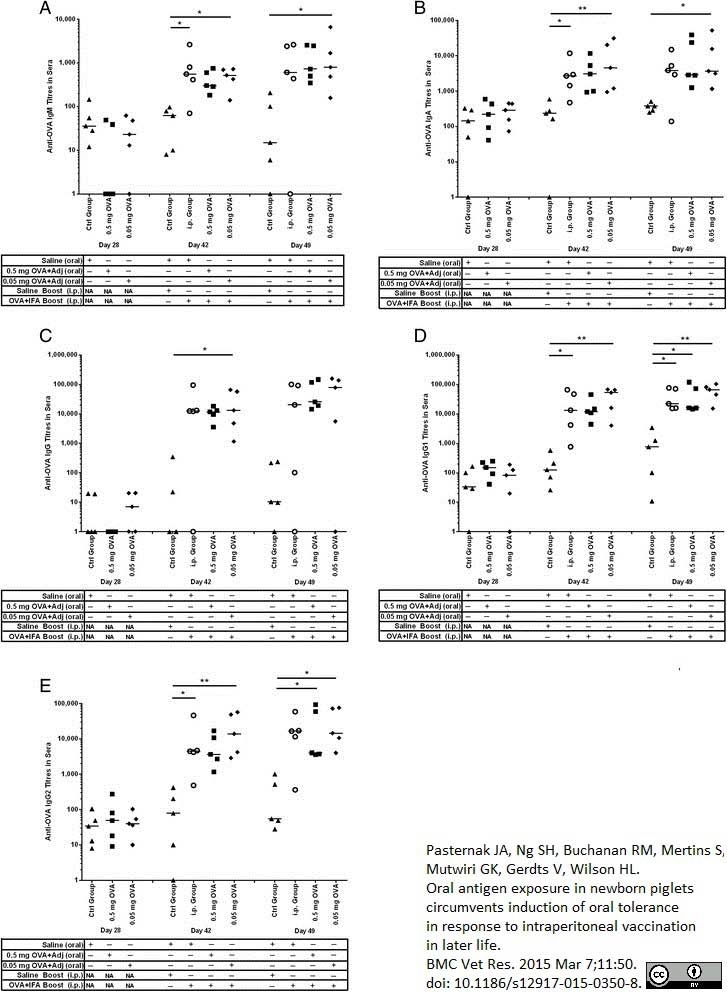
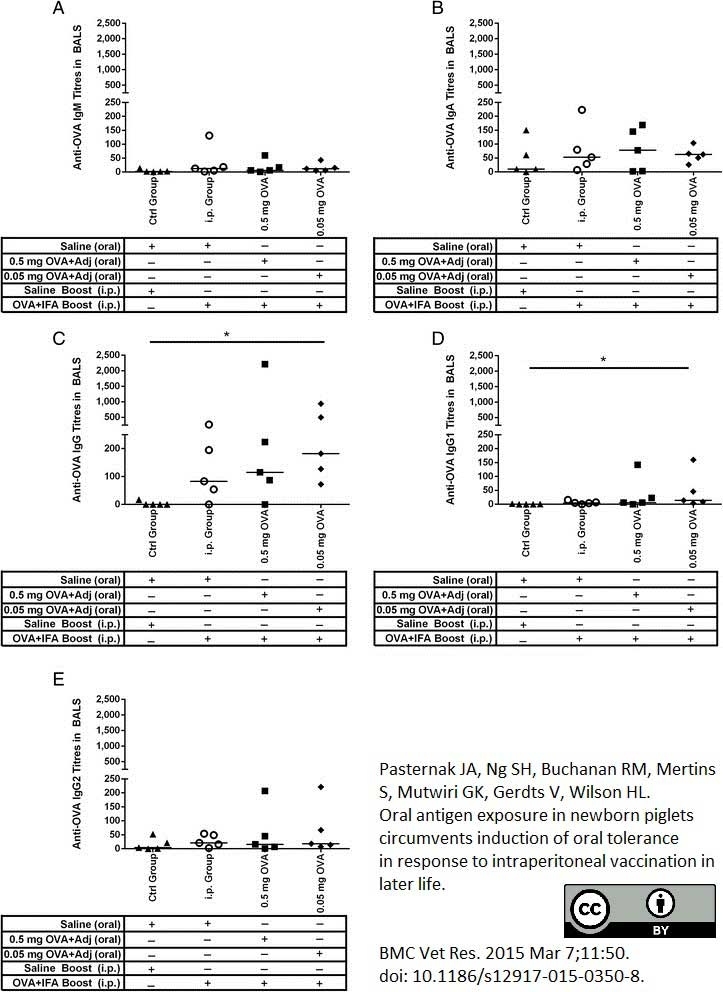
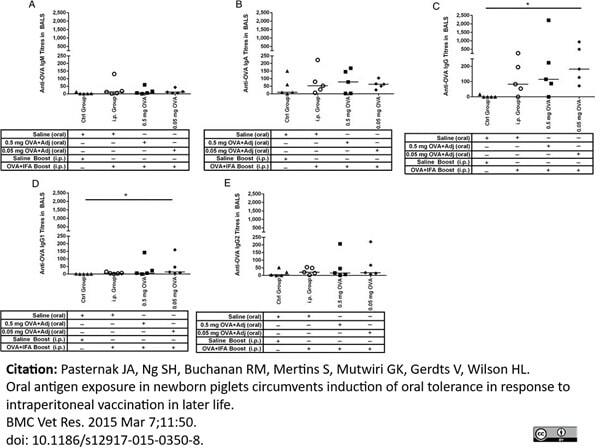
Pasternak, J.A. et al. (2015) Oral antigen exposure in newborn piglets circumvents induction of oral tolerance in response to intraperitoneal vaccination in later life.
BMC Vet Res. 11 (1): 350. -
Makadiya, N. et al. (2016) S1 domain of the porcine epidemic diarrhea virus spike protein as a vaccine antigen.
Virol J. 13: 57. -
Aubrey, L. et al. (2022) A Bivalent Live Attenuated Influenza Virus Vaccine Protects against Drifted H1N2 and H3N2 Clinical Isolates in Swine
Viruses. 15 (1): 46.
Further Reading
-
Navarro, P. et al. (2000) Porcine IgA allotypes are not equally transcribed or expressed in heterozygous swine.
Mol Immunol. 37: 653-64.
MCA638GA
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Pig ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up