c-Myc antibody | 9E10






Mouse anti c-Myc:Biotin
- Product Type
- Monoclonal Antibody
- Clone
- 9E10
- Isotype
- IgG1
- Specificity
- c-Myc
| Mouse anti c-myc antibody, clone 9E10 detects the p62c-myc proto-oncogene protein, which is involved in the regulation of the cell cycle and cell growth. C-myc is primarily located to the cell nucleus, but has also been shown to localized to the cytoplasm in several cell lines (Craig et al. 1993). Overexpression of c-myc has been reported in a wide variety of human cancers ( Mouse anti c-myc antibody, clone 9E10 recognizes the sequence EQKLISEEDL and may be used to detect proteins and peptides labelled with molecular tags containing this sequence ( |
- Target Species
- Human
- Species Cross-Reactivity
-
Target Species Cross Reactivity Epitope tag - N.B. Antibody reactivity and working conditions may vary between species.
- Product Form
- Purified IgG conjugated to biotin - liquid
- Preparation
- Purified IgG prepared by affinity chromatography on Protein G from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- 0.09% sodium azide (NaN3)
1% bovine serum albumin - Immunogen
- Synthetic peptide sequence corresponding to the C-terminal region (residues 408-439) of human c-myc conjugated to keyhole limpet hemocyanin.
- Approx. Protein Concentrations
- IgG concentration 0.1 mg/ml
- Fusion Partners
- Spleen cells from immunised BALB/c mice were fused with cells of the SP2/0 myeloma cell line.
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
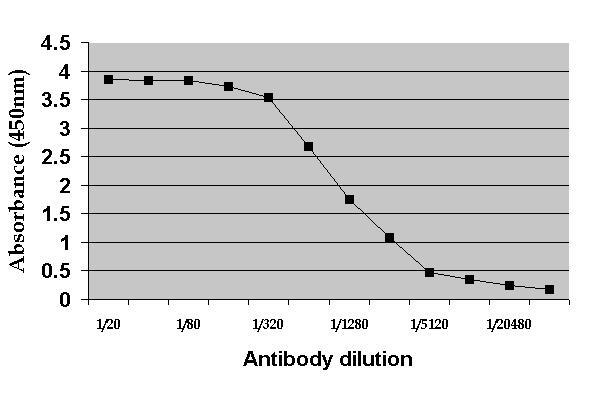
| ELISA | 1/20 | 1/50 | |
| Immunohistology - Frozen | |||
| Immunohistology - Paraffin | |||
| Western Blotting 1 |
- 1 9E10 recognizes c-myc under non-reducing conditions
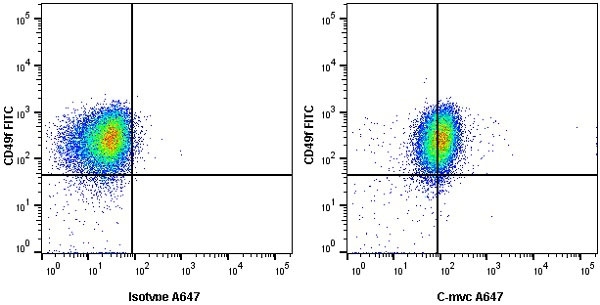
- Flow Cytometry
- Use 10μl of the suggested working dilution to label 1x106 cells in 100μl
References for c-Myc antibody
-
Evan, G.I. et al. (1985) Isolation of monoclonal antibodies specific for human c-myc proto-oncogene product.
Mol Cell Biol. 5 (12): 3610-6. -
Spandidos, D.A. et al. (1987) Elevated expression of the myc gene in human benign and malignant breast lesions compared to normal tissue.
Anticancer Res. 7 (6): 1299-304. -
Borodina, I. et al. (2010) Display of wasp venom allergens on the cell surface of Saccharomyces cerevisiae.
Microb Cell Fact. 9: 74. -
Groeger, G. et al. (2007) Co-operative Cdc42 and Rho signalling mediates ephrinB-triggered endothelial cell retraction.
Biochem J. 404: 23-9. -
Hilpert, K. et al. (2001) Anti-c-myc antibody 9E10: epitope key positions and variability characterized using peptide spot synthesis on cellulose.
Protein Eng. 14: 803-6. -
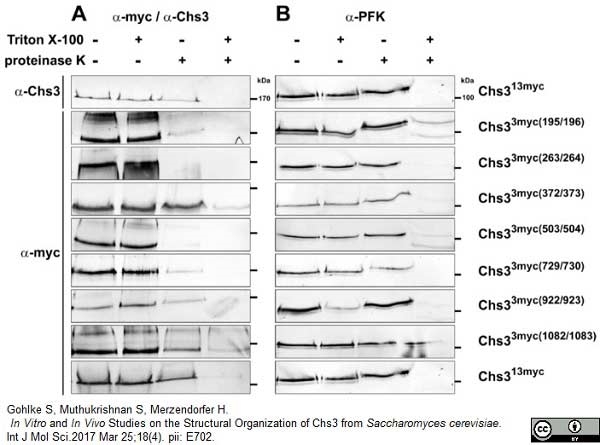
Gohlke, S. et al. (2017) In Vitro and In Vivo Studies on the Structural Organization of Chs3 from Saccharomyces cerevisiae.
Int J Mol Sci. 18 (4): pii: E702. -
Gray, P. et al. (2010) Identification of a novel human MD-2 splice variant that negatively regulates Lipopolysaccharide-induced TLR4 signaling.
J Immunol. 184: 6359-66. -
Duriseti, S. et al. (2010) Antagonistic anti-urokinase plasminogen activator receptor (uPAR) antibodies significantly inhibit uPAR-mediated cellular signaling and migration.
J Biol Chem. 285: 26878-88.
View The Latest Product References
-
Tan, P.H. et al. (2005) Creation of tolerogenic human dendritic cells via intracellular CTLA4: a novel strategy with potential in clinical immunosuppression.
Blood. 106: 2936-43. -
Wallace, S.W. et al. (2010) Cdc42 regulates apical junction formation in human bronchial epithelial cells through PAK4 and Par6B.
Mol Biol Cell. 21: 2996-3006. -
Rowshanravan, B. et al. (2014) RasGAP mediates neuronal survival in Drosophila through direct regulation of Rab5-dependent endocytosis.
J Cell Sci. 127: 2849-61. -
Taylor K et al. (2015) Nanocell targeting using engineered bispecific antibodies.
MAbs. 7 (1): 53-65. -
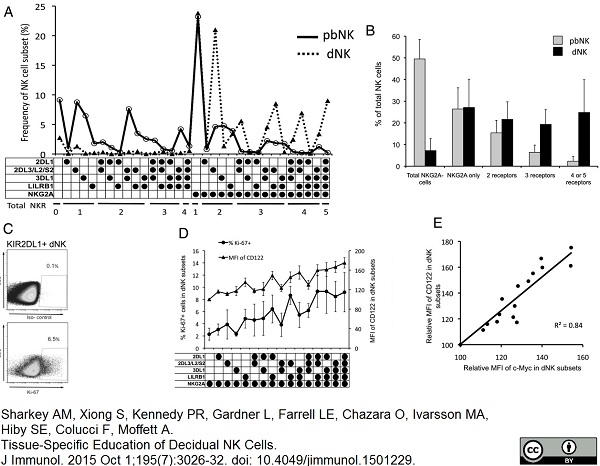
Sharkey, A.M. et al. (2015) Tissue-Specific Education of Decidual NK Cells.
J Immunol. 195 (7): 3026-32. -
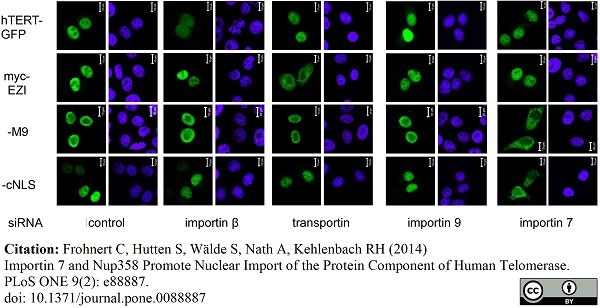
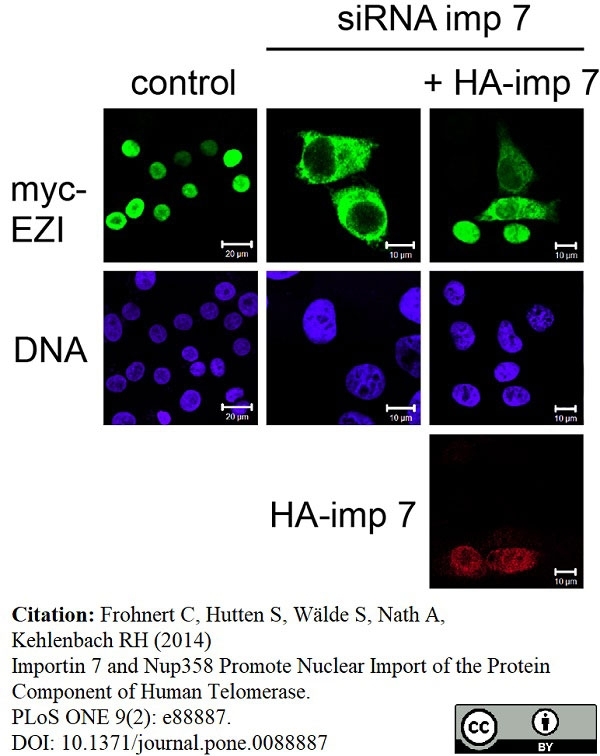
Frohnert, C. et al. (2014) Importin 7 and Nup358 promote nuclear import of the protein component of human telomerase.
PLoS One. 9 (2): e88887. -
Hage, N. et al. (2015) Improved expression and purification of the Helicobacter pylori adhesin BabA through the incorporation of a hexa-lysine tag.
Protein Expr Purif. 106: 25-30. -
Mann, J.K. & Park, S. (2015) Epitope-Specific Binder Design by Yeast Surface Display.
Methods Mol Biol. 1319: 143-54. -
Paraskevopoulou, V. et al. (2019) Introduction of a C-terminal hexa-lysine tag increases thermal stability of the LacDiNac binding adhesin (LabA) exodomain from Helicobacter pylori.
Protein Expr Purif. 163: 105446. -
Lim, H.K. et al. (2010) Flow cytometric analysis of genetic FRET detectors containing variable substrate sequences.
Biotechnol Prog. 26 (6): 1765-71. -
Walker, L.M. et al. (2009) Efficient recovery of high-affinity antibodies from a single-chain Fab yeast display library.
J Mol Biol. 389 (2): 365-75. -
Matos, J. et al. (2013) Cell-cycle kinases coordinate the resolution of recombination intermediates with chromosome segregation.
Cell Rep. 4 (1): 76-86. -
Paraskevopoulou, V. et al. (2020) Structural and binding characterization of the LacdiNAc-specific adhesin (LabA; HopD) exodomain from Helicobacter pylori.
Curr Res Struct Biol. 15 Dec [Epub ahead of print]. -
Kalusche, S. et al. (2020) Lactobacilli Expressing Broadly Neutralizing Nanobodies against HIV-1 as Potential Vectors for HIV-1 Prophylaxis?
Vaccines (Basel). 8 (4) Dec 13 [Epub ahead of print]. -
Hollandsworth, H.M. et al. (2020) Fluorophore-conjugated Helicobacter pylori recombinant membrane protein (HopQ) labels primary colon cancer and metastases in orthotopic mouse models by binding CEA-related cell adhesion molecules.
Transl Oncol. 13 (12): 100857. -
Paraskevopoulou, V. et al. (2021) Structural and binding characterization of the LacdiNAc-specific adhesin (LabA; HopD) exodomain from Helicobacter pylori.
Curr Res Struct Biol. 3: 19-29. -
Low, S. et al. (2020) VHH antibody targeting the chemokine receptor CX3CR1 inhibits progression of atherosclerosis.
MAbs. 12 (1): 1709322.
Further Reading
-
Nesbit, C. et al. (1999) MYC oncogenes and human neoplastic disease.
Oncogene. 18: 3004-16. -
Krauß, N. et al. (2008) The structure of the anti-c-myc antibody 9E10 Fab fragment/epitope peptide complex reveals a novel binding mode dominated by the heavy chain hypervariable loops.
Proteins. 73: 552-65.
- RRID
- AB_323950
- UniProt
- P01106
- Entrez Gene
- MYC
- GO Terms
- GO:0005515 protein binding
- GO:0001658 branching involved in ureteric bud morphogenesis
- GO:0003700 sequence-specific DNA binding transcription factor activity
- GO:0005654 nucleoplasm
- GO:0005730 nucleolus
- GO:0006357 regulation of transcription from RNA polymerase II promoter
- GO:0006879 cellular iron ion homeostasis
- GO:0007050 cell cycle arrest
- GO:0008283 cell proliferation
- View More GO Terms
- GO:0042493 response to drug
- GO:0016563 transcription activator activity
- GO:0032204 regulation of telomere maintenance
- GO:0070888 E-box binding
- GO:0090096 positive regulation of metanephric cap mesenchymal cell proliferation
MCA2200B
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Human ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up