CD3 antibody | CA17.2A12











Mouse anti Dog CD3:FITC
- Product Type
- Monoclonal Antibody
- Clone
- CA17.2A12
- Isotype
- IgG1
- Specificity
- CD3
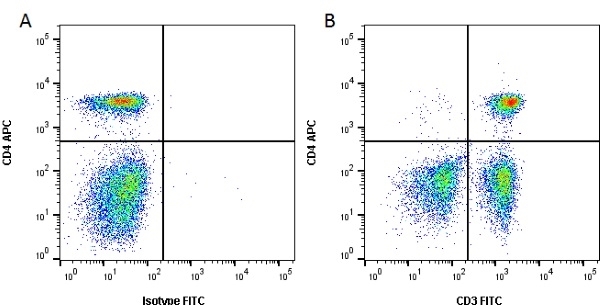
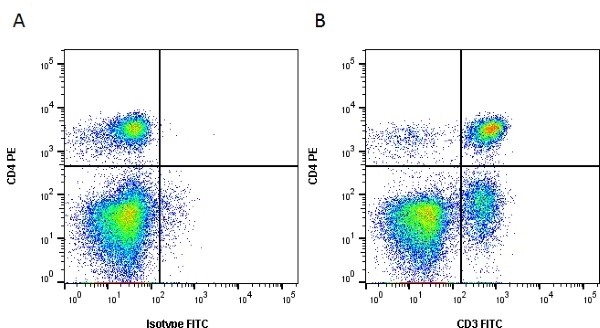
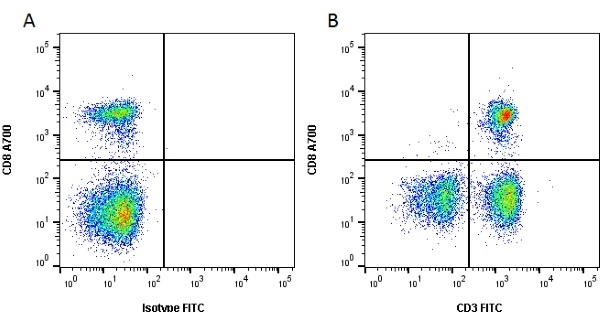
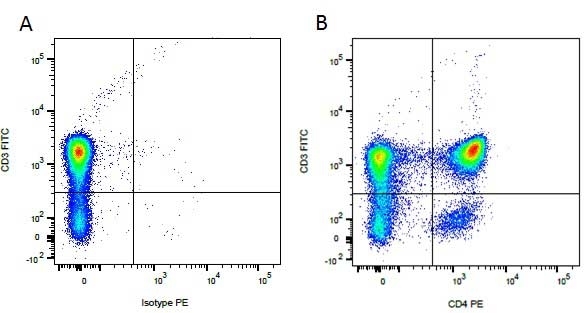
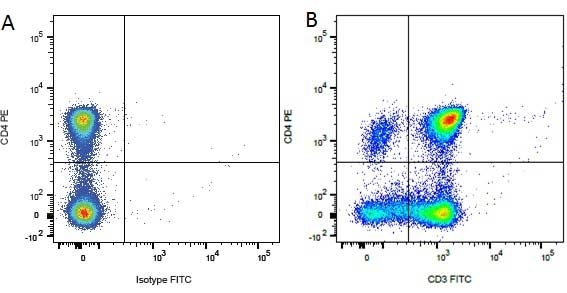
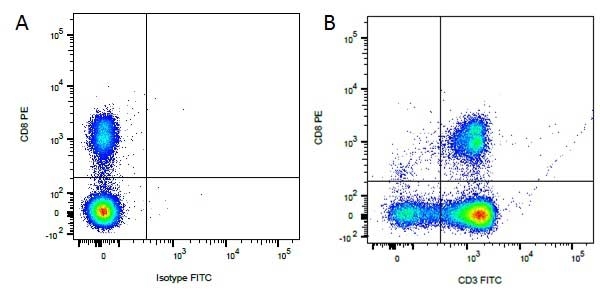
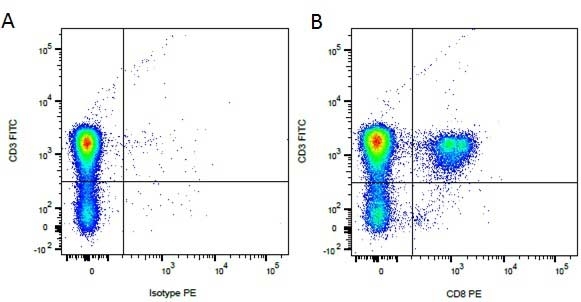
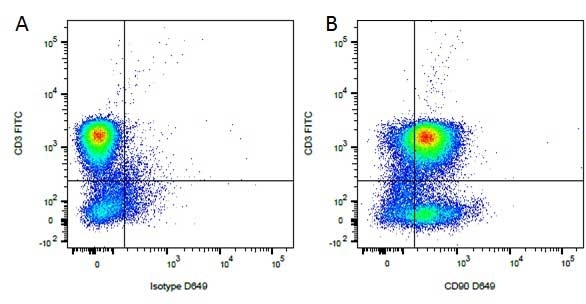
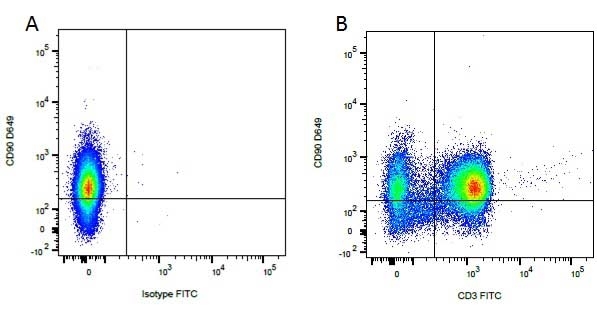
| Mouse anti Dog CD3 antibody, clone CA17.2A12 recognizes the canine CD3 cell surface antigen, expressed by thymocytes and mature T lymphocytes. CD3 is engaged in the surface expression of the T-cell antigen receptor (TCR) and the signal transduction pathway resulting from MHC ligand binding to the TCR. CD3 is made up of a number of invariant subchains of the immunoglobulin superfamily. Mouse anti Dog CD3 clone CA17.2A12 is a valuable flow cytometric and immunohistologic tool for canine lymphoma detection of T-cell origin (Miniscalco et al. 2003). |
- Target Species
- Dog
- Product Form
- Purified IgG conjugated to Fluorescein Isothiocyanate Isomer 1 (FITC) - liquid
- Preparation
- Purified IgG prepared by affinity chromatography on Protein G from tissue culture supernatant
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- 0.09% sodium azide (NaN3)
1% bovine serum albumin - Immunogen
- Affinity enriched TCR/CD3 membrane proteins isolated from thymocytes and the T cell line CLGL-90
- Approx. Protein Concentrations
- IgG concentration 0.1 mg/ml
- Max Ex/Em
-
Fluorophore Excitation Max (nm) Emission Max (nm) FITC 490 525 - Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended. This product is photosensitive and should be protected from light.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| Flow Cytometry | Neat | 1/10 |
- Flow Cytometry
- Use 10μl of the suggested working dilution to label 106 cells or 100μl whole blood.
N.B.. MCA1774F should not be used with MCA1781PE (mouse anti canine B-cells), in dual colour flow cytometry, due to non-specific interactions between the two reagents.
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Mouse IgG1 Negative Control:FITC | MCA928F | F | 100 Tests |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse IgG1 Negative Control:FITC | ||||||
References for CD3 antibody
-
Moore, P.F. and Rossitto, P.V. (1993) Development of monoclonal antibodies to canine T cell receptor complex (TCR/CD3) and their utilisation in the diagnosis of T cell neoplasia.
Vet. Pathol. 30: 457. Abstract 117. -
Vernau, W and Moore, P. F. (1999) An immunophenotypic study of canine leukemias and preliminary assessment of clonality by polymerase chain reaction.
Vet Immunol Immunopathol. 69:145-64. -
Moreno, J. et al (1999) The immune response and PBMC subsets in canine visceral leishmaniasis before, and after, chemotherapy.
Vet Immunol Immunopathol. 71:181-95. -
McDonough, S. P. and Moore, P. F. (2000) Clinical, hematologic, and immunophenotypic characterization of canine large granular lymphocytosis.
Vet Pathol. 37:637-46. -
Byrne, K. et al (2000) A standardized gating technique for the generation of flow cytometry data for normal canine and normal feline blood lymphocytes.
Vet Immunol Immunopathol. 73:167-82. -
Out, T.A. et al. (2002) Local T-cell activation after segmental allergen challenge in the lungs of allergic dogs.
Immunology. 105: 499-508. -
Zentek, J. et al. (2002) Morphology and immunopathology of the small and large intestine in dogs with nonspecific dietary sensitivity.
J Nutr. 132: 1652S-4S. -
Hsiao, Y.W. et al (2004) Tumor-infiltrating lymphocyte secretion of IL-6 antagonizes tumor-derived TGF-beta 1 and restores the lymphokine-activated killing activity.
J Immunol. 172: 1508-14.
View The Latest Product References
-
Moore, P.F. et al. (2006) Canine hemophagocytic histiocytic sarcoma: a proliferative disorder of CD11d+ macrophages.
Vet Pathol. 43 (5): 632-45. -
Ting-De Ravin, S.S. et al. (2006) Correction of canine X-linked severe combined immunodeficiency by in vivo retroviral gene therapy.
Blood. 107: 3091-7. -
Miranda, S. et al. (2007) Characterization of circulating lymphocyte subpopulations in canine leishmaniasis throughout treatment with antimonials and allopurinol.
Vet Parasitol. 144: 251-60. -
Altmann, S. et al. (2008) High Mobility Group Box 1-Protein expression in canine haematopoietic cells and influence on canine peripheral blood mononuclear cell proliferative activity
Vet Immunol Immunopathol. 126: 367-72. -
Huang, Y.C. et al. (2008) CD5-low expression lymphocytes in canine peripheral blood show characteristics of natural killer cells.
J Leukoc Biol. 84: 1501-10. -
Hai, M. et al. (2008) Potential genotoxicity from integration sites in CLAD dogs treated successfully with gammaretroviral vector-mediated gene therapy.
Gene Ther. 15: 1067-71. -
Fellman, C.L. et al. (2011) Cyclosporine A affects the in vitro expression of T cell activation-related molecules and cytokines in dogs.
Vet Immunol Immunopathol. 140: 175-80. -
Watabe, A. et al. (2011) Alterations of lymphocyte subpopulations in healthy dogs with aging and in dogs with cancer.
Vet Immunol Immunopathol. 142: 189-200. -
Machado, G.F. et al. (2011) Intravascular Lymphomatosis in the Central Nervous System of Dogs: Immunohistochemical Investigation in Two Cases
Braz J Vet Pathol 4: 47-51. -
Maiolini, A. et al. (2012) Toll-like receptors 4 and 9 are responsible for the maintenance of the inflammatory reaction in canine steroid-responsive meningitis-arteritis, a large animal model for neutrophilic meningitis.
J Neuroinflammation. 9: 226. -
Villaescusa A et al. (2012) Evaluation of peripheral blood lymphocyte subsets in family-owned dogs naturally infected by Ehrlichia canis.
Comp Immunol Microbiol Infect Dis. 35 (4): 391-6. -
Aricò, A. et al. (2013) The role of vascular endothelial growth factor and matrix metalloproteinases in canine lymphoma: in vivo and in vitro study.
BMC Vet Res. 9: 94. -
Michael, H.T. et al. (2013) Isolation and characterization of canine natural killer cells.
Vet Immunol Immunopathol. 155 (3): 211-7. -
Duz, A.L. et al. (2014) The TcI and TcII Trypanosoma cruzi experimental infections induce distinct immune responses and cardiac fibrosis in dogs.
Mem Inst Oswaldo Cruz. 109 (8): 1005-13. -
Aresu, L. et al. (2014) VEGF and MMP-9: biomarkers for canine lymphoma.
Vet Comp Oncol. 12: 29-36. -
Perosso, J. et al. (2014) Alteration of sFAS and sFAS ligand expression during canine visceral leishmaniosis.
Vet Parasitol. 205 (3-4): 417-23. -
Gelain, M.E. et al. (2014) CD44 in canine leukemia: analysis of mRNA and protein expression in peripheral blood.
Vet Immunol Immunopathol. 159 (1-2): 91-6. -
Constantinoiu, C.C. et al. (2015) Mucosal tolerance of the hookworm Ancylostoma caninum in the gut of naturally infected wild dogs.
Parasite Immunol. 37 (10): 510-20. -
Grøndahl-Rosado C et al. (2015) NCR1+ cells in dogs show phenotypic characteristics of natural killer cells.
Vet Res Commun. 39 (1): 19-30. -
Miller, J. et al. (2015) Humoral and Cellular Immune Response in Canine Hypothyroidism.
J Comp Pathol. 153 (1): 28-37. -
Mie, K. et al. (2016) Change in peripheral blood lymphocyte count in dogs following adoptive immunotherapy using lymphokine-activated T killer cells combined with palliative tumor resection.
Vet Immunol Immunopathol. 177: 58-63. -
Schaut, R.G. et al. (2016) Recovery of antigen-specific T cell responses from dogs infected with Leishmania (L.) infantum by use of vaccine associated TLR-agonist adjuvant.
Vaccine. 34 (44): 5225-34. -
Riondato, F. et al. (2016) Analytical and diagnostic validation of a flow cytometric strategy to quantify blood and marrow infiltration in dogs with large B-cell lymphoma.
Cytometry B Clin Cytom. 90 (6): 525-530. -
Bonnefont-Rebeix, C. et al. (2016) Characterization of a novel canine T-cell line established from a spontaneously occurring aggressive T-cell lymphoma with large granular cell morphology.
Immunobiology. 221 (1): 12-22. -
Schaut, R.G. et al. (2016) Regulatory IgDhi B Cells Suppress T Cell Function via IL-10 and PD-L1 during Progressive Visceral Leishmaniasis.
J Immunol. 196 (10): 4100-9. -
McGill, J.L. et al. (2016) Vaccination with an Attenuated Mutant of Ehrlichia chaffeensis Induces Pathogen-Specific CD4+ T Cell Immunity and Protection from Tick-Transmitted Wild-Type Challenge in the Canine Host.
PLoS One. 11 (2): e0148229. -
Martins, G.C. et al. (2018) Clinical-pathological and immunological biomarkers in dogs with atopic dermatitis.
Vet Immunol Immunopathol. 205: 58-64. -
Schmidli, M.R. et al. (2018) Inflammatory pattern of the infrapatellar fat pad in dogs with canine cruciate ligament disease.
BMC Vet Res. 14 (1): 161. -
Akiyama, S. et al. (2019) Th17 cells increase during maturation in peripheral blood of healthy dogs.
Vet Immunol Immunopathol. 209: 17-21. -
Aguiar-Soares, R.D.O. et al. (2020) Phase I and II Clinical Trial Comparing the LBSap, Leishmune(®), and Leish-Tec(®) Vaccines against Canine Visceral Leishmaniasis.
Vaccines (Basel). 8 (4): 690. -
Sayag, D. et al. (2020) Proof-of-concept study: Evaluation of plasma and urinary electrolytes as markers of response to L-asparaginase therapy in dogs with high-grade lymphoma.
Vet Clin Pathol. 49 (3): 476-83. -
Enciso, N. et al. (2020) Regenerative potential of allogeneic adipose tissue-derived mesenchymal cells in canine cutaneous wounds.
Acta Vet Scand. 62 (1): 13. -
Marchetti, C. et al. (2020) Profile of gamma-delta (γδ) T lymphocytes in the peripheral blood of crossbreed dogs during stages of life and implication in aging.
BMC Vet Res. 16 (1): 278. -
Lee, J. et al. (2021) Canine Natural Killer Cell-Derived Exosomes Exhibit Antitumor Activity in a Mouse Model of Canine Mammary Tumor.
Biomed Res Int. 2021: 6690704. -
Rotolo, A. et al. (2021) Genetic re-direction of canine primary T cells for clinical trial use in pet dogs with spontaneous cancer
STAR Protocols. 2 (4): 100905. -
Grudzien, M. et al. (2021) A newly established canine NK-type cell line and its cytotoxic properties.
Vet Comp Oncol. 19 (3): 567-77. -
Yang, Y. et al. (2021) Canine Multicentric Large B Cell Lymphoma with Increased Mott Cells Diagnosed by Flow Cytometry
Journal of Veterinary Clinics. 38 (1): 36-40. -
Lee, S.H. et al. (2021) Safety and immunological effects of recombinant canine IL-15 in dogs.
Cytokine. 148: 155599. -
Knebel, A. et al. (2021) Measurement of canine Th17 cells by flow cytometry.
Vet Immunol Immunopathol. 243: 110366. -
Troupel, T. et al. (2022) Generalised idiopathic polymyositis mimicking masticatory myositis in a dog
Vety Rec Case Rep. 10: e452. -
do Prado Duzanski, A. et al. (2022) Cell-mediated immunity and expression of MHC class I and class II molecules in dogs naturally infected by canine transmissible venereal tumor: Is there complete spontaneous regression outside the experimental CTVT?
Research in Veterinary Science. 145: 193-204. -
Konno, H. et al. (2022) An experimental challenge model for Leishmania donovani in beagle dogs, showing a similar pattern of parasite burden in the peripheral blood and liver.
Parasitol Res. 121 (12): 3569-3579. -
Rotolo, A. et al. (2023) Unedited allogeneic iNKT cells show extended persistence in MHC-mismatched canine recipients.
Cell Rep Med. 4 (10): 101241. -
Wesolowski, M. et al. (2023) Long-term changes of Th17 and regulatory T cells in peripheral blood of dogs with spinal cord injury after intervertebral disc herniation.
BMC Vet Res. 19 (1): 90. -
Yamauchi, A. et al. (2023) Negative Influence of Aging on Differentiation and Proliferation of CD8(+) T-Cells in Dogs.
Vet Sci. 10 (9): 541. -
Martini, V. et al. (2018) A retrospective study of flow cytometric characterization of suspected extranodal lymphomas in dogs.
J Vet Diagn Invest. 30 (6): 830-6. -
Archer, T.M. et al. (2018) In vivo effects of aspirin and cyclosporine on regulatory T cells and T-cell cytokine production in healthy dogs.
Vet Immunol Immunopathol. 197: 63-8. -
Lee, G.W. et al. (2021) Case Report: Long-Term Survival of a Dog With Chronic Lymphocytic Leukemia Treated With Chlorambucil, Prednisolone, and Imatinib.
Front Vet Sci. 8: 625527. -
Sainz, Á. et al. (2021) Effect of chemically modified tetracycline-8 (CMT-8) on hematology, blood chemistry, cytokines and peripheral blood lymphocyte subsets of healthy dogs.
Res Vet Sci. 136: 200-8. -
Hughes, K. et al. (2024) Canine T zone lymphoma is a tumor of mature, previously activated αβ T cells
Vet Immunol Immunopathol. 110725. -
Anthonyraj, S. et al. (2024) Chicory root powder included as a prebiotic in different cereal-based diets for dogs: Influences on gut health, metabolic and immunological status
Bioactive Carbohydrates and Dietary Fibre. : 100414. [Pre-proof] -
Sheng, R. et al. (2023) Prognostic significance of CD25 expression in dogs with a noninvasive diagnosis of B-cell lymphoma treated with CHOP chemotherapy.
Vet Comp Oncol. 21 (1): 28-35. -
Miguelena Chamorro, B. et al. (2023) Characterization of Canine Peyer's Patches by Multidimensional Analysis: Insights from Immunofluorescence, Flow Cytometry, and Single-Cell RNA Sequencing.
Immunohorizons. 7 (11): 788-805.
- RRID
- AB_2291174
- UniProt
- P27597
- Entrez Gene
- CD3E
- GO Terms
- GO:0016021 integral to membrane
- GO:0004888 transmembrane receptor activity
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Dog ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up