Green Fluorescent Protein antibody


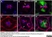
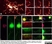




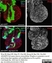
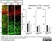

Sheep anti Green Fluorescent Protein
- Product Type
- Polyclonal Antibody
- Isotype
- Polyclonal IgG
- Specificity
- Green Fluorescent Protein
| Sheep anti Green Fluorescent Protein antibody recognizes green fluorescent protein (GFP), a ~27 kDa protein derived from the jellyfish Aequorea victoria. GFP fluoresces green (509nm) when excited by blue light (395nm) and is commonly used as a marker of gene expression. |
- Product Form
- Purified IgG - liquid
- Preparation
- Purified IgG prepared by affinity chromatography on Protein G.
- Buffer Solution
- Phosphate buffered saline
- Preservative Stabilisers
- 0.09% Sodium Azide (NaN3)
- Immunogen
- Green fluorescent protein from Aequorea victoria.
- Approx. Protein Concentrations
- IgG concentration 5.0 mg/ml
- Regulatory
- For research purposes only
- Guarantee
- 12 months from date of despatch
Avoid repeated freezing and thawing as this may denature the antibody. Storage in frost-free freezers is not recommended.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| ELISA | |||
| Immunofluorescence |
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Rabbit anti Sheep IgG (H/L):Biotin | 5184-2304 | C E F WB | 1.5 ml |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Rabbit anti Sheep IgG (H/L):Biotin | ||||||
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Rabbit anti mCherry | AHP2326 | WB | 50 µg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Rabbit anti mCherry | ||||||
| Rabbit anti Red Fluorescent Protein | AHP2987 | WB | 50 µg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Rabbit anti Red Fluorescent Protein | ||||||
| Mouse anti mCherry | MCA6020 | WB | 50 µg |
|
Log in | ||
| List Price | Your Price | ||||||
|
|
Log in | ||||||
| Description | Mouse anti mCherry | ||||||
References for Green Fluorescent Protein antibody
-
Mason, D.E. et al. (1987) Arterial blood gas tensions in the horse during recovery from anesthesia.
J Am Vet Med Assoc. 190 (8): 989-94. -
Soza-Ried, C. et al. (2008) Maintenance of thymic epithelial phenotype requires extrinsic signals in mouse and zebrafish.
J Immunol. 181: 5272-7. -
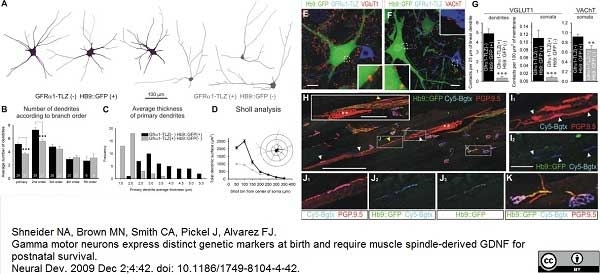
Shneider, N.A. et al. (2009) Gamma motor neurons express distinct genetic markers at birth and require muscle spindle-derived GDNF for postnatal survival.
Neural Dev. 4: 42. -
Siembab, V.C. et al. (2010) Target selection of proprioceptive and motor axon synapses on neonatal V1-derived Ia inhibitory interneurons and Renshaw cells.
J Comp Neurol. 518: 4675-701. -
Collins, R.T. et al. (2010) MAZe: a tool for mosaic analysis of gene function in zebrafish.
Nat Methods. 7: 219-23. -
Wu, L. et al. (2011) Properties of a distinct subpopulation of GABAergic commissural interneurons that are part of the locomotor circuitry in the neonatal spinal cord.
J Neurosci. 31 (13): 4821-33. -
Lopez, K.A. et al. (2011) Convection-enhanced delivery of topotecan into a PDGF-driven model of glioblastoma prolongs survival and ablates both tumor-initiating cells and recruited glial progenitors.
Cancer Res. 71: 3963-71. -
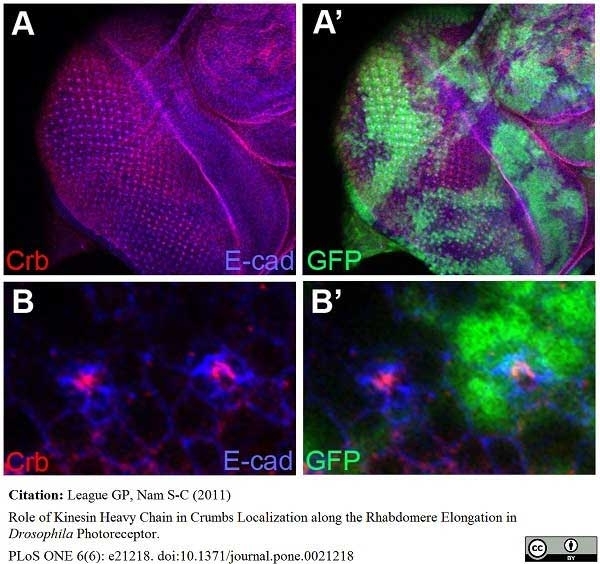
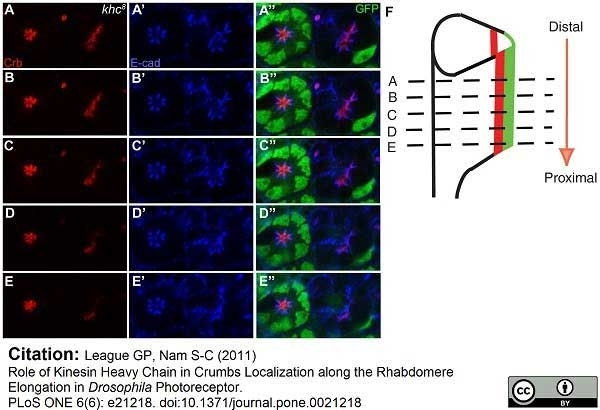
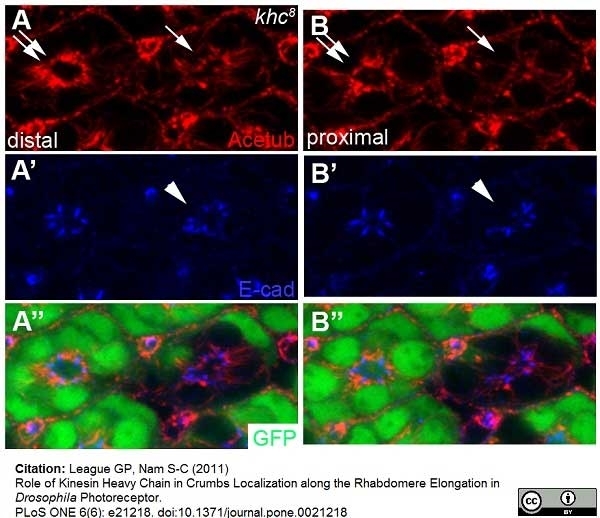
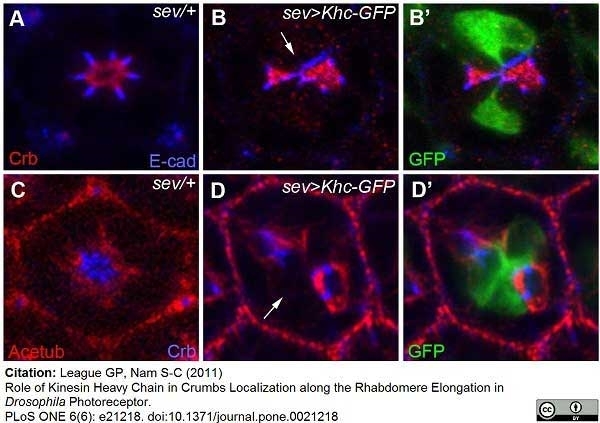
League, G.P. and Nam, S.C. (2011) Role of kinesin heavy chain in Crumbs localization along the rhabdomere elongation in Drosophila photoreceptor.
PLoS One. 6:e21218.
View The Latest Product References
-
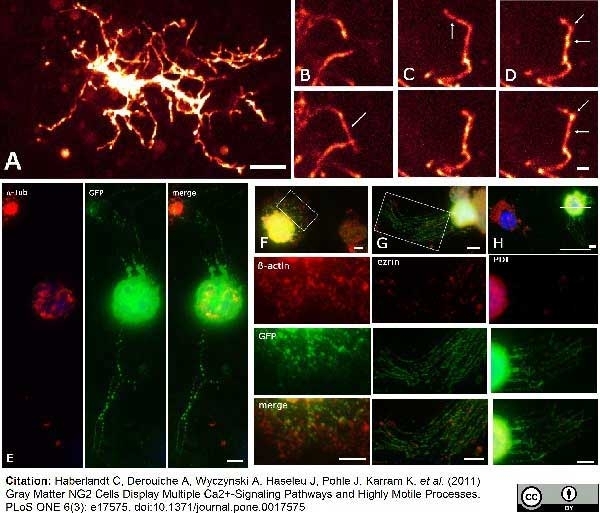
Haberlandt, C. et al. (2011) Gray matter NG2 cells display multiple Ca2+-signaling pathways and highly motile processes.
PLoS One. 6: e17575. -
Srinivasan, S. et al. (2012) The receptor tyrosine phosphatase Lar regulates adhesion between Drosophila male germline stem cells and the niche.
Development. 139: 1381-90. -
Cheung, L.S. et al. (2013) Dynamic model for the coordination of two enhancers of broad by EGFR signaling.
Proc Natl Acad Sci U S A. 110: 17939-44. -
Li, X. et al. (2013) Temporal patterning of Drosophila medulla neuroblasts controls neural fates.
Nature. 498: 456-62. -
Behnia, R. et al. (2014) Processing properties of ON and OFF pathways for Drosophila motion detection.
Nature. 512: 427-30. -
de Nooij, J.C. et al. (2015) The PDZ-domain protein Whirlin facilitates mechanosensory signaling in mammalian proprioceptors.
J Neurosci. 35 (7): 3073-84. -
Scotti, M. et al. (2015) A Hoxa13:Cre mouse strain for conditional gene manipulation in developing limb, hindgut, and urogenital system.
Genesis. 53 (6): 366-76. -
Sun, G.J. et al. (2015) Latent tri-lineage potential of adult hippocampal neural stem cells revealed by Nf1 inactivation.
Nat Neurosci. 18 (12): 1722-4. -
Crouch, E.E. et al. (2015) Regional and stage-specific effects of prospectively purified vascular cells on the adult V-SVZ neural stem cell lineage.
J Neurosci. 35 (11): 4528-39. -
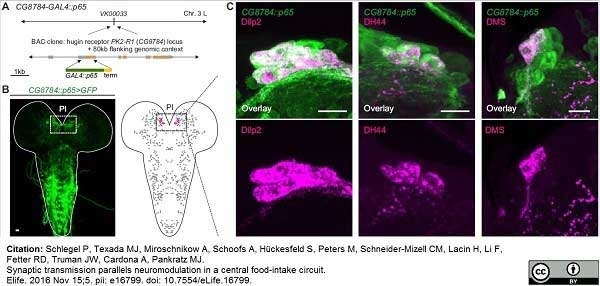
Schlegel, P. et al. (2016) Synaptic transmission parallels neuromodulation in a central food-intake circuit.
eLife 2016;10.7554/eLife.16799 -
Gushchina, S. et al. (2018) Increased expression of colony-stimulating factor-1 in mouse spinal cord with experimental autoimmune encephalomyelitis correlates with microglial activation and neuronal loss.
Glia. 66 (10): 2108-25. -
Sagner, A. et al. (2018) Olig2 and Hes regulatory dynamics during motor neuron differentiation revealed by single cell transcriptomics.
PLoS Biol. 16 (2): e2003127. -
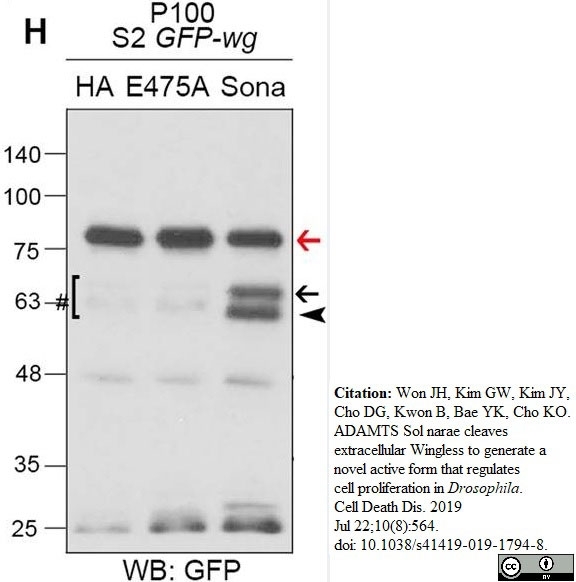
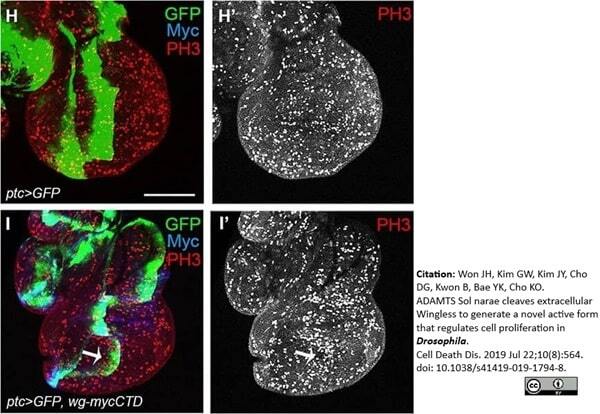
Won, J.H. et al. (2019) ADAMTS Sol narae cleaves extracellular Wingless to generate a novel active form that regulates cell proliferation in Drosophila.
Cell Death Dis. 10 (8): 564. -
Balaskas, N. et al. (2019) Positional Strategies for Connection Specificity and Synaptic Organization in Spinal Sensory-Motor Circuits.
Neuron. 102 (6): 1143-1156.e4. -
Zhang, R. et al. (2019) Id4 Downstream of Notch2 Maintains Neural Stem Cell Quiescence in the Adult Hippocampus.
Cell Rep. 28 (6): 1485-1498.e6. -
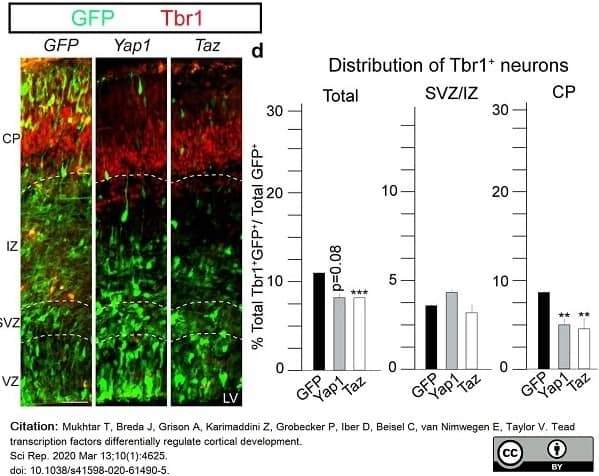
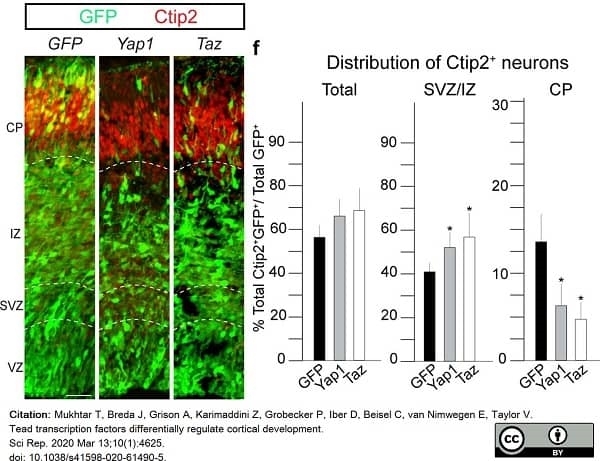
Mukhtar, T. et al. (2020) Tead transcription factors differentially regulate cortical development.
Sci Rep. 10 (1): 4625. -
Heath, S.L. et al. (2020) Circuit Mechanisms Underlying Chromatic Encoding in Drosophila Photoreceptors.
Curr Biol. 30 (2): 264-275.e8. -
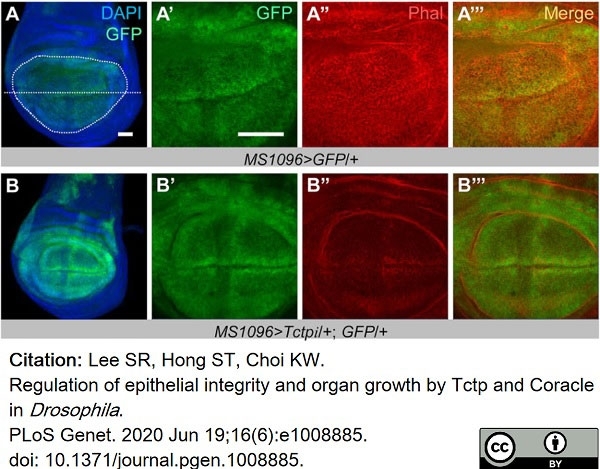
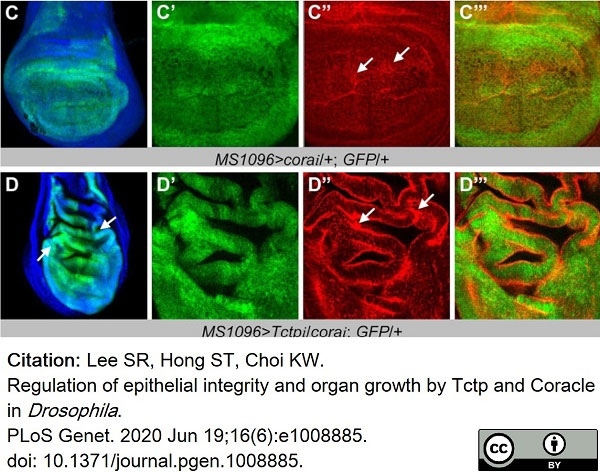
Lee, S.R. et al. (2020) Regulation of epithelial integrity and organ growth by Tctp and Coracle in Drosophila..
PLoS Genet. 16 (6): e1008885. -
Choquet, C. et al. (2020) Nkx2-5 defines distinct scaffold and recruitment phases during formation of the murine cardiac Purkinje fiber network.
Nat Commun. 11 (1): 5300. -
Del Valle Rodríguez, A. et al. (2020) A network approach to analyze neuronal lineage and layer innervation in the Drosophila optic lobes.
PLoS One. 15 (2): e0227897. -
Poupault, C. et al. (2021) A combinatorial cis-regulatory logic restricts color-sensing Rhodopsins to specific photoreceptor subsets in Drosophila..
PLoS Genet. 17 (6): e1009613. -
Oliver, K.M. et al. (2021) Molecular correlates of muscle spindle and Golgi tendon organ afferents.
Nat Commun. 12 (1): 1451. -
Zhu, H. et al. (2022) A comprehensive temporal patterning gene network in Drosophila medulla. neuroblasts revealed by single-cell RNA sequencing.
Nat Commun. 13 (1): 1247. -
Parmigiani, E. & Giachino, C. (2022) Genetic Inactivation of Notch1 Synergizes with Loss of Trp53 to Induce Tumor Formation in the Adult Mouse Forebrain.
Cancers (Basel). 14 (21): 5409. -
Zhang, Y. et al. (2023) Notch-dependent binary fate choice regulates the Netrin pathway to control axon guidance of Drosophila visual projection neurons.
Cell Rep. 42 (3): 112143. -
Liau, E.S. et al. (2023) Single-cell transcriptomic analysis reveals diversity within mammalian spinal motor neurons.
Nat Commun. 14 (1): 46. -
Zhang, Y. et al. (2023) Axon targeting of Drosophila medulla projection neurons requires diffusible Netrin and is coordinated with neuroblast temporal patterning.
Cell Rep. 42 (3): 112144. -
Tsuboguchi, S. et al. (2023) TDP-43 differentially propagates to induce antero- and retrograde degeneration in the corticospinal circuits in mouse focal ALS models.
Acta Neuropathol. 146 (4): 611-29. -
Klima, M.L. et al. (2023) Anti-inflammatory effects of hunger are transmitted to the periphery via projection-specific AgRP circuits
Cell Reports. 42 (11): 113338. -
Hoang, P.T. et al. (2018) Subtype Diversification and Synaptic Specificity of Stem Cell-Derived Spinal Interneurons.
Neuron. 100 (1): 135-149.e7. -
Mukhtar, T. et al. (2022) Temporal and sequential transcriptional dynamics define lineage shifts in corticogenesis.
EMBO J. 41 (24): e111132. -
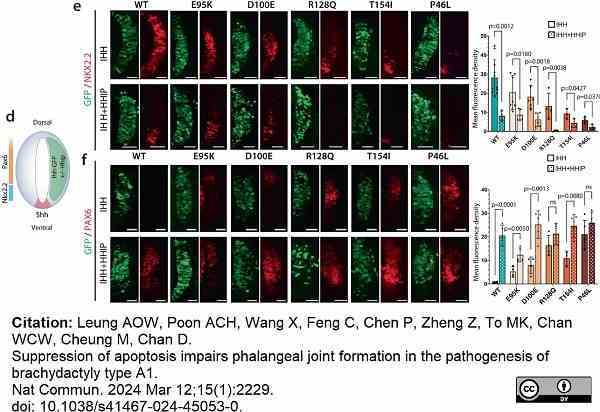
Leung, A.O.W. et al. (2024) Suppression of apoptosis impairs phalangeal joint formation in the pathogenesis of brachydactyly type A1.
Nat Commun. 15 (1): 2229.
Further Reading
-
Adams, K.L. et al. (2015) Foxp1-mediated programming of limb-innervating motor neurons from mouse and human embryonic stem cells.
Nat Commun. 6: 6778.
- RRID
- AB_619712
- UniProt
- P42212
- GO Terms
- GO:0006091 generation of precursor metabolites and energy
- GO:0008218 bioluminescence
- GO:0018298 protein-chromophore linkage
4745-1051
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
Always be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up