Fluorochromes and Light in Flow Cytometry
Author: Mike Blundell | Reviewer: Chloe Fenton
What Are Fluorophores and How Do They Work in Flow Cytometry
Fluorophores are fluorescent markers used to detect the expression of cellular molecules such as proteins or nucleic acids. They functionally accept light energy (for example, from a laser) at a given wavelength and re-emit it at a longer wavelength. These two processes are called excitation and emission. Emission follows excitation extremely rapidly, commonly in nanoseconds and is known as fluorescence. Before considering the different types of fluorophores available for flow cytometry, it is necessary to understand the principles of light absorbance and emission.
Properties of Light Relevant to Fluorescence
Light is a form of electromagnetic energy that travels in waves. These waves have both a frequency and length, the latter of which determines the color of the light. The light that can be visualized by the human eye represents a narrow wavelength band (380-700 nm) between ultraviolet (UV) and infrared (IR) radiation (Figure 7). Sunlight, for example, contains UV and IR light that, although invisible to the eye can be felt as warmth on the skin and measured scientifically using photodetectors. The visible spectrum can be further subdivided according to color, red, orange, yellow, green, blue and violet. Red light has a longer wavelength and lower energy, whereas violet light has a shorter wavelength and higher energy.
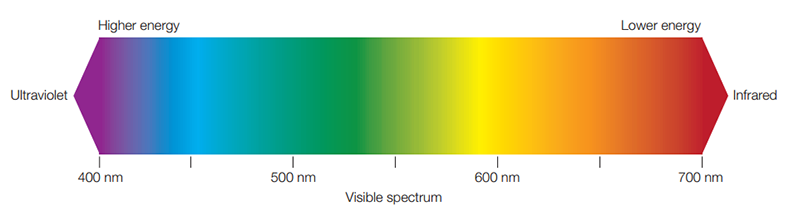
Fig. 7. The electromagnetic spectrum
Flow Cytometry Basics Guide Download
Get your own copy to peruse at your leisure
Download our updated Flow Cytometry Basics Guide to have practical advice, best practice examples, and a basic overview of all the important flow cytometry principles in one handy location.
Resources
Bio-Rad is committed to helping you succeed in Flow Cytometry by sharing knowledge and best practices from our experts. Below are some extremely useful resources to enhance your journey of discovery and support your success.
