VEGF-A


Recombinant Human VEGF-A
- Product Type
- Recombinant Protein
- Specificity
- VEGF-A
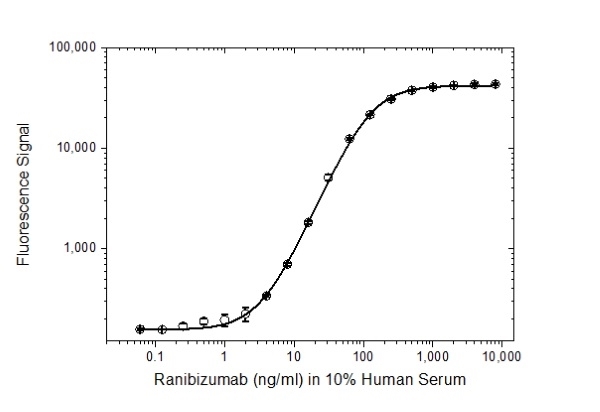
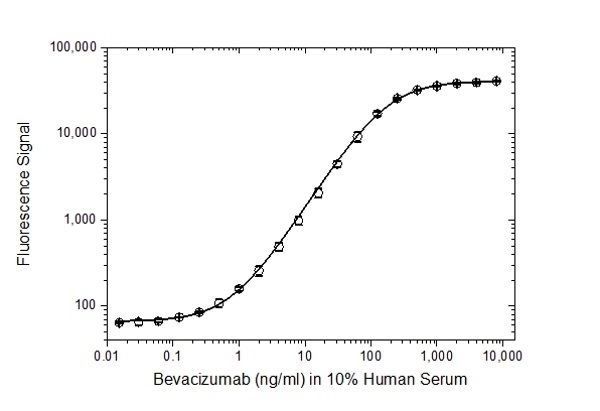
| Recombinant Human VEGF-A Vascular endothelial growth factor alpha forms a homodimer and is a potent stimulator of angiogenesis of both normal and cancerous cells. It acts as a regulator of vasculogenesis, angiogenesis and endothelial cell growth. VEGF-A is secreted by many different cell types such as endothelial cells (Nissen et al. 1998), smooth muscle cells (Brogi et al. 1994), neutrophils (Gaudry et al. 1997), platelets (Banks et al. 1998), macrophages and 60% of all tumours ( The sequence of this recombinant protein product is amino acid (aa) 27 - aa 141 + CDKPRR but deficient from aa 142-226, and it is closest to isoform 121. Isoform 121 and 111 lack exons 6 and 7, and for this reason neither of these isoforms bind to the extracellular matrix (Krilleke et al. 2017). This product has been demonstrated for use, in a pharmacokinetic (PK) antigen capture ELISA with the monoclonal antibody drugs ranibizumab and bevacizumab using anti-ranibizumab antibodies for detection. |
- Target Species
- Human
- Product Form
- Purified recombinant protein - lyophilized
- Reconstitution
- Centrifuge vial prior to reconstitution. Reconstitute to 0.5 mg/ml by adding 0.4 ml ddH2O.
Care should be taken during reconstitution as the protein may appear as a film at the bottom of the vial. Mix gently after reconstitution. Do not vortex. - Preparation
- Recombinant protein expressed in E. coli and purified by affinity chromatography
- Source
- E.coli
- Buffer Solution
- 20 mM Phosphate Buffer, 0.1 M Sodium Chloride
- Preservative Stabilisers
- 1% Trehalose
- Purity
- ≥90% determined by SDS-PAGE under reducing conditions and visualized by coomassie blue staining
- Approx. Protein Concentrations
- 0.5 mg/ml after reconstitution
- Protein Molecular Weight
- Predicted 14 kDa. The apparent molecular mass of the human VEGF-A monomer is approximately 17 kDa determined by SDS-PAGE under reducing conditions, and 17 kDa (monomer) and 30 kDa (dimer) under non-reducing conditions.
- Regulatory
- For research purposes only
- Guarantee
- Guaranteed for 3 months from the date of reconstitution or until the date of expiry, whichever comes first. Please see label for expiry date.
This product should be stored undiluted.
Storage in frost-free freezers is not recommended. Avoid repeated freezing and thawing as this may denature the protein.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| ELISA |
| Description | Product Code | Applications | Pack Size | List Price | Your Price | Quantity | |
|---|---|---|---|---|---|---|---|
| Human anti Ranibizumab (Drug/Target Complex) | HCA304 | E | 0.1 mg | Log in | |||
| List Price | Your Price | ||||||
| Log in | |||||||
| Description | Human anti Ranibizumab (Drug/Target Complex) | ||||||
| Human anti Ranibizumab | HCA307 | E | 0.1 mg | Log in | |||
| List Price | Your Price | ||||||
| Log in | |||||||
| Description | Human anti Ranibizumab | ||||||
Further Reading
-
Krilleke, D. et al. (2007) Molecular mapping and functional characterization of the VEGF164 heparin-binding domain.
J Biol Chem. 282 (38): 28045-56. -
Berse, B. et al. (1992) Vascular permeability factor (vascular endothelial growth factor) gene is expressed differentially in normal tissues, macrophages, and tumors.
Mol Biol Cell. 3 (2): 211-20. -
Gaudry, M. et al. (1997) Intracellular pool of vascular endothelial growth factor in human neutrophils.
Blood. 90 (10): 4153-61. -
Banks, R.E. et al. (1998) Release of the angiogenic cytokine vascular endothelial growth factor (VEGF) from platelets: significance for VEGF measurements and cancer biology.
Br J Cancer. 77 (6): 956-64. -
Brogi, E. et al. (1994) Indirect angiogenic cytokines upregulate VEGF and bFGF gene expression in vascular smooth muscle cells, whereas hypoxia upregulates VEGF expression only.
Circulation. 90 (2): 649-52. -
Nissen, N.N. et al. (1998) Vascular endothelial growth factor mediates angiogenic activity during the proliferative phase of wound healing.
Am J Pathol. 152 (6): 1445-52.
- UniProt
- P15692
PHP293
If you cannot find the batch/lot you are looking for please contact our technical support team for assistance.
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Human ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up