EGF
Recombinant Human EGF
- Product Type
- Recombinant Protein
- Specificity
- EGF
Filter by Application:
TC Reset| Recombinant human epidermal growth factor is 6.2kDa globular protein comprosed of 53 amino acids. EGF is a polypeptide growth factor which stimulates the proliferation of a wide range of epidermal and epithelial cells. |
- Target Species
- Human
- Species Cross-Reactivity
-
Target Species Cross Reactivity Mouse - N.B. Antibody reactivity and working conditions may vary between species.
- Product Form
- Purified recombinant protein - lyophilized
- Reconstitution
- Reconstitute with 0.5ml distilled water
Care should be taken during reconstitution as the protein may appear as a film at the bottom of the vial. Bio-Rad recommend that the vial is gently mixed after reconstitution. For extended storage, the addition of 5% trehalose is recommended - Preparation
- Purified recombinant human EGF expressed in E. coli
- Source
- E.coli
- Preservative Stabilisers
- None present
- Carrier Free
- Yes
- Activity
- 1 x 107 units/mg
- Purity
- >98% by SDS PAGE and HPLC analysis
- Approx. Protein Concentrations
- 1.0 mg/ml after reconstitution.
- Protein Molecular Weight
- 6.2 kD (53 Amino acid sequence)
- Endotoxin Level
- < 0.1 ng/ug
- Regulatory
- For research purposes only
- Guarantee
- Guaranteed for 3 months from the date of reconstitution or until the date of expiry, whichever comes first. Please see label for expiry date.
This product should be stored undiluted.
Storage in frost free freezers is not recommended. Avoid repeated freezing and thawing as this may denature the protein. Should this product contain a precipitate we recommend microcentrifugation before use.
| Application Name | Verified | Min Dilution | Max Dilution |
|---|---|---|---|
| ELISA | 0.2 | 0.4ng/well | |
| Functional Assays | 0.5 | 25ng/ml | |
| Western Blotting | 1.5 | 3.0ng/lane |
- ELISA
- Recombinant human EGF may be used as athe standard in ELISA applications with either a purified human EGF antibody (AHP767) or a biotinylated human EGF antibody (AHP767B).
- Western Blotting
- Recombinant human EGF may be used as the positive control for Wester Blotting application with either a purified human EGF antibody (AHP767) or a biotinylated human EGF antibody (AHP767B)
References for EGF
-
Tomlins, C. & Storey, A. (2010) Cutaneous HPV5 E6 causes increased expression of Osteoprotegerin and Interleukin 6 which contribute to evasion of UV-induced apoptosis.
Carcinogenesis. 31 (12): 2155-64. -
Wray, H. et al. (2012) α6 Integrin and CD44 enrich for a primary keratinocyte population that displays resistance to UV-induced apoptosis.
PLoS One. 7 (10): e46968. -
Chen, W. et al. (2016) Tissue Kallikrein Inhibitors Based on the Sunflower Trypsin Inhibitor Scaffold - A Potential Therapeutic Intervention for Skin Diseases.
PLoS One. 11 (11): e0166268. -
Zhang, X. et al. (2015) Wnt signaling regulates the stemness of lung cancer stem cells and its inhibitors exert anticancer effect on lung cancer SPC-A1 cells.
Med Oncol. 32 (4): 95. -
Roth, K. et al. (2021) Clinically relevant aberrant Filip1l DNA methylation detected in a murine model of cutaneous squamous cell carcinoma.
EBioMedicine. 67: 103383. -
Inman, G.J. et al. (2018) The genomic landscape of cutaneous SCC reveals drivers and a novel azathioprine associated mutational signature.
Nat Commun. 9 (1): 3667. -
Aiderus, A. et al. (2021) Transposon mutagenesis identifies cooperating genetic drivers during keratinocyte transformation and cutaneous squamous cell carcinoma progression.
PLoS Genet. 17 (8): e1009094. -
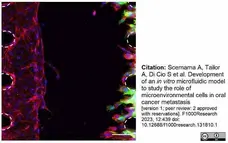
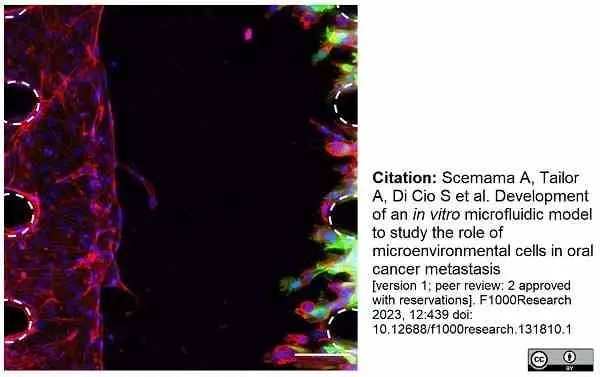
Scemama, A. et al. (2023) Development of an in vitro microfluidic model to study the role of microenvironmental cells in oral cancer metastasis
F1000Research. 12: 439.
View The Latest Product References
-
Zhang, Y. et al. (2020) Sulfoxythiocarbamate S-4 inhibits HSP90 in human cutaneous squamous cell carcinoma cells.
Eur J Pharmacol. 889: 173609.
- Synonyms
- Epidermal Growth Factor
- UniProt
- P01133
- Entrez Gene
- EGF
- GO Terms
- GO:0005886 plasma membrane
- GO:0006260 DNA replication
- GO:0005509 calcium ion binding
- GO:0001525 angiogenesis
- GO:0002576 platelet degranulation
- GO:0016021 integral to membrane
- GO:0005154 epidermal growth factor receptor binding
- GO:0030297 transmembrane receptor protein tyrosine kinase activator activity
- GO:0045741 positive regulation of epidermal growth factor receptor activity
- View More GO Terms
- GO:0007173 epidermal growth factor receptor signaling pathway
- GO:0008083 growth factor activity
- GO:0030168 platelet activation
- GO:0031093 platelet alpha granule lumen
- GO:0035413 positive regulation of catenin import into nucleus
- GO:0042059 negative regulation of epidermal growth factor receptor signaling pathway
- GO:0043406 positive regulation of MAP kinase activity
- GO:0045840 positive regulation of mitosis
- GO:0051048 negative regulation of secretion
- GO:0090279 regulation of calcium ion import
- GO:2000008 regulation of protein localization at cell surface
Please Note: All Products are "FOR RESEARCH PURPOSES ONLY"
View all Anti-Human ProductsAlways be the first to know.
When we launch new products and resources to help you achieve more in the lab.
Yes, sign me up